クロフィブラート 化学特性,用途語,生産方法
外観
無色~わずかにうすい黄色, 澄明の液体
溶解性
エタノール、アセトン及びジエチルエーテルに極めて溶けやすく、水にほとんど溶けない。
解説
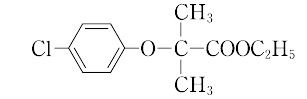
2-(4-chlorophenoxy)-2-methylpropanoic acid ethyl ester.C12H15ClO3(242.71).p-クロロフェノール,アセトン,水酸化ナトリウムをクロロホルム中で加熱還流後,得られたp-クロロフェノキシイソ酪酸をエチルエステル化すると得られる.
"無色~淡黄色の液体.沸点150 ℃(2.66 kPa).
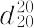
"1.137~1.144.
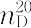
"1.500~1.505.水に不溶,有機溶媒に易溶.コレステロール生合成経路を阻害し,血中コレステロールを減少させる.高脂血症治療薬として使用される.LD50 1.95 g/kg(ラット,経口).[CAS 637-07-0]
森北出版「化学辞典(第2版)
用途
脂質の過酸化物の生理作用の研究。
医薬用
クロフィブラートは,抗高脂血症(脂質異常症)剤で、血中のコレステロールを低下させる。無色ないし淡黄色の澄明な油状の液体で、特異なにおいがあり、味は初め苦くのちに甘い。カプセル剤として用いられる。作用機序としては、コレステロールの合成を阻害し、中性脂肪を低下させ、尿酸の排泄(はいせつ)を促進させる作用を有する。1日0.75~1.5グラムを2~3回に分けて内服する。[幸保文治]
効能
脂質異常症治療薬, トリグリセリド合成阻害薬, ペルオキシソーム増殖因子活性化受容体(PPAR)α作動薬
商品名
クロフィブラート (鶴原製薬)
確認試験
(1) 本品のエタノール(99.5)溶液(1→10000)につき,紫外
可視吸光度測定法〈2.24〉により吸収スペクトルを測定し,
本品のスペクトルと本品の参照スペクトル1又はクロフィブ
ラート標準品について同様に操作して得られたスペクトルを
比較するとき,両者のスペクトルは同一波長のところに同様
の強度の吸収を認める.また,本品のエタノール(99.5)溶液
(1→100000)につき,紫外可視吸光度測定法〈2.24〉により吸
収スペクトルを測定し,本品のスペクトルと本品の参照スペ
クトル2又はクロフィブラート標準品について同様に操作し
て得られたスペクトルを比較するとき,両者のスペクトルは
同一波長のところに同様の強度の吸収を認める.
(2) 本品につき,赤外吸収スペクトル測定法〈2.25〉の液
膜法により試験を行い,本品のスペクトルと本品の参照スペ
クトル又はクロフィブラート標準品のスペクトルを比較するとき,両者のスペクトルは同一波数のところに同様の強度の
吸収を認める.
定量法
本品約0.5gを精密に量り,0.1mol/L水酸化カリウム・
エタノール液50mLを正確に加え,二酸化炭素吸収管(ソー
ダ石灰)を付けた還流冷却器を用いて水浴中でしばしば振り
混ぜながら2時間加熱する.冷後,直ちに過量の水酸化カリ
ウムを0.1mol/L塩酸で滴定〈2.50〉する(指示薬:フェノール
フタレイン試液3滴).同様の方法で空試験を行う.
0.1mol/L水酸化カリウム・エタノール液1mL
=24.27mg C
12H
15ClO
3
純度試験
(1) 酸 本品2.0gを中和エタノール100mLに溶かし,フ
ェノールフタレイン試液1滴及び0.1mol/L水酸化ナトリウム
液0.20mLを加えるとき,液の色は赤色である.
(2) 重金属〈1.07〉 本品2.0gをとり,第2法により操作し,
試験を行う.比較液には鉛標準液2.0mLを加える(10ppm以
下).
