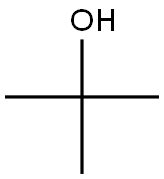
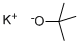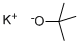Potassium tert-butoxide
- CAS No.
- 865-47-4
- Chemical Name:
- Potassium tert-butoxide
- Synonyms
- t-BuOK;POTASSIUM TERT-BUTYLATE;POTASSIUM T-BUTOXIDE;KTB;Potassium Tertiary Butoxide;tert-BuOK;Potassium tert-butanolate;tert-Butoxypotassium;tert-butoxide, potassium;PotassiuM tert-butyl alcohol
- CBNumber:
- CB1335932
- Molecular Formula:
- C4H9KO
- Molecular Weight:
- 112.21
- MDL Number:
- MFCD00012162
- MOL File:
- 865-47-4.mol
- MSDS File:
- SDS
| Melting point | 256-258 °C (dec.) (lit.) |
|---|---|
| Boiling point | 275°C |
| Density | 0.910 g/mL at 20 °C |
| vapor pressure | 1 mm Hg ( 220 °C) |
| Flash point | 54 °F |
| storage temp. | Store below +30°C. |
| solubility | Soluble in hexane, toluene, diethyl ether and terahydrofuran. |
| pka | pK1:18(25°C) |
| form | Solution |
| color | White to off-white |
| Specific Gravity | 0.902 |
| PH | 13 (5g/l, H2O, 20℃)Hydrolysis |
| Water Solubility | REACTS |
| Sensitive | Moisture Sensitive |
| Hydrolytic Sensitivity | 7: reacts slowly with moisture/water |
| BRN | 3556712 |
| Stability | Stable, but reacts violently with water and acids, possibly leading to fire. Incompatible with water, acids, halogenated hydrocarbons, alcohols, strong oxidizing agents, ketones, carbon dioxide. |
| InChIKey | LPNYRYFBWFDTMA-UHFFFAOYSA-N |
| CAS DataBase Reference | 865-47-4(CAS DataBase Reference) |
| FDA UNII | VR838VHE0V |
| EPA Substance Registry System | 2-Methyl-2-propanol, potassium salt (865-47-4) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS02,GHS05 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H228-H260-H314 | |||||||||
| Precautionary statements | P210-P231+P232-P260-P280-P303+P361+P353-P305+P351+P338 | |||||||||
| Hazard Codes | F,C,Xi | |||||||||
| Risk Statements | 11-19-22-34-35-14-20-36/37-37 | |||||||||
| Safety Statements | 16-26-36/37/39-43-45-7/9-8-43A-33-27-29 | |||||||||
| RIDADR | UN 3274 3/PG 2 | |||||||||
| WGK Germany | 1 | |||||||||
| F | 10-21 | |||||||||
| Autoignition Temperature | 483 °C | |||||||||
| Hazard Note | Flammable solid/Corrosive | |||||||||
| TSCA | Yes | |||||||||
| HazardClass | 8 | |||||||||
| PackingGroup | I | |||||||||
| HS Code | 29051900 | |||||||||
| NFPA 704 |
|
Potassium tert-butoxide price More Price(71)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | 8.43849 | Potassium tert-butylate (~ 20% solution in tetrahydrofuran) for synthesis | 865-47-4 | 50mL | $86.1 | 2024-03-01 | Buy |
| Sigma-Aldrich | 8.04918 | Potassium tert-butylate for synthesis | 865-47-4 | 100g | $67.1 | 2024-03-01 | Buy |
| Sigma-Aldrich | 8.43849 | Potassium tert-butylate (~ 20% solution in tetrahydrofuran) for synthesis | 865-47-4 | 250ML | $244 | 2024-03-01 | Buy |
| Sigma-Aldrich | 8.04918 | Potassium tert-butylate for synthesis | 865-47-4 | 250g | $134 | 2024-03-01 | Buy |
| Sigma-Aldrich | 8.04918 | Potassium tert-butylate for synthesis | 865-47-4 | 2.5kg | $324 | 2024-03-01 | Buy |
Potassium tert-butoxide Chemical Properties,Uses,Production
Strong organic alkaline
