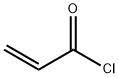
Acryloyl chloride
- CAS No.
- 814-68-6
- Chemical Name:
- Acryloyl chloride
- Synonyms
- prop-2-enoyl chloride;oyL;2-PROPENOYL CHLORIDE;acrylyl chloride;ACRYLIC ACID CHLORIDE;Propenoyl chloride;chloridkyselinyakrylove;Acryloyl chloride, 97%, for synthesis, stabilized with p-Hydroxyanisole;ACRYLOYL CHLORIDE;propenoylchloride
- CBNumber:
- CB4854427
- Molecular Formula:
- C3H3ClO
- Molecular Weight:
- 90.51
- MDL Number:
- MFCD00000731
- MOL File:
- 814-68-6.mol
| Melting point | 76 °C(Solv: isopropyl ether (108-20-3)) |
|---|---|
| Boiling point | 72-76 °C (lit.) |
| Density | 1.114 g/mL at 25 °C (lit.) |
| vapor density | >1 (vs air) |
| vapor pressure | 1.93 psi ( 20 °C) |
| refractive index |
n |
| Flash point | 61 °F |
| storage temp. | 2-8°C |
| solubility | Chloroform (Soluble), Methanol (Slightly) |
| form | Oil |
| color | Colourless to Pale Yellow |
| Water Solubility | Miscible with water. |
| Sensitive | Moisture & Light Sensitive |
| BRN | 635744 |
| Exposure limits |
ACGIH: TWA 5 mg/m3 (Skin) NIOSH: TWA 5 mg/m3 |
| Stability | Stable, but reacts violently with water. Incompatible with alcohols, oxidizing agents, strong bases. Light-sensitive. Highly flammable. |
| InChIKey | HFBMWMNUJJDEQZ-UHFFFAOYSA-N |
| CAS DataBase Reference | 814-68-6(CAS DataBase Reference) |
| EWG's Food Scores | 1 |
| FDA UNII | 8K23O56TG5 |
| NIST Chemistry Reference | 2-Propenoyl chloride(814-68-6) |
| EPA Substance Registry System | Acrylyl chloride (814-68-6) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |    GHS02,GHS05,GHS06 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H225-H290-H302-H314-H330 | |||||||||
| Precautionary statements | P210-P280-P301+P312-P303+P361+P353-P304+P340+P310-P305+P351+P338 | |||||||||
| Hazard Codes | F,T+ | |||||||||
| Risk Statements | 11-14-26-34 | |||||||||
| Safety Statements | 16-26-28-36/37/39-45-7/9 | |||||||||
| RIDADR | UN 3383 6.1/PG 1 | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | AT7350000 | |||||||||
| F | 8-10 | |||||||||
| TSCA | Yes | |||||||||
| HazardClass | 3 | |||||||||
| PackingGroup | II | |||||||||
| HS Code | 29161980 | |||||||||
| NFPA 704 |
|
Acryloyl chloride price More Price(14)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | 549797 | Acryloyl chloride 97.0%, contains <210ppm MEHQ as stabilizer | 814-68-6 | 5g | $76.2 | 2024-03-01 | Buy |
| Sigma-Aldrich | 549797 | Acryloyl chloride 97.0%, contains <210ppm MEHQ as stabilizer | 814-68-6 | 500g | $646 | 2024-03-01 | Buy |
| Alfa Aesar | L10363 | Acryloyl chloride, 96%, stab. with 400ppm phenothiazine | 814-68-6 | 10g | $40.5 | 2021-12-16 | Buy |
| Alfa Aesar | L10363 | Acryloyl chloride, 96%, stab. with 400ppm phenothiazine | 814-68-6 | 50g | $60.6 | 2021-12-16 | Buy |
| Sigma-Aldrich | A24109 | Acryloyl chloride ≥97%, contains ~400ppm phenothiazine as stabilizer | 814-68-6 | 5g | $66.7 | 2024-03-01 | Buy |
Acryloyl chloride Chemical Properties,Uses,Production
Chemical Properties
Acryloyl chloride is a highly flammable, polymerizable, and toxic (inhalation), light yellow liquid. A lacrimator.
Uses
Acryloyl chloride is used in the production of plastics. It plays an important role in the preparation of acrylate monomers and polymers. It also acts as a substrate for cross-metathesis. Further, it is utilized in organic synthesis for the introduction of acrylic groups into other compounds.
Synthesis Reference(s)
Journal of the American Chemical Society, 72, p. 72, 1950 DOI: 10.1021/ja01157a020