(3) ヒ素〈1.11〉 本品5.0gに硝酸20mL及び硫酸5mLを加
え,白煙が発するまで加熱する.必要ならば,冷後,更に硝
酸5mLを加え,白煙が発生するまで加熱し,この操作を液
が無色~淡黄色となるまで繰り返す.冷後,シュウ酸アンモ
ニウム飽和溶液15mLを加え,再び白煙が発生するまで加熱
し,冷後,水を加
えて25mLとする.この液5mLを検液とし,
試験を行う.
標準色:本品を用いないで同様に操作して調製した液
5mLを発生瓶にとり,ヒ素標準液2.0mLを加え,以下
検液の試験と同様に操作する(2ppm以下).
(4) 4-クロロフェノール 本品1.0gをとり,内標準溶液
1mLを正確に加え,更に移動相を加えて5mLとし,試料溶
液とする.別に4-クロロフェノール10mgをとり,ヘキサ
ン/2-プロパノール混液(9:1)に溶かし,正確に100mLと
する.この液10mLを正確に量り,ヘキサン/2-プロパノ
ール混液(9:1)を加えて正確に50mLとする.この液6mLを
正確に量り,内標準溶液4mLを正確に加え,更に移動相を
加えて20mLとし,標準溶液とする.試料溶液及び標準溶液
20μLにつき,次の条件で液体クロマトグラフィー〈2.01〉に
より試験を行う.それぞれの液の内標準物質のピーク面積に
対する4-クロロフェノールのピーク面積の比Q T及びQ Sを
求めるとき,Q TはQ Sより大きくない.
内標準溶液 4-エトキシフェノールの移動相溶液(1→
30000)
操作条件
検出器:紫外吸光光度計(測定波長:275nm)
カラム:内径約4mm,長さ約30cmのステンレス管に5
~10μmの液体クロマトグラフィー用シアノプロピル
シリル化シリカゲルを充てんする.
カラム温度:25℃付近の一定温度
移動相:ヘキサン/2-プロパノール/酢酸(100)混液
(1970:30:1)
流量:クロフィブラートの保持時間が約2分になるよう
に調整する.
カラムの選定:本品10.0g,4-クロロフェノール6mg
及び4-エトキシフェノール6mgをヘキサン1000mL
に溶かす.この液20μLにつき,上記の条件で操作す
るとき,クロフィブラート,4-クロロフェノール,
4-エトキシフェノールの順に溶出し,クロフィブラ
ートと4-クロロフェノールの分離度が5以上及び4-
クロロフェノールと4-エトキシフェノールの分離度
が2.0以上のものを用いる.
貯法
保存条件 遮光して保存する.
容器 気密容器.
強熱残分
0.1%以下(1g).
使用上の注意
光によって徐々に分解する
説明
Researchers in France observed in 1953 that structures derived
from dehydrocholic acid, phenylethyl acetic acid, and certain
other disubstituted acetic acids exhibited hypocholesterolemic
properties in rats and humans. Several years later, Thorp and
Waring discovered clof ibrate as an effective compound for
lowering lipids in animal models, with minimal toxicity. Its
mode of action was initially attributed to seasonal variations in
adrenal and thyroid function, and the administration of
androsterone was found to potentiate the hypocholesterolemic
effect of this compound. Subsequently, several clinical trials
were performed which showed that clofibrate decreases lipid
levels in hypercholesterolemic patients, mainly as the result of
a reduction in the very-low-density lipoprotein (VLDL), and
less in the low-density lipoprotein (LDL) fraction, and that the
coadministration of androsterone was not necessary for its
hypolipidemic effect. Despite reported hepatomegaly in rats
following long-term treatment with clofibrate, this drug was
approved in the United States in 1967 for the treatment of
hyperlipidemias.
Clofibrate can be chemically synthesized by the condensation
of phenol with ethyl 2-chloro-2-methylpropionate in the
presence of a dehydrochlorinating agent, followed by chlorination
and purification. It can also be synthesized by the
condensation of p-chlorophenol with acetone and chloroform
followed by esterifying the resultant acid to give clofibrate.
