| Identification | More | [Name]
Acetophenone | [CAS]
98-86-2 | [Synonyms]
1-Phenylethanone
ACETOPHENONE
ACETPHENONE
ACETYLBENZENE
AKOS BBS-00003215
alpha-Acetophenone
BENZOYL METHIDE
FEMA 2009
HYPNONE
METHYL PHENYL KETONE
PHENYL METHYL KETONE
1-Phenyl-1-ethanone
1-phenyl-ethanon
1-Phenylethanone (acetophenone)
1-phenylethanone[qr]
Acetofenon
Acetophenon
acetophenon[qr]
acetyl-benzen
acetylbenzene[qr] | [EINECS(EC#)]
202-708-7 | [Molecular Formula]
C8H8O | [MDL Number]
MFCD00008724 | [Molecular Weight]
120.15 | [MOL File]
98-86-2.mol |
| Chemical Properties | Back Directory | [Description]
Acetophenone is the simplest aromatic ketone and is a clear liquid/crystal and very slightly
soluble in water with a sweet pungent taste and odour resembling oranges. It is used as
a polymerisation catalyst for the manufacture of olefins. Acetophenone is used in perfumery
as a fragrance ingredient in soaps, detergents, creams, lotions, and perfumes; as a
flavouring agent in foods, non-alcoholic beverages, and tobacco; as a specialty solvent for plastics and resins; as a catalyst for the polymerisation of olefins; and as a photosensitiser
in organic syntheses. Acetophenone is a raw material for the synthesis of some pharmaceuticals
and is also listed as an approved excipient by the U.S. FDA. Acetophenone occurs
naturally in many foods such as apple, apricot, banana, and beef. Acetophenone has been detected in ambient
air and drinking water; exposure of the general public may occur through the inhalation
of contaminated air or the consumption of contaminated water. It is highly flammable
and will get easily ignited by heat, sparks, or flames, and the vapours may form explosive
mixtures with air. | [Appearance]
Acetophenone is a colorless, oily liquid with a sweet, floral odor. | [Melting point ]
19-20 °C (lit.) | [Boiling point ]
202 °C (lit.) | [density ]
1.03 g/mL at 25 °C(lit.)
| [vapor density ]
4.1 (vs air)
| [vapor pressure ]
0.45 mm Hg ( 25 °C)
| [FEMA ]
2009 | [refractive index ]
n20/D 1.534(lit.)
| [Fp ]
180 °F
| [storage temp. ]
2-8°C | [solubility ]
6.1g/l | [form ]
Liquid | [color ]
Clear colorless to light yellow | [Odor]
Pungent, floral odor | [Relative polarity]
4.4 | [Stability:]
Stable. Incompatible with strong oxidizing agents, strong bases, strong reducing agents. Combustible. | [explosive limit]
1.4-5.2%(V) | [Odor Type]
floral | [Water Solubility ]
5.5 g/L (20 ºC) | [JECFA Number]
806 | [Merck ]
14,73 | [BRN ]
605842 | [Dielectric constant]
17.4(25℃) | [Exposure limits]
No exposure limits are set. The health hazard
from exposure to this compound should be
low, due to its low vapor pressure and low
toxicity. | [InChIKey]
KWOLFJPFCHCOCG-UHFFFAOYSA-N | [LogP]
1.65 at 20℃ | [CAS DataBase Reference]
98-86-2(CAS DataBase Reference) | [NIST Chemistry Reference]
Acetophenone(98-86-2) | [EPA Substance Registry System]
98-86-2(EPA Substance) |
| Safety Data | Back Directory | [Hazard Codes ]
Xn,T,F | [Risk Statements ]
R22:Harmful if swallowed.
R36:Irritating to the eyes.
R63:Possible risk of harm to the unborn child.
R43:May cause sensitization by skin contact.
R36/37/38:Irritating to eyes, respiratory system and skin .
R23/24/25:Toxic by inhalation, in contact with skin and if swallowed .
R45:May cause cancer.
R39/23/24/25:Toxic: danger of very serious irreversible effects through inhalation, in contact with skin and if swallowed .
R11:Highly Flammable. | [Safety Statements ]
S26:In case of contact with eyes, rinse immediately with plenty of water and seek medical advice .
S36/37:Wear suitable protective clothing and gloves .
S24/25:Avoid contact with skin and eyes .
S23:Do not breathe gas/fumes/vapor/spray (appropriate wording to be specified by the manufacturer) .