Potassium tert-butoxide is an important kind of organic base with its alkalinity being stronger than potassium hydroxide. Because of the induction effect of three methyl groups in the (CH3) 3CO-, it has stronger alkalinity and activity than other potassium alkoxides, thus being a very excellent catalyst. In addition, as a kind of strong alkaline, potassium tert-butoxide is widely used in the organic synthesis of chemical, pharmaceutical, and pesticide such as trans-esterification, condensation, rearrangement, polymerization, loop opening and the production of heavy metal orthoester. It can be used to catalyze the Michael addition reaction, Pinacol rearrangement reaction and Ramberg-Backlund rearrangement reaction. Potassium tert-butoxide is used as a condensing agent, which can be used to catalyze Darzens condensation reaction and Stobbe condensation reaction. It is also the most effective alkaline for traditional akloxide-haloform reaction generating dihalogenated carbene. Therefore, potassium tert-butoxide has attracted more and more attention from chemical, pharmaceutical, pesticide and other industries. As potassium tert-butoxide has such a wide application, the domestic and international demand for high purity potassium tert-butoxide is very large. However, due to its production cost is higher than other alkali metal alkoxides, the production technology needs to be improved, so in-depth study of potassium tert-butoxide is particularly important.
chemical properties
Potassium tert-butoxide is a strong alkaline condensing agent with its alkalinity is stronger than sodium methoxide and sodium ethoxide. At room temperature, this product is white or white-like solid powder with its chemical formula being (CH3) 3C-O-K, molecular weight being 112.21, density being 0.929, the melting point of 256-258°C and the boiling point being 275°C. It is easy to absorb moisture. It is soluble in tert-butanol with the solution being relative stable, and commonly used in the dehydrohalogenation reaction of halogenated hydrocarbon. Being exposure to air decomposition, it can be decomposed into potassium oxide and tert-butyl alcohol when coming across water. It can be manufactured through the reaction between butanol and potassium, followed by the vacuum distillation of tert-butanol in the system.
Potassium tert-butoxide is used as a strong non-nucleophilic base in organic chemistry. It is widely used the condensation reaction, rearrangement and ring-opening reaction in organic synthesis. People generally apply tert-butanol solution. Potassium tert-butoxide is a kind of organic alkaline corrosion products with strong moisture absorption property and should be sealed for storage. We need to pay attention to prevention of fire and sunshine during the storage and transportation. It should be stored in a cool, ventilated and dry place.
reaction
Potassium tert-butoxide may be used as a base in the intramolecular cyclization of iodo arene to afford benzopyran via microwave method of synthesis.
Potassium tert-butoxide has been used as a strong base in the enantioselective synthesis of amines by transfer hydrogenation of N-(tertbutylsulfinyl)imines.
Take 2, 6, 6-trimethylcyclohexane-2-alkenyl formaldehyde to have reaction with allyl Grignard agent to generate cyclohexenyl butenol, followed by the oxidation under copper-zinc catalyst to generate cyclohexane alkenyl ketene, followed by isomerization in the alkali solution of potassium tert-butoxide, being able to generate Damascenone.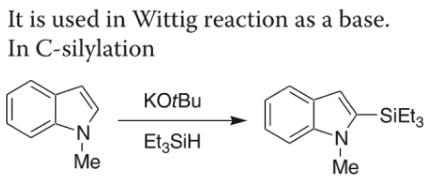
Darzen reaction: aldehydes and ketones, in the presence of alkaline reagents, can have ester-aldol condensation reaction with α-halo acid ester, while having the hydrogen halogen lost, generating α, β-epoxy ester. Common alkaline reagents include sodium ethoxide, sodium amide, sodium metal, potassium tert-butoxide and so on.