化学的特性
Clear Colorless Oil
使用
Clofibrate is a lipid-lowering agent (antilipidemic) used for
controlling high cholesterol (anticholesteremic) and triacylglyceride
levels in the blood. It increases lipoprotein lipase
activity to promote the conversion of VLDL to LDL, thereby reducing VLDL levels. It is indicated only in subjects with
increased concentrations of VLDL and intermediate-density
lipoproteins (IDL) who have failed to respond adequately to
gemfibrozil or nicotinic acid. Clofibrate is of limited utility for
patients with either familial hypercholesterolemia or polygenic
hypercholesterolemia, as comparatively more effective drugs
are available for lowering the concentration of LDL in these
patients.
Clofibrate has no effect on hyperchylomicronemia, nor
does it affect concentrations of high-density lipoproteins
(HDL). Thus, clofibrate appears to have specific efficacy only in
patients with familial type-III hyperlipoproteinemia. There is
no substantial evidence proving efficacy of clofibrate in preventing
deaths from coronary artery disease. Clofibrate has
been used to prevent or control polydipsia, polyuria, and
dehydration in a limited number of patients with mild to
moderate neurohypophyseal diabetes insipidus. A 5-year
multicenter study reported failure of clofibrate in reducing or
preventing mortality in cardiovascular disorders, which has
provided a setback for the prophylactic use of this drug.
定義
ChEBI: The ethyl ester of clofibric acid.
世界保健機関(WHO)
Clofibrate, an antihyperlipidaemic agent, was introduced in 1967
and was subsequently extensively studied in the primary and secondary prevention
of ischaemic heart disease. Following reports, published in 1978, of increased
mortality among patients receiving clofibrate in a WHO-sponsored cooperative trial
concerned with the primary prevention of ischaemic heart disease, the drug was
withdrawn in some countries and its approved indications were severely restricted in many others. These restrictions have become the norm for more recently
developed analogues of clofibrate.
(Reference: (WHODI) WHO Drug Information, 2, 6, 1979)
一般的な説明
Clofibrate, ethyl 2-(p-chlorophenoxy)-2-methylpropionate (Atromid-S), is a stable, colorless topale yellow liquid with a faint odor and a characteristictaste. It is soluble in organic solvents but insoluble in water.
Clofibrate is prepared by a Williamson synthesis, condensingp-chlorophenol with ethyl -bromoisobutyrate, or bythe interaction of a mixture of acetone, p-chlorophenol, andchloroform in the presence of excess potassium hydroxide.The acid obtained by either of these methods is esterified togive clofibrate. Both acid and ester are active; the latter, however,is preferred for medicinal use. Clofibrate is hydrolyzedrapidly to 2-p-chlorophenoxy-2-methylpropionic acid by esterasesin vivo and, bound to serum albumin, circulates inblood. The acid has been investigated as a hypolipidemicagent. It is absorbed more slowly and to a smaller extent thanis the ester. The aluminum salt of the acid gives even lowerblood levels than p-chlorophenoxy-2-methylpropionic acid.
危険性
Questionable carcinogen; toxic; causes
nausea, vomiting, diarrhea, weakness, stiffness,
cramps, and muscle tenderness.
生物活性
PPAR agonist (EC 50 values are 50, 500 and > 100 μ M at PPAR α , PPAR γ and PPAR δ respectively). Antihyperlipoproteinemic.
作用機序
The three structurally related fibrates available in the
United States are gemfibrozil (Lopid), fenofibrate
(Tricor) and clofibrate (Atromid-S).They share common
uses and toxicities. The fibrates typically lower VLDL
triglyceride by 40% or more and elevate plasma HDL
cholesterol by 10 to 15%. The reduction of plasma
triglycerides in humans appears due to increased lipoprotein
lipase (LPL) activity. The fibrates activate a nuclear
receptor (transcription factor) termed peroxisomal proliferation
activated receptor (PPAR) that is a member of
the steroid hormone receptor superfamily. PPAR increases
transcription of the LPL gene and decreases transcription
of the apolipoprotein CIII gene (apo CIII).
Since LPL is responsible for catabolism of VLDL triglyceride
and apo CIII is an inhibitor of LPL activity, the
combined consequences of these changes are increased
LPL activity and enhanced removal of triglyceride from
the circulation.
