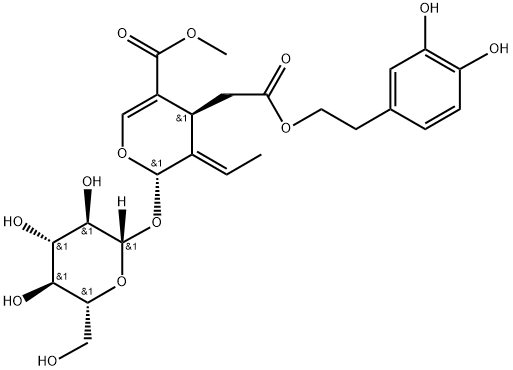
Oleuropein
- CAS No.
- 32619-42-4
- Chemical Name:
- Oleuropein
- Synonyms
- Olive Leaf Extract;2-(3,4-dihydroxyphenyl)ethyl [2S-(2alpha,3E,4beta)]-3-ethylidene-2-(beta-D-glucopyranosyloxy)-3,4-dihydro-5-(methoxycarbonyl)-2H-pyran-4-acetate;2H-Pyran-4-acetic acid, 3-ethylidene-2-(beta-D-glucopyranosyloxy)-3,4-dihydro-5-(methoxycarbonyl)-, 2-(3,4-dihydroxyphenyl)ethyl ester, [2S-(2alpha,3E,4beta)];Methyl (4S,5E,6S)-4-[2-[2-(3,4-dihydroxyphenyl)ethoxy]-2-oxoethyl]-5-ethylidene-6-[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy-4H-pyran-3-carboxylate;A688;A-688;OLEUROPEIN;Oleuropein ext;Oleuropein CRS;Oleuropein, >
- CBNumber:
- CB9154313
- Molecular Formula:
- C25H32O13
- Molecular Weight:
- 540.52
- MDL Number:
- MFCD16879008
- MOL File:
- 32619-42-4.mol
- MSDS File:
- SDS
| Melting point | 89-90°C |
|---|---|
| alpha | D20 -147° (H2O, alcohol, or acetone); D20 -127° |
| Boiling point | 772.9±60.0 °C(Predicted) |
| Density | 1.50±0.1 g/cm3(Predicted) |
| storage temp. | Inert atmosphere,2-8°C |
| solubility | DMSO (Slightly), Ethanol (Slightly, Sonicated), Methanol (Slightly) |
| pka | 9.70±0.10(Predicted) |
| color | Light Beige to Dark Brown |
| Stability | Hygroscopic, Light Sensitive |
| InChIKey | RFWGABANNQMHMZ-JUVTZQQOSA-N |
| SMILES | C([C@@H]1C(=CO[C@@H](O[C@@]2([H])[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O2)O)/C/1=C/C)C(=O)OC)C(=O)OCCC1C=CC(O)=C(O)C=1 |&1:1,5,7,9,10,12,14,r| |
| LogP | -0.865 (est) |
| CAS DataBase Reference | 32619-42-4(CAS DataBase Reference) |
| FDA UNII | 2O4553545L |
SAFETY
Risk and Safety Statements
| Safety Statements | 24/25 |
|---|---|
| WGK Germany | 3 |
| HS Code | 29389090 |
Oleuropein price More Price(48)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | 12247 | Oleuropein ≥98.0% | 32619-42-4 | 10mg | $212 | 2024-03-01 | Buy |
| Cayman Chemical | 21220 | Oleuropein ≥98% | 32619-42-4 | 25mg | $57 | 2024-03-01 | Buy |
| Cayman Chemical | 21220 | Oleuropein ≥98% | 32619-42-4 | 50mg | $95 | 2024-03-01 | Buy |
| Sigma-Aldrich | Y0000805 | Oleuropein European Pharmacopoeia (EP) Reference Standard | 32619-42-4 | y0000805 | $220 | 2024-03-01 | Buy |
| Sigma-Aldrich | O8889 | Oleuropein Synthetic | 32619-42-4 | 500mg | $499 | 2024-03-01 | Buy |
Oleuropein Chemical Properties,Uses,Production
A kind of natural plant extract
Oleuropein is a kind of natural plant extract obtained mainly from leaves of the olive tree (Olea europaea), an evergreen tree in the genus Olea of the family Oleaceae. Olea europaea is a world’s famous woody oleiferous plant and fruit tree specie. Cultivars of the Olea europaea have high food values and contain abundant high-quality edible vegetable oil, the olive oil, which are famous and economically important subtropical fruit trees. Leaves of Olea europaea are broad, simple and opposite. The flower is white and epigynous, with a four-lobed corolla, two stamens and two stigmas. The ovary bears two chambers, with two ovules per chamber. The drupe is oval-shaped, containing oil. There are as many as 500 Olea europaea cultivars, of which about 140 have been widely planted. China is the hometown of Chinese olive (Canavium album), and also is the largest country in the world that cultivates Canavium album. In China, the Canavium album distributes mainly in the province of Fujian, and it is also cultivated in provinces such as Guangdong, Guangxi, Zhejiang, Sichuan, and Taiwan. In the world, the countries that cultivate Canavium album also include Thailand, Vietnam, Myanmar, Laos, the Philippines, India and Malaysia. Olea europaea is a light-loving plant, vigorous in growth and drought tolerant. It mainly distributes in the Mediterranean countries of Europe and the California region. Greece, Italy, Tunisia and Spain are its major producing countries. Presently, it has been introduced into countries all over the world for cultivation. In 1950s, a small amount of Olea europaea was introduced into China, and was cultivated in China's western and northwestern regions, such as Gansu, and other provinces and cities such as Shaanxi, Sichuan, Chongqing, Fujian, Guangxi, Guangdong, and Hunan. The original wild olive trees still can be found in mountain areas of Sichuan, Guizhou and Hunan of China.