S53:Avoid exposure-obtain special instruction before use .
S45:In case of accident or if you feel unwell, seek medical advice immediately (show label where possible) .
S16:Keep away from sources of ignition-No smoking .
S7:Keep container tightly closed . | [RIDADR ]
UN 1593 6.1/PG 3 | [WGK Germany ]
1
| [RTECS ]
AM5250000
| [F ]
8 | [Autoignition Temperature]
570 °C | [TSCA ]
Yes | [PackingGroup ]
III | [HS Code ]
29143900 | [Safety Profile]
Poison by intraperitoneal and subcutaneous routesModerately toxic by ingestion. A skin and severe eye irritant. Mutation data reported. Narcotic in high concentration. A hypnotic. Flammable liquid. To fight fire, use foam, CO2, dry chemical. When heated to decomposition it emits acrid smoke and fumes. See also IGTONES | [Hazardous Substances Data]
98-86-2(Hazardous Substances Data) | [Toxicity]
LD50 orally in rats: 0.90 g/kg (Smyth, Carpenter) |
| Hazard Information | Back Directory | [General Description]
A colorless liquid with a sweet pungent taste and odor resembling the odor of oranges. Melting point 20.5°C (68.9°F); freezes under cool conditions. Slightly soluble in water and denser than water. Hence sinks in water. Vapor heavier than air. Flash point 180°F. A mild irritant to skin and eyes. Vapors can be narcotic in high concentrations. Used as a flavoring, solvent, and polymerization catalyst. | [Reactivity Profile]
ACETOPHENONE(98-86-2) reacts with many acids and bases liberating heat and flammable gases (e.g., H2). Reacts with many oxidizing agents. Reacts with reducing agents such as hydrides, alkali metals, and nitrides to produce flammable gas (H2) and heat. The amount of heat in these reactions may be sufficient to start a fire in the unreacted portion. Incompatible with isocyanates, aldehydes, cyanides, peroxides, and anhydrides. | [Air & Water Reactions]
Slightly soluble in water. | [Health Hazard]
No toxicity expected from inhalation or ingestion except slight narcotic effect. Liquid can cause eye and skin irritation on contact. | [Potential Exposure]
Acetophenone is used as a solvent and in perfume manufacture to impact a pleasant jasmine or orange-blossom odor. It is used as a catalyst in olefin polymerization and as a flavorant in tobacco. It is also used in the synthesis of pharmaceuticals | [First aid]
Remove any contact lenses at once, then flush eyes, wash contaminated areas of body with soap and water. If this chemical gets into the eyes, remove any contact lenses at once and irrigate immediately for at least 15 minutes, occasionally lifting upper and lower lids. If this chemical contacts the skin, remove contaminated clothing and wash immediately with soap and water. If this chemical has been inhaled, remove from exposure, begin rescue breathing (using universal precautions, including resuscitation mask) if breathing has stopped and CPR if heart action has stopped. Transfer promptly to a medical facility. When this chemical has been swallowed, get medical attention. Give large quantities of water and induce vomiting. Do not make an unconscious person vomit. | [Shipping]
UN1993 Flammable liquids, n.o.s., Hazard Class: 3; Labels: 3-Flammable liquid, Technical Name Required. | [Incompatibilities]
May form explosive mixture with air. See flash point, above. Reacts violently with strong oxidizers, many acids, bases, amines, amides, and inorganic hydroxides; alkali metals; hydrides, and nitrides. Reacts with reducing agents; alkali metals; hydrides, nitrides. Contact with all preceding materials release heat and flammable gases, including hydrogen; the heat may be sufficient enough to result in fire. Incompatible with aldehydes, aliphatic amines, alkanolamines, cyanides, isocyanates, organic acids, peroxides; perchloric acid. May attack plastics, and some rubbers and coatings | [Waste Disposal]
Consult with environmental regulatory agencies for guidance on acceptable disposal practices. Generators of waste containing this contaminant (≥100 kg/mo) must conform with EPA regulations governing storage, transportation, treatment, and waste disposal. Incineration, preferably with a flammable solvent | [Occurrence]