The elevation of HDL levels by fibrates may be due
to two drug actions: induced synthesis of apo-A1, the
principal apoprotein of HDL, and increased assembly
of new HDL particles in the circulation. Surface components
of VLDL contribute to formation of HDL, as
the VLDL particles are reduced in size through the action
of LPL.The increased rate of catabolism of VLDL
caused by the fibrates would provide more components
for assembly of HDL particles.
臨床応用
Clofibrate is the drug of choice in the treatment of typeIII hyperlipoproteinemias and may also be useful, to a lesserextent, in types IIb and IV hyperlipoproteinemias. The drugis not effective in types I and IIa.
Clofibrate can lower plasma concentrations of both triglyceridesand cholesterol, but it has a more consistent clinicaleffect on triglycerides. It also affects lipoprotein plasmalevels by enhancing removal of triglycerides from the circulationand causes reduction of VLDL by stimulatinglipoprotein lipase to increase the catabolism of this lipoproteinto LDL. Clofibrate lowers triglyceride levels in theserum much more than cholesterol levels and decreases levelsof FFAs and phospholipids. The lowering of cholesterollevels may result from more than one mechanism. Clofibrateinhibits the incorporation of acetate into the synthesis ofcholesterol, between the acetate and mevalonate step, by inhibitingsn-glyceryl-3-phosphate acyltransferase. Clofibratealso regulates cholesterol synthesis in the liver by inhibitingmicrosomal reduction of 3-hydroxy-3-methylglutaryl-CoA(HMG-CoA), catalyzed by HMG-CoA reductase. Clofibratemay lower plasma lipids by means other than impairment ofcholesterol biosynthesis, such as increasing excretionthrough the biliary tract.
副作用
The fibrates are generally well tolerated, with GI
distress being the most likely complaint. Other adverse
effects include myositis and erectile dysfunction, particularly
with clofibrate. There is ongoing concern about
the fibrates increasing the risk of gallstones, although
the extent of risk is unclear. Because clofibrate was associated
with increased mortality in early clinical trials,
it should be considered as a second-line drug.
安全性プロファイル
Poison by intravenous route.Moderately toxic by ingestion and other routes. Anexperimental teratogen. Other experimental reproductiveeffects. Reduces plasma lipid levels. Human systemiceffects by ingestion: muscle weakness, muscle spasms, andfever. Q
薬物相互作用
The fibrates potentiate the actions of the coumarin
anticoagulants, such as warfarin, so care should be taken
to reduce the dose of simultaneously administered anticoagulants,
and plasma prothrombin should be frequently
measured until the level stabilizes. As mentioned
earlier, great care should be given to combining
a statin with a fibrate, since this combination may increase
the risk of myositis and perhaps rhabdomyolysis.
環境運命予測
Clofibrate characteristically reduces plasma triglycerides by
lowering the concentration of VLDL within 2–5 days after
initiation of therapy. In a majority of patients, total cholesterol
and LDL concentrations in plasma fall slightly. However, some
patients who exhibit a large fall in VLDL may show a paradoxical
rise in LDL, resulting in minimal net effect on total
cholesterol levels.
The drug has several proposed antilipidemic actions,
including increased triglyceride and VLDL clearance, mobilization
of cholesterol from tissues, increased fecal excretion of
neutral sterols, decreased hepatic lipoprotein synthesis and/or
secretion, decreased free fatty acid release, and decreased
triglyceride synthesis. The precise mechanisms by which clofibrate
lowers serum concentrations of triglycerides and cholesterol
are not known.
代謝
The pro-drug fenofibrate undergoes rapid hydrolysis to produce fenofibric acid. This active metabolite can then be further metabolized
by oxidative or conjugative pathways. Gemfibrozil is slightly different in that it does not require initial bioactivation; however, similar to
fenofibric acid, it can be oxidized or conjugated. Oxidation of the aromatic methyl groups produces inactive hydroxymethyl and carboxylic
acid analogues. As a drug class, fibrates and their oxidized analogues are primarily excreted as glucuronide conjugates in the urine.
Oxidization requires the CYP3A4 isozyme; however, because of the ability of these compounds to be conjugated and eliminated either
with or without oxidation, drug interactions with other compounds affecting the CYP3A4 system are less important here than with other
drug classes.
クロフィブラート 上流と下流の製品情報
原材料
準備製品