Figure 1 is an image of the olive (Olea europaea) tree.
The olive leaf contains mainly the oleuropein and its glycosides, flavonoids and flavonoid glycosides, biflavonoids and biflavonoid glycosides, and low molecular weight tannins. Secoiridoids are main active ingredients of olive leaf. Secoiridoids is one of the cyclopentane monoterpenes derivatives generated from the cycloreversion of iridoids, and it only takes a small part of the iridoids. As an important non-toxic polyphenolic secoiridoid glycoside, oleuropein is widely present in plants of genera Olea, Syringa, Ligustrum, osmanthus and jasminum of the family Oleaceae. Studies have shown that oleuropein has antioxidant, anti-inflammatory, anti-bacterial, anti-viral, anti-cancer, anti-tumor and hypoglycemic effects, and has been gradually used in medicine, health food products, cosmetics and other industries. The application prospect is promising.
The above information is edited by the Chemicalbook of Liu Huimin.
Physicochemical property
Fine crystals (recrystallized from ethyl acetate), melting point 87-89℃, hygroscopic; optical rotation-147° (c=1, water, ethanol or propanol) and characteristic mutarotation (the optical rotation changed into-127 °, 9 hours after dissolved in water); easily soluble in ethanol, acetone, glacial acetic acid and 5% sodium hydroxide solution; soluble in water, alcohol, ethyl acetate and butyl acetate; and almost insoluble in ether, petroleum ether, chloroform and carbon tetrachloride. The product may decompose when exposed to air and sunlight, thus it is recommended to be sealed and stored under cold and dark conditions.
Extraction methods
The commonly used methods to extract oleuropein currently are lixiviation, ultrasonic extraction, microwave-assisted extraction, supercritical CO2 extraction, dispersive liquid-liquid micro-extraction, superheated liquid extraction, and low-temperature vacuum distillation. A comprehensive analysis and comparison of different extraction methods was performed by Pujun Xie et al., who hold the opinion that the combination of ultrasonic technique and low-pressure vacuum is a method of high-efficiency and low energy-consumption that currently suitable for industrial production and easy to operate. Pujun Xie et al. also believes that the apparatus used in the extraction of natural products may be directly linked to such advanced analytical techniques as HPLC, GC-MS and other equipments in the future to realize online analysis and determination, which can not only increase the speed of analysis and determination, but also control the quality of the product, and which is also the future research on the extraction and separation of natural products.
Purification methods
1. Jianzhong Ye et al. obtained a crude extract of oleuropein using a reflux-heating extraction process, then purified it through AB-8 resin (the optimum condition for the purification was as follows: sample concentration, 2 g/l; eluent, 70% ethanol/water mixture; flow rate, 3 ml/min; eluent amount, 3 BV), and finally got the oleuropein with purity of up to 47.90%, flavonoid content of 16.4%, and the yield of 6.43%.
2. Shan Fu et al compared the adsorption of oleuropein among six different macroporous resins and found that the D101 macroporous resin presents good adsorption capacity and selectivity for oleuropein. The D101 macroporous resin was then used in the purification with the optimum adsorption condition as follows: sample concentration, 41.06 mg/ml; eluent, 50% ethanol; flow rate, 5ml/min; eluate amount, 260 ml. The resin can be used repeatedly. The purity of oleuropein can reach up to 55% or more after the purification by macroporous resin, and can reach up to 76% or more after further purification by dextran gel.