Stobbe reaction: aldehydes, ketones and succinate ester, in the presence of alkaline catalysts (such as sodium ethoxide, potassium tert-butoxide, sodium chloride, etc.) have reaction similar to the aldol condensation reaction, generating succinate or its monoester with the methylene replaced by hydrocarbon.
KOtBu: A Privileged Reagent for Electron Transfer Reactions
Potassium tert-butoxide mediated C–C, C–N, C–O and C–S bond forming reactions
Uses
Potassium tert-butoxide, as a strong alkaline, is widely used in the condensation, rearrangement and ring opening reaction in organic production in the fields of chemical, pharmaceutical and pesticide.
It can also be used:
To synthesize aliphatic and aromatic amides from corresponding esters and amines.
As a base in the intramolecular cyclization of aryl ethers, amines, and amides.
As a catalyst to prepare styrene derivatives from aryl halides and alkenes by Mizoroki-Heck reaction.
preparation
The potassium metal was added to the freshly distilled tert-butanol under nitrogen atmosphere, refluxed to until the potassium was completely melted. Further incubate for 1 hour. The excess amount of t-butyl alcohol was distilled off with the remaining white solid subjecting to vacuum drying under reduced pressure at 180-190 °C for 10 h. Then we can obtain the crystal powder of potassium tert-butanol. It needs to saved be under the nitrogen atmosphere for using. It should be kept away from air & water, or will become pink. The yield, calculated according to potassium, is more than 99%. The chemical reaction equation for the reaction between tert-butyl alcohol and metal potassium for preparation of potassium tert-butoxide is as follows: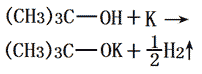
Precautions
Potassium tert-butoxide has two types, liquid and solid. Usually the liquid industrial product is the tert-butanol solution of tert-butanol potassium with the color of the product being light yellow or milky white, being slightly turbid, in which the content of potassium tert-butoxide being 10% to 12%; the solid product is generally white or white powder in which the potassium tert-butoxide content is 95% to 97%.
Potassium tert-butoxide is organic alkaline corrosion product with strong moisture absorption property. It should be generally sealed for storage, generally stored in a cool, dry, ventilated warehouse. It should be kept away from heat and isolated from fire isolation as well as being protected from sun exposure. Potassium tert-butoxide has a strong corrosive effect on the skin. During the handling and loading process, the operator should wear a protective mask to prevent corrosion and burning by potassium tert-butoxide.
The above information is edited by Tongtong from Chemicalbook.
Chemical Properties
white crystalline powder
Uses
Usually used for greener amidation of esters.
Uses
Potassium tert-Butoxide is used in the synthesis of many organic compounds primarily as a strong base. In particular, it is used as a reagent in the base- catalyzed carbonylation of amines for the synthesis of N-Formamides.
General Description
This product has been enhanced for catalytic efficiency.
Fire Hazard
Potassium tert-butoxide is a flammable solid.
It ignites on heating. Being very strongly
basic, its reactions with acids are highly
exothermic. Contact of solid powder with
drops of sulfuric acid and vapors of acetic
acid caused ignition after an induction period
of 0.5 and 3 minutes, respectively (Manwaring
1973). Ignition occurs upon reactions
with many common solvents of the
type ketone, lower alcohols, esters, and halogenated
hydrocarbons. Such solvents include
acetone, methyl ethyl ketone, methyl isobutyl
ketone, methanol, ethanol, n-propanol, isopropanol,
ethyl acetate, n-propyl formate,
n-butyl acetate, chloroform, methylene chloride,
carbon tetrachloride, epichlorohydrin,
dimethyl carbonate, and diethyl sulfate
(NFPA 1997). Such ignition may arise from
accidental contact of the alkoxide with these
solvents and may be attributed to sudden
release of energy from exothermic reactions.
