Stickstoff Chemische Eigenschaften,Einsatz,Produktion Methoden
ERSCHEINUNGSBILD
GERUCHLOSE FARBLOSE TIEFKALTE FLüSSIGKEIT.
ERSCHEINUNGSBILD
GERUCHLOSES FARBLOSES KOMPRIMIERTES GAS.
PHYSIKALISCHE GEFAHREN
Das Gas vermischt sich leicht mit Luft.
PHYSIKALISCHE GEFAHREN
Das Gas ist im kalten Zustand schwerer als Luft. Es kann sich in tiefer gelegenen Bereichen sammeln und den Luftsauerstoff verdrängen.
ARBEITSPLATZGRENZWERTE
TLV: Erstickungsgefahr durch Sauerstoffverdrängung. (ACGIH 2005).
MAK nicht festgelegt (DFG 2005).
ARBEITSPLATZGRENZWERTE
TLV: Erstickungsgefahr durch Sauerstoffverdrängung. (ACGIH 2005).
MAK nicht festgelegt (DFG 2005).
AUFNAHMEWEGE
Aufnahme in den Körper durch Inhalation.
AUFNAHMEWEGE
Aufnahme in den Körper durch Inhalation.
INHALATIONSGEFAHREN
Beim Entweichen aus dem Behälter verdampft die Flüssigkeit sehr schnell, wobei die Luft verdrängt wird. Ernste Erstickungsgefahr in geschlossenen Räumen. (S. Anm.)
INHALATIONSGEFAHREN
Beim Entweichen aus dem Behälter kann das Gas die Luft verdrängen. Erstickungsgefahr in geschlossenen Räumen. (S. Anm.)
WIRKUNGEN BEI KURZZEITEXPOSITION
WIRKUNGEN BEI KURZZEITEXPOSITION: Die Flüssigkeit kann Erfrierungen hervorrufen.
LECKAGE
Belüftung. Wasserstrahl NIEMALS auf die Flüssigkeit richten. Chemikalienschutzanzug mit umgebungsluftunabhängigem Atemschutzgerät.
LECKAGE
Belüftung. Persönliche Schutzausrüstung: Umgebungsluftunabhängiges Atemschutzgerät.
S-Sätze Betriebsanweisung:
S38:Bei unzureichender Belüftung Atemschutzgerät anlegen.
Beschreibung
Nitrogen makes up the major portion of the atmosphere
(78.08 percent by volume, 75.5 percent
by weight). It is a colorless, odorless,
tasteless, nontoxic, almost totally inert gas, and
is colorless as a liquid. Nitrogen is nonflammable,
will not support combustion, and is not life
supporting. It combines with some of the more
active metals such as lithium and magnesium to
form nitrides, and at high temperatures it will
also combine with hydrogen, oxygen, and other
elements. It is used as an inert protection against
atmospheric contamination in many nonwelding
applications. Nitrogen is only slightly soluble in
water and most other liquids, and is a poor conductor
of heat and electricity. As a liquid at
cryogenic temperatures it is nonmagnetic. It is
shipped as a nonliquefied gas at pressures of
2000 psig (13 790 kPa) or above, and also as a
cryogenic fluid at pressures and temperatures
below 200 psig (1380 kPa) and -261°F
(-163°C).
Chemische Eigenschaften
Nitrogen occurs naturally as approximately 78% v/v of the atmosphere. It is a nonreactive, noncombustible, colorless, tasteless, and odorless gas. It is often used under refrigeration as a cryogenic liquid. The boiling point is -195.8 °C and -320 °F. Nitrogen is not combustible. Nitrogen can combine with oxygen at high temperatures to form oxides and may form ammonia in contact with hydrogen at elevated temperatures. Cyanides can form if nitrogen is heated with carbon in presence of alkalies or barium oxide. If nitrogen comes in contact with ozone, nitrogen can oxidize explosively.It is usually handled as a compressed gas, stored in metal cylinders.
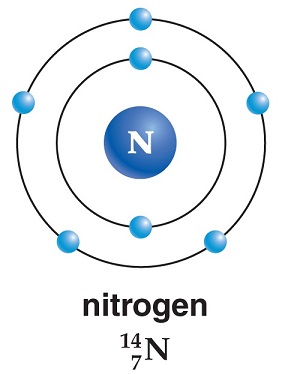
Physikalische Eigenschaften
In its natural gaseous state, nitrogen is a relatively inert diatomic molecule (N2) that iscolorless, odorless, and tasteless, yet it is responsible for hundreds of active compounds. Itmakes up about 78% of the air we breathe. We are constantly taking it into our lungs withno stimulation or sensation; therefore, we really do not detect its presence. When liquefied, itis still colorless and odorless and resembles water in density. The melting point of nitrogen is–209.86°C, its boiling point is –195.8°C, and its density as a gas is 0.0012506 g/cm
3.
Isotopes
There are 19 isotopes of nitrogen, two of which are stable. The stable ones andtheir proportion to the natural abundance of nitrogen on Earth follow: N-14 = 99.634%and N-15 = 0.366%. The other 17 isotopes are radioactive and man-made in nuclearreactors and have half-lives ranging from a few nanoseconds to 9.965 minutes.