Determination methods
1. Jianzhang Dang et al. established a HPLC method for determining the oleuropein content in olive leaves. The column used was ZORBAX Eclipse XDB-C18 (4.6 mm x 250 mm, 5um), the mobile phase was 0.01 mol/l phosphate solution/acetonitrile at a ratio of 79/21, the flow rate was 0.8 ml/min, and the detection wavelength was set at 254 nm.
2. Yuanyuan Zheng established an indirect method for determining the oleuropein content of olive leave extract. As the total flavonoids and oleuropein are different in absorbance characteristics in two color systems, the content of oleuropein could be estimated indirectly by the UV-VIS subtraction method when determining the content of the total flavonoids. Zheng found that the indirect method was more accurate and reliable in determining the oleuropein content in olive leaves, compared with that of the HPLC method. Zheng also suggested that the indirect method is rapid, simple, and low in cost, and could replace the current HPLC method.
Pharmacological effect
According to the study, oleuropein has anti-viral, anti-tumor, antioxidant, antibacterial, hypoglycemic and antihypertensive effects. It is also effective in prevention of coronary heart disease and atherosclerosis. The application of oleuropein has been more and more widely with the discovery of a variety of pharmacological activities. Oleuropein of high purity has been used mainly in skin care products, which can protect skin cells from UV damage and effectively maintain the skin in soft and elastic status. Microherb oleuropein, which contains 80% of oleuropein, designed specifically for skin care products. As it contains high content of active ingredient and is light in color, the product is very suitable for cosmetic formulation.
Description
Oleuropein is a phenolic compound extracted from olive (Canarium album (Lour.) Raeusch., Gan Lan) leaves mainly. The olive tree belongs to family Oleaceae Olea genus of evergreen trees, and it is the world famous fruit and woody species. There is highly edible value for cultivating trees. Affiliated to famous economic forest and subtropical fruit trees, they are rich in olive oil which is considered as high-quality edible oil .
Chemical Properties
Brown Solid
Physical properties
Appearance: crystal in ethyl acetate. Solubility: very soluble in ethanol, acetone, glacial acetic acid, and 5% NaOH solution; soluble in water, butanol, ethyl acetate, and butyl acetate; insoluble in ether, petroleum ether, chloroform, and carbon tetrachloride. Melting point: 87–89 °C. Specific optical rotation: 147° in water/ethanol/ propane with a concentration of 1 mol/L; able to convert to 127° in water in 9 h.
History
The usage of olive ramification (including olive oil) in human health dates back to several centuries ago. For centuries, olive oil has been added to cosmetics and pharmacological agent. However, the unrefined mixture is applied primarily, and later studies have found that the main component of it which can be absorbed is olive polyphenols . Olive polyphenol, which gives a unique bitter taste, is mainly from the seeds, leaves, and immature bark of the olive (14% of dry weight). The content and purity are affected by fruit ripening stage, production, and extraction technology. In 1959, oleuropein was isolated, and the chemical structure was identified . In addition, oleuropein can be found in other kinds of plants, such as Lauraceae, Syringa, Ligustrum, Hibiscus and Jasminum. Up to now, the amount of Oleaceae containing oleuropein is 25 at least, including jasmine, cloves, lobular lilac, etc. Based on the molecular structure, pharmacological effects have been studied further. Clinical and experiment statistics demonstrated it is beneficial to human health. People gradually realized the function of its antioxidative, anti-inflammatory, and antitumor effects, atherosclerosis prevention, blood glucose hypoglycemia reduction, and nerve protection.
Uses
olive leaf extract is astringent and anti-septic. It also has vasodialating capacities, as well as being perfuming and skin-conditioning. olive leaf extract has demonstrated some anti-oxidant and freeradical scavenger activities. olive leaves contain both bioflavonoids and polyphenols. The extract generally found in anti-aging products.
Uses
Oleuropein is a tyrosol ester derivative of elenolic acid and phenolic glycoside found in olive oil. Oleuropein shows antioxidant, anti-ischemic and hypolipidemic activity. Oleuropein has also been used as an immunomodulator .
Definition
ChEBI: Oleuropein is a secoiridoid glycoside that is the methyl ester of 3,4-dihydro-2H-pyran-5-carboxylic acid which is substituted at positions 2, 3, and 4 by hydroxy, ethylidene, and carboxymethyl groups, respectively and in which the anomeric hydroxy group at position 2 has been converted into its beta-D-glucoside and the carboxylic acid moiety of the carboxymethyl substituent has been converted to the corresponding 3,4-dihydroxyphenethyl ester (the 2S,3E,4S stereoisomer). The most important phenolic compound present in olive cultivars. It has a role as a plant metabolite, a radical scavenger, an anti-inflammatory agent, an antineoplastic agent, an antihypertensive agent, a NF-kappaB inhibitor, an apoptosis inducer, an antioxidant and a nutraceutical. It is a secoiridoid glycoside, a beta-D-glucoside, a methyl ester, a member of catechols, a diester and a member of pyrans.