However, slow mixing of the powder
with excess solvent will dissipate the
heat. It reacts violently with water, producing
tert-butanol and potassium hydroxide, as
follows:
K―OC―(C4H9)3+H2O →
tert-C4H9OH +KOH
The addition of potassium tert-butoxide
to the solvent dimethyl sulfoxide can cause
ignition of the latter (Bretherick 1995).
Flammability and Explosibility
Flammable
Purification Methods
It sublimes at 220o/1mm. The last traces of tert-BuOH are removed by heating at 150-160o/2mm for 1hour. It is best prepared afresh as likely impurities are tert-BuOH, KOH and K2CO3 depending on its exposure to air. Its solubility at 25-26o in hexane, toluene, Et2O, and THF is 0.27%, 2.27%, 4.34% and 25.0%, respectively. [Feuer et.al. J Am Chem Soc 78 4364, Doering & Urban J Am Chem Soc 78 5938 1956, Beilstein 1 IV 1612.]
Potassium tert-butoxide Preparation Products And Raw materials
Raw materials
Preparation Products
1of8
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| career henan chemical co | +86-0371-86658258 | sales@coreychem.com | China | 29914 | 58 |
| Shandong Believe Chemical PTE.LTD | +86-18553607090 +8618553607090 | 1093908296@qq.com | China | 1684 | 58 |
| Hebei Mojin Biotechnology Co., Ltd | +8613288715578 | sales@hbmojin.com | China | 12453 | 58 |
| Hebei Yanxi Chemical Co., Ltd. | +8617531190177 | peter@yan-xi.com | China | 5993 | 58 |
| Hebei Yibangte Import and Export Co. , Ltd. | +86-0311-85030958 +8615532196582 | lisa@yibangte.com | China | 2988 | 58 |
| Hunan aslsen technology co.,ltd | +8615308460054 | aslsc@aslsen.com | China | 124 | 58 |
| Hebei Saisier Technology Co., LTD | +86-18400010335 +86-13102810335 | admin@hbsaisier.cn | China | 424 | 58 |
| Springchem New Material Technology Co.,Limited | +86-021-62885108 +8613917661608 | info@spring-chem.com | China | 2068 | 57 |
| Capot Chemical Co.,Ltd. | 571-85586718 +8613336195806 | sales@capotchem.com | China | 29797 | 60 |
| Hangzhou Bayee Chemical Co., Ltd. | 0086-571-86990109 | rachelhoo@bayeechem.com | China | 104 | 55 |
Related articles
- Potassium tert-butoxide: physicochemical property and Application
- Potassium tert-butoxide is also known as Potassium 2-methylpropan-2-olate or t-BuOK.
- Apr 17,2024
- The Uses of Potassium Tert-butoxide
- The passage introduces the uses of Potassium tert-butoxide.
- Nov 21,2022
View Lastest Price from Potassium tert-butoxide manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2024-04-19 | Potassium tert-butoxide
865-47-4
|
US $6.00 / KG | 1KG | More than 99% | 2000KG/MONTH | Hebei Saisier Technology Co., LTD | |
 |
2024-04-08 | Potassium Tert-Butoxide
865-47-4
|
US $0.00-0.00 / kg | 1kg | 99% | 20tons | Hunan aslsen technology co.,ltd | |
 |
2023-12-25 | Potassium tert-butoxide
865-47-4
|
US $0.00 / kg | 1kg | 99% | 1000000 | Hebei Jingbo New Material Technology Co., Ltd |
-

- Potassium tert-butoxide
865-47-4
- US $6.00 / KG
- More than 99%
- Hebei Saisier Technology Co., LTD
-

- Potassium Tert-Butoxide
865-47-4
- US $0.00-0.00 / kg
- 99%
- Hunan aslsen technology co.,ltd
-

- Potassium tert-butoxide
865-47-4
- US $0.00 / kg
- 99%
- Hebei Jingbo New Material Technology Co., Ltd