Reported found in cocoa, beef, raspberry, peas, and concord grape | [Definition]
ChEBI: A methyl ketone that is acetone in which one of the hydrogens of the methyl group has been replaced by a phenyl group. | [Preparation]
From benzene and acetylchloride in the presence of aluminum chloride or by catalytic oxidation of ethyl benzene; also
prepared by fractional distillation and crystallization from the essential oil of Stirlingia latifolia. | [Production Methods]
Most methyl phenyl ketone originates from the Hock process for the production of phenol from isopropylbenzene (→Phenol); it is isolated from the residue of this process. In addition, acetophenone can be obtained as a main product by selective decomposition of cumene hydroperoxide in the presence of copper catalysts at 100℃:
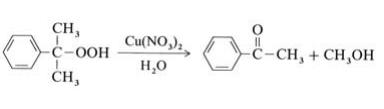
A second possibility is the oxidation of ethylbenzene with air or oxygen at 130℃ and 0.5 MPa. Catalysts used include cobalt salts or manganese salts of naphthenic or fatty acids. Conversion of ethylbenzene is limited to ca. 25 % to minimize the byproducts 1-phenylethanol and benzoic acid. A third method is the Friedel – Crafts acetylation of benzene with acetic anhydride, but this is not of industrial importance. | [Aroma threshold values]
Detection: 170 ppb; Recognition: 2.9 ppm | [Taste threshold values]
Taste characteristics at 10 ppm: sweet, nutty, benzaldehyde with musty, fruity notes | [Synthesis Reference(s)]
Canadian Journal of Chemistry, 56, p. 2269, 1978 DOI: 10.1139/v78-373
Chemical and Pharmaceutical Bulletin, 31, p. 4209, 1983 DOI: 10.1248/cpb.31.4209 | [Fire Hazard]
Combustible liquid; flash point (closed cup)
82°C (180°F); vapor pressure 1 torr at
37°C (98.6°F); vapor density 4.1 (air = 1);
autoignition temperature 570°C (1058°F);
fire-extinguishing agent: dry chemical, foam,
or CO2; water may cause frothing, but it can
be used to flush and dilute the spill. Its reaction
with strong oxidizers may be violent. | [Flammability and Explosibility]
Notclassified | [Carcinogenicity]
No carcinogenicity studies were
identified for acetophenone. The U.S. EPA has classified
acetophenone as a Category D, not classifiable as to human
carcinogenicity. | [Environmental Fate]
It is unclear what mechanism is responsible for the central
nervous system depression observed following high doses of
acetophenone. In vitro evaluations have demonstrated that
acetophenone suppresses voltage-gated ion channels in olfactory
receptor cells and retinal neurons; however, it is unclear if
this is related to any of the observed toxicity in animal studies. | [Metabolism]
At one time, acetophenone was used as a hypnotic. Its conversion to benzoic acid and methylphenylcarbinol in dogs and rabbits was observed by a number of early workers. Small amounts are also excreted as mandelic acid. In the rabbit about half the dose is excreted as methylphenylcarbinyl glucuronide and about 20 % as hippuric acid. It is probable that the ketone is first asymmetrically reduced to the carbinol, which is the precursor of benzoic and mandelic acids. | [Purification Methods]
Dry it by fractional distillation or by standing with anhydrous CaSO4 or CaCl2 for several days, followed by fractional distillation under reduced pressure (from P2O5, optional), and careful, slow and repeated partial crystallisations from the liquid at 0o excluding light and moisture. It can also be crystallised at low temperatures from isopentane. Distillation can be followed by purification using gas-liquid chromatography [Earls & Jones J Chem Soc, Faraday Trans 1 71 2186 1975.] [Beilstein 7 H 271, 7 IV 619.] § A commercial polystyrene supported version is available — scavenger resin (for diol substrates). | [Toxicity evaluation]
The production and use of acetophenone as a specialty solvent
and fragrance and flavor additive may result in environmental
releases to the air, water, and soil. Acetophenone is slightly
soluble in water and freely soluble in alcohol, chloroform, fatty
oils, and glycerol. The vapor pressure at 25 °C is 0.4 mmHg,
the Henry’s law constant is estimated to be 1.04×10�5
atmm3 mol�-1, and the octanol/water partition coefficient
(log Kow) is 1.58.