Pharmacology
According to the study, the pharmacological effects including antiviral, antitumor,
antioxidative, and antimicrobial and cardiovascular protection are investigated. In
cardiovascular system, olive leaf extract can reduce the discomfort caused by inadequate
arterial blood flow induced by angina and intermittent claudication. Oleuropein can decrease the degree of LDL oxidation and protect from coronary heart disease. The intensity of vascular smooth muscle can be decreased, and the ability of decreasing blood pressure can be achieved. Oleuropein regulates enzymes which participates in specific metabolism of protein, carbohydrate, and lipid and activates pepsin, trypsin, lipase, glycerol dehydrogenase, and GAPDH . The activity of other enzymes is also inhibited by oleuropein.
Oleuropein plays a role in resisting the virus; it can suppress VHSV and acts as a specific HIV inhibitor. It also shows significant activity against respiratory syncytial virus .
Oleuropein destroys the structure of actin filaments in cellular and noncellular detection and inhibits the proliferation and migration of tumor cells. The replication, movement, and invasion of tumor cells can be prevented. However, all of the above are reversible in normal cells .
Clinical Use
In the United States and other European countries, olive leaf extract serves as a dietary supplement, and its recommended dose is 50–100 mg as an immunomodulator. FDA also approves olive leaf extracts to be used as antioxidants in foods. In addition, 80% of oleuropein are used for skin care products mainly, and it can protect skin cells from UV. At the same time, it maintains soft and elastic skin effectively to protect skin. With light color and high content, oleuropein is very suitable for cosmetic formula design. In the cream and liquid formula, it is added at a concentration of 0.5–1%. The other pharmacological effects are still in preclinical study.
Oleuropein Preparation Products And Raw materials
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Changsha Staherb Natural Ingredients Co., Ltd. | +86-0731-84213302 +8618374838656 | sales@staherb.cn | China | 1025 | 58 |
| PNP Biotech Co. Ltd | +8618516098983 | sales@pnpbiotech.com | China | 1001 | 58 |
| Chengdu ChenLv Herb Co.,Ltd | +undefined13608205856 | maryextract@126.com | China | 127 | 58 |
| Hebei Mojin Biotechnology Co., Ltd | +8613288715578 | sales@hbmojin.com | China | 12457 | 58 |
| Hebei Yibangte Import and Export Co. , Ltd. | +86-0311-85030958 +8615532196582 | lisa@yibangte.com | China | 2987 | 58 |
| Shaanxi Haibo Biotechnology Co., Ltd | +undefined18602966907 | qinhe02@xaltbio.com | China | 1000 | 58 |
| Nantong Guangyuan Chemicl Co,Ltd | +undefined17712220823 | admin@guyunchem.com | China | 616 | 58 |
| Henan Fengda Chemical Co., Ltd | +86-371-86557731 +86-13613820652 | info@fdachem.com | China | 7613 | 58 |
| Capot Chemical Co.,Ltd. | 571-85586718 +8613336195806 | sales@capotchem.com | China | 29797 | 60 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21695 | 55 |
View Lastest Price from Oleuropein manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2024-04-23 | oleuropein
32619-42-4
|
US $0.00-0.00 / kg | 1kg | 5-30% | 1000kg | Changsha Staherb Natural Ingredients Co., Ltd. | |
 |
2024-04-23 | Olive Leaf Extract
32619-42-4
|
US $10.00 / kg | 1kg | 10%-50% | 10000 | Changsha Staherb Natural Ingredients Co., Ltd. | |
 |
2024-04-23 | Olive leaf extract | US $0.00 / KG | 1KG | 1-98% HPLC | 1000KG | Changsha Staherb Natural Ingredients Co., Ltd. |
-

- oleuropein
32619-42-4
- US $0.00-0.00 / kg
- 5-30%
- Changsha Staherb Natural Ingredients Co., Ltd.
-

- Olive Leaf Extract
32619-42-4
- US $10.00 / kg
- 10%-50%
- Changsha Staherb Natural Ingredients Co., Ltd.
-

- Olive leaf extract
- US $0.00 / KG
- 1-98% HPLC
- Changsha Staherb Natural Ingredients Co., Ltd.