Origin of Name
From the two Greek words nitron and genes, which together stand for
“soda or saltpeter forming.”
Occurrence
Nitrogen is the 30th most abundant element on Earth. There is an almost unlimited sourceof nitrogen available to us considering that our atmosphere constitutes 4/5, or over 78%, ofthe nitrogen by volume. Over 33 million tons of nitrogen is produced each year by liquefyingair and then using fractional distillation to produce nitrogen as well as other gases in the atmosphere. During this process the air is cooled and then slowly warmed to fractionaltemperature points at which each specific gas in the air will “boil” off. (Note: Oxygen, argon,carbon dioxide, and nitrogen all have specific boiling points and these gases can be used tocollect the specific gas during the fractionation process.) When the temperature –reaches–195.8°C, the nitrogen is boiled off and collected.
There is a balance of nitrogen with other gases in the atmosphere that is maintained bywhat is called the nitrogen cycle. This cycle includes several processes, including nitrogen fixationof bacteria in the soil by legumes (bean and pea plants). Lightning produces nitrogen, asdo industrial waste gases and the decomposition products of organic material (i.e., organicproteins and amino acids in plants and animals contain nitrogen). In time, these sourcesreplace the nitrogen in the atmosphere to complete the cycle.
Ammonia (NH
3) is the first binary molecule discovered in outer space of our galaxy, theMilky Way. It may also be the main compound that forms the rings of the planet Saturn.
Charakteristisch
There are approximately 4,000 trillion tons of gas in the atmosphere, and nitrogen makesup about 78% of these gases. It is slightly soluble in water and alcohol. It is noncombustibleand is considered an asphyxiant gas (i.e., breathing pure nitrogen will deprive the body ofoxygen).
Although nitrogen is considered an inert element, it forms some compounds that are veryactive. Of the diatomic molecules, such as CO
2 , it is difficult to separate the two atoms innitrogen’s molecules because of their strong binding energy. This is the reason that, along withcarbon dioxide, nitrogen gas is stable. However, once separated, the individual atoms of nitrogen(N) become very reactive and do combine with hundreds of other elements.
Nitrogen can be liquefied easily, making it useful in many applications wherein sustainedcooling is needed. At high temperatures, nitrogen reacts with many metals to form nitrides.
Verwenden
In manufacture of ammonia, nitric acid, nitrates, cyanides, etc.; in manufacture of explosives; in filling high-temp thermometers, incandescent bulbs; to form an inert atm for preservation of materials, for use in dry boxes or glove bags. Liquid nitrogen in food-freezing processes; in the laboratory as a coolant. Pharmaceutic aid (air displacement).
Vorbereitung Methode
Nitrogen is obtained commercially, in large quantities, by the
fractional distillation of liquefied air.
Definition
Nitrogen, N2, is a colorless,odorless, inert gas that comprises 80%of the earth's atmosphere. It serves as a diluent and controls natural burning and respiration rates, which would be much faster in higher concentrations of oxygen. Nitrogen is soluble in water and alcohol, but is essentially insoluble in most other liquids. It is essential to practically all forms of life and its compounds serve as foods or fertilizers. Nitrogen is used in the manufacture of ammonia and nitric acid. Nitrogen is essentially an inert gas at ambient and moderate temperatures. Therefore, it is easily handled by most metals.At elevated temperatures, nitrogen can be aggressive to metals and alloys.
Allgemeine Beschreibung
A colorless odorless gas. Noncombustible and nontoxic. Makes up the major portion of the atmosphere, but will not support life by itself. Used in food processing, in purging air conditioning and refrigeration systems, and in pressurizing aircraft tires. May cause asphyxiation by displacement of air. Under prolonged exposure to fire or heat containers may rupture violently and rocket.
Air & Water Reaktionen
Slightly soluble in water.
Reaktivität anzeigen
These substances undergo no chemical reactions under any known circumstances except those under extreme conditions (liquid Nitrogen reacts violently in mixture with magnesium powder when a fuse is lit. Due to formation of magnesium nitride). Otherwise, they are nonflammable, noncombustible and nontoxic. They can asphyxiate.
Hazard
Nitrogen is nontoxic, but it is an asphyxiate gas that cannot, by itself, support oxidation(combustion) or support life. If you breathe pure nitrogen for any period of time, you will die—not because the nitrogen gas is a poison, but because your body will be deprived of oxygen.
Nitrogen oxides are formed under certain conditions when nitrogen combines with oxygen,thus contributing to pollution. One source is from the internal combustion engine thatproduces NO similar to lightning. Once released, it combines with more oxygen to form ,which is a very reactive polluting gas. Nitrogen dioxide NO
2 is the main cause of “brown”smog over some cities and is harmful to plants, animals, and humans. To make matter worse,if there is adequate sunlight at the time of the smog, the ultraviolet light of the sun will breakdown the N and O of the NO
2 to form free radicals of oxygen that are reactive, forming ozone(O
3), which is itself a strong oxidizing agent that adds to pollution.