If released to the soil, acetophenone is expected to have high
mobility, with volatilization from moist soils. If released to the
water, volatilization is anticipated from the surface and acetophenone
is not anticipated to adsorb to sediment. The potential
for bioconcentration in aquatic organisms is predicted to
be low. If released to the atmosphere, the half-life is anticipated
to be 6 days, with degradation occurring by reaction with
photochemically produced hydroxyl radicals. |
| Questions And Answer | Back Directory | [General description]
Acetophenone is also known as acetyl benzene with the boiling point (℃) being 202.3, relative density (water = 1) being 1.03 (20 ℃) and the relative vapor density (air = 1) being 4.14. It is the simplest aromatic ketones with its aromatic core (benzene ring) being directly connected with a carbonyl group. It is presented in some kinds of essential oils of some plants in its free-state form. It is pure colorless crystals. Most commercially available product appears as pale yellow oily liquid with hawthorn like aroma. It is only slightly soluble in water and is easily soluble in many organic solvents and can be evaporated together with the steam. The molecular structure of acetophenone: methyl C atom is bonded through sp3 hybrid orbital while the benzene ring and the carbonyl C atom are bonded through sp2 hybrid orbital. Addition reaction and [alpha] active hydrogen reaction can occur for the carbonyl group of acetophenone. It can also have electrophilic substitution reaction in its benzene ring with the major product generated being in the meta-position. Acetophenone can be produced from the reaction between benzene with acetyl chloride, acetic anhydride or acetate under the catalysis of aluminum trichloride. Furthermore, when ethylbenzene is catalyzed and oxidized into styrene, it can also generate acetophenone as by-product. Acetophenone is mainly used as the raw materials for pharmaceuticals and other kind of organic synthesis. It can also used for the preparation of spices, soap and cigarettes as well as being used as the solvents of cellulose ether, cellulose ester and resin and plasticizer. It has hypnotic effect. Currently, acetophenone is mostly obtained via the by-product of reaction between phenol and acetone via cumene oxidation. It may be obtained through the acetylation on benzene through acetyl chloride.
| [Characterization of acetophenone production via acylation reaction of Friedel-Craffs]
1, the catalyst (aluminum trichloride) used in the acylation greatly exceeds the amount of the catalyst used in the alkylation reaction, the resulting acetophenone can form complex with an equivalent amount of aluminum chloride while by-product acetic acid formed in the reaction can also form salt with an equivalent of aluminum chloride. Therefore, during the acylation reaction, one molecule of acid anhydride can consume two or more molecules of aluminum chloride.
2, acetophenone/aluminum chloride complex formed during the reaction is stable in anhydrous medium, only when the reaction mixture undergoes hydrolysis, the complex is destroyed with precipitating acetophenone. After the aluminum chloride forms complex with acetophenone, it no longer participates in the reaction, therefore, the amount of aluminum chloride should meet the requirement that: after the formation of complex, there still remains certain part as the catalyst of the acylation reaction.
3, since the aluminum chloride can form a complex with substance containing a carboxyl group, the raw material, acetic anhydride can also form molecular complex with aluminum chloride and no longer participate in the reaction, meaning that acetic anhydride can’t been quantitatively converted to acetophenone; Furthermore, in the reaction, when the amount of aluminum chloride is in excess, it can act as the acylating agent for making the acetate into acetyl chloride to participate into the reaction.
4, in the reaction system, the amount of benzene is also in large excess because the benzene is not only the reactant but also the solvent in the reaction, therefore, only the yield of acetyl can be used as the reference reagent.
5, the characteristics of the acylation reaction: pure product, high yield (since acyl does not have isomerization, nor has multiple substitution)
| [Content Analysis]
Accurately weigh sample of about 1g, determine it according to the method 1 in the "determination of aldehydes and ketones" (OT-7), wherein the heating time was 1h and the equivalency factors (e) in the calculation was 60.08.
| [Chemical Properties]
The pure product is white plate-like crystal. The general merchandises always contain impurities. At temperature above 20 °C, it is colorless or slightly yellow transparent liquid above with strong acacia-like sweet aroma. It has the melting point of 19.7 ℃, boiling point of 202 ℃ and the flash point of 76 ℃. It can be subject to spontaneous burning. It is extremely easily to be dissolved in propylene glycol and non-volatile oil, soluble in chloroform, ether and ethanol (1ml dissolved in 5mL 50% ethanol), slightly soluble in water and propylene glycol and insoluble in glycerol.
The natural product is presented in labdanum oil and orris oil.
| [Uses]
Methyl phenyl ketone is mainly converted to resins (synthetic resins) by reaction with formaldehyde. It is also used as a photoinitiator for special printing plates as well as for organic syntheses, particularly for pharmaceuticals. Certain derivatives, such as 1-phenylethanol (obtained by hydrogenation) and its acetate, are used as fragrances. Furthermore, methyl phenyl ketone is used for the synthesis of optical brighteners. |
|
|