General Description
A liquid. Boiling point 75°C. Used to make plastics. Polymerization in a closed container can cause pressurization and explosive rupture.
Air & Water Reactions
Polymerizes readily upon exposure to oxygen in the air. Reacts exothermically with water to give hydrochloric acid and acrylic acid.
Reactivity Profile
Acrylyl chloride is incompatible with strong oxidizing agents, alcohols, amines, alkali. Polymerizes readily upon exposure to oxygen. May react vigorously or explosively if mixed with diisopropyl ether or other ethers in the presence of trace amounts of metal salts [J. Haz. Mat., 1981, 4, 291].
Fire Hazard
When heated to decomposition, Acrylyl chloride emits toxic fumes of chlorides. Decomposes in water.
Potential Exposure
May be used as a monomer in preparation of specialty polymers or as a chemical intermediate
Shipping
UN3383 Poisonous Toxic by inhalation liquid, flammable, n.o.s. with an LC50 # 200 mL/m3 and saturated vapor concentration ≥500 LC50, Hazard class: 6.1; Labels: 6.1-Poisonous materials, 3-Flammable liquid, Technical Name Required, Inhalation Hazard Zone A. UN2924 Flammable liquids, corrosive, n.o.s., Hazard Class: 3; Labels: 3-Flammable liquid, 8-Corrosive material, Technical Name Required
Incompatibilities
Use MEHQ (monomethyl ether of hydroquinone) as an inhibitor. Incompatible with oxidizers, polymerizes on contact with oxygen; alcohols, amines, alkalis. Reacts violently with water, releasing hydrochloric acid and acrylic acid. Attacks some metals.
Acryloyl chloride Preparation Products And Raw materials
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Henan Fengda Chemical Co., Ltd | +86-371-86557731 +86-13613820652 | info@fdachem.com | China | 7378 | 58 |
| Capot Chemical Co.,Ltd. | 571-85586718 +8613336195806 | sales@capotchem.com | China | 29797 | 60 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21695 | 55 |
| Shanghai Time Chemicals CO., Ltd. | +86-021-57951555 +8617317452075 | jack.li@time-chemicals.com | China | 1807 | 55 |
| Zjartschem | +86-571-8723 8903 jocelynpan@zjarts.com | jocelynpan@zjarts.com | CHINA | 993 | 58 |
| Tianjin Zhongxin Chemtech Co., Ltd. | +86-022-66880623 +8618622897568 | sales@tjzxchem.com | China | 559 | 58 |
| Shanxi Naipu Import and Export Co.,Ltd | +86-13734021967 +8613734021967 | kaia@neputrading.com | China | 1011 | 58 |
| career henan chemical co | +86-0371-86658258 | sales@coreychem.com | China | 29914 | 58 |
| SHANDONG ZHI SHANG CHEMICAL CO.LTD | +86 18953170293 | sales@sdzschem.com | China | 2931 | 58 |
| Hubei Jusheng Technology Co.,Ltd. | 18871490254 | linda@hubeijusheng.com | CHINA | 28180 | 58 |
View Lastest Price from Acryloyl chloride manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2023-12-26 | Acryloyl chloride
814-68-6
|
US $100.00-1.00 / KG | 1KG | 99% | g-kg-tons, free sample is available | Henan Fengda Chemical Co., Ltd | |
 |
2023-08-04 | Acryloyl chloride
814-68-6
|
US $0.00-0.00 / kg | 1kg | 0.99 | 500000kg | Hebei Yibangte Import and Export Co. , Ltd. | |
 |
2023-08-04 | Acryloyl chloride
814-68-6
|
US $0.00-0.00 / kg | 1kg | 0.99 | 500000kg | Hebei Yibangte Import and Export Co. , Ltd. |
-

- Acryloyl chloride
814-68-6
- US $100.00-1.00 / KG
- 99%
- Henan Fengda Chemical Co., Ltd
-

- Acryloyl chloride
814-68-6
- US $0.00-0.00 / kg
- 0.99
- Hebei Yibangte Import and Export Co. , Ltd.
-

- Acryloyl chloride
814-68-6
- US $0.00-0.00 / kg
- 0.99
- Hebei Yibangte Import and Export Co. , Ltd.