Several of the oxygen, hydrogen, and halogen compounds of nitrogen are toxic wheninhaled. A common error made in using household cleaners is to mix or use together ammoniacleaning fluids (containing nitrogen) and Clorox-type cleaning fluids (containing chlorine).The combined fumes can be deadly in any confined area. NEVER mix Clorox with ammoniatypecleaning fluids.
Health Hazard
Vapors may cause dizziness or asphyxiation without warning. Vapors from liquefied gas are initially heavier than air and spread along ground.
Brandgefahr
Non-flammable gases. Containers may explode when heated. Ruptured cylinders may rocket.
Industrielle Verwendung
Nitrogen is often called an inert gas, and is used for some inert atmospheres for metal treating and in lightbulbs to prevent arcing, but it is not chemically inert. It is a necessary element in animal and plant life, and is a constituent of many useful compounds. Nitrogen combines with many metals to form hard nitrides useful as wear-resistant metals. Small amounts of nitrogen in steels inhibit grain growth at high temperatures, and also increase the strength of some steels. It is also used to produce a hard surface on steels.
Because of the importance of nitrogen compounds in agriculture and chemical industry, much of the industrial interest in elementary nitrogen has been in processes for converting elemental nitrogen into nitrogen compounds. The principal methods for doing this are the direct synthesis of ammonia from nitrogen and hydrogen, the electric arc process, which involves the direct combination ofN
2 and O
2 to nitric oxide, and the cyanamide process.
Materials Uses
Gaseous nitrogen is noncorrosive and inert, and
may consequently be contained in systems constructed
of any common metals and designed to
safely withstand the pressures involved. At the
temperature of liquid nitrogen, ordinary carbon
steels and most alloy steels lose their ductility
and are considered unsafe for liquid nitrogen
service. Satisfactory materials for use with liquid
nitrogen include austenitic stainless steel
(for example, types 304 and 316) and other
nickel-chromium alloys, copper, Monel, brass,
and aluminum.
Pharmakologie
Atropine
does not reactivate the phosphorylated AChE but competes
with acetylcholine for binding with the muscarinic
acetylcholine receptor acting as an antagonist.
Sicherheitsprofil
Low toxicity. In high
concentrations it is a simple as-p~h yxiant.
The release of nitrogen from solution in the
blood, with formation of small bubbles, is
the cause of most of the symptoms and
changes found in compressed air illness
(caisson disease). It is a narcotic at hgh
concentration and hgh pressure. Both the
narcotic effects and the bends are hazards of
compressed air atmospheres such as found
in underwater dving. Nonflammable gas.
Can react violently with lithium,
neodymium, titanium under the proper
condtions. See also ARGON.
Sicherheit(Safety)
Nitrogen is generally regarded as a nontoxic and nonirritant
material. However, it is an asphyxiant and inhalation of large
quantities is therefore hazardous.
mögliche Exposition
Nitrogen is present in the air we
breathe. Health effects may occur at concentrations above 80%. It has many medical and industrial uses including the
quick freezing of food. The gas is used for purging, heat
treating; food freezing; annealing, cooling, oil recovery; in
the inert blanketing of sensitive materials and as a reactant
in chemical synthesis of ammonia.
Lager
Nitrogen is stable and chemically unreactive. It should be stored in
tightly sealed metal cylinders in a cool, dry place.
Versand/Shipping
UN1066 Nitrogen, compressed, Hazard Class:,
Hazard Class: 2.2; Labels: 2.2-Nonflammable compressed
gas; UN1977 Nitrogen, refrigerated liquid cryogenic liquid,
Hazard Class:, Hazard Class: 2.2; Labels: 2.2-
Nonflammable compressed gas. Cylinders must be transported
in a secure upright position, in a well-ventilated
truck. Protect cylinder and labels from physical damage.
The owner of the compressed gas cylinder is the only entity
allowed by federal law (49CFR) to transport and refill
them. It is a violation of transportation regulations to refill
compressed gas cylinders without the express written permission
of the owner.
Inkompatibilitäten
Containers may explode when heated.
Liquid nitrogen is very unreactive, nonflammable, noncombustible
and nontoxic. Contact with water may result in
vigorous or violent boiling and extremely rapid vaporization.
If the water is hot, there is the possibility that a liquid
“superheat” explosion may occur. Pressures may build to
dangerous levels if the liquid contacts water in a closed
container.
Waste disposal
Return refillable compressed
gas cylinders to supplier. Vent to atmosphere.
Regulatory Status
GRAS listed. Included in the FDA Inactive Ingredients Database
(injections; dental preparations; nasal sprays; oral solutions; rectal
gels). Accepted for use as a food additive in Europe. Included in
parenteral and nonparenteral medicines licensed in the UK and
USA. Included in the Canadian List of Acceptable Non-medicinal
Ingredients.
Stickstoff Upstream-Materialien And Downstream Produkte
Upstream-Materialien
Downstream Produkte

