| Company Name: |
BOC Sciences
|
| Tel: |
|
| Email: |
info@bocsci.com |
| Products Intro: |
Product Name:18-hydroxydesoxycorticosterone
CAS:379-68-0
|
| Company Name: |
ChemeGen(Shanghai) Biotechnology Co.,Ltd.
|
| Tel: |
18818260767 |
| Email: |
sales@chemegen.com |
| Products Intro: |
Product Name:18-hydroxy-11-deoxy Corticosterone
CAS:379-68-0
Purity:98% Package:10 mg;50 mg;100 mg;500 mg;1 g;5 g;10 g
|
| Company Name: |
Energy Chemical
|
| Tel: |
021-58432009 400-005-6266 |
| Email: |
marketing@energy-chemical.com |
| Products Intro: |
Product Name:11-Deoxy-18-hydroxycorticosterone
CAS:379-68-0
Purity:NULL Package:10mg;1mg;2.5mg;5mg Remarks:NULL
|
|
| | 18-HYDROXY-11-DEOXYCORTICOSTERONE Basic information |
| Product Name: | 18-HYDROXY-11-DEOXYCORTICOSTERONE | | Synonyms: | (8R,9S,10R,13R,14S,17S)-17-(2-hydroxyethanoyl)-13-(hydroxymethyl)-10-methyl-1,2,6,7,8,9,11,12,14,15,16,17-dodecahydrocyclopenta[a]phenanthren-3-one;(8R,9S,10R,13R,14S,17S)-17-glycoloyl-10-methyl-13-methylol-1,2,6,7,8,9,11,12,14,15,16,17-dodecahydrocyclopenta[a]phenanthren-3-one;11-DEOXY-18-HYDROXYCORTICOSTERONE;18-HYDROXY-11-DEOXYCORTICOSTERONE;18-HYDROXYDEOXYCORTICOSTERONE;18-HYDROXY DOC;18,20-EPOXY-20,21-DIHYDROXY-4-PREGNEN-3-ONE;18,21-DIHYDROXY-4-PREGNENE-3,20-DIONE | | CAS: | 379-68-0 | | MF: | C21H30O4 | | MW: | 346.46 | | EINECS: | 2068343 | | Product Categories: | | | Mol File: | 379-68-0.mol | 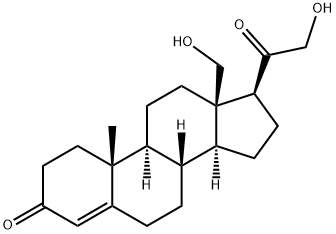 |
| | 18-HYDROXY-11-DEOXYCORTICOSTERONE Chemical Properties |
| Melting point | 171-173 °C | | storage temp. | 2-8°C | | solubility | Chloroform (Slightly, Sonicated), Methanol (Slightly, Heated) | | form | Solid | | color | White to Off-White | | LogP | 1.571 (est) |
| | 18-HYDROXY-11-DEOXYCORTICOSTERONE Usage And Synthesis |
| Description | 18-hydroxy-11-deoxy Corticosterone (18-OH-DOC) is a mineralocorticoid secreted by the zona fasciculata of the adrenal gland. Its biosynthesis is regulated by adrenocorticotropic hormone (ACTH; ) as well as angiotensin II , which increases 18-OH-DOC production in isolated human adrenal glomerulosa cells. 18-OH-DOC can be formed via conversion of 11-deoxy corticosterone (DOC; ) in human SK-MEL188 melanoma cells. 18-OH-DOC is an intermediate in the metabolism of progesterone and can be converted to aldosterone by the capsular portion of rat adrenal glands. Continuous infusion of 18-OH-DOC (200 μg/rat per day) increases systolic blood pressure in uninephrectomized saline-drinking rats. Plasma levels of 18-OH-DOC are elevated in a db/db mouse model of type 2 diabetes. | | Uses | 11-Deoxy-18-hydroxycorticosterone is an analog of Corticosterone (C695700); a glucocorticoid and intermediate in the biosynthesis of Aldosterone (A514700) which is an adrenocortical steroid isolated from the adrenal cortex. | | Definition | ChEBI: 18-hydroxydeoxycorticosterone is a 21-hydroxy steroid, a 3-oxo-Delta(4) steroid, a 20-oxo steroid and a 18-hydroxy steroid. | | Purification Methods | Recrystallise 18-hydroxy-11-deoxycorticosterone from Et2O/Me2CO to give crystals m 200-205o. When it is recrystallised from M2CO, it has m 191-195o. It has UV with max at 240nm. The 21-O-acetoxy-18-hydroxy derivative has m 158-159o (from Et2O/*C6H6), and the 21-O-acetoxy-18,20-epoxy derivative has m 149-154o (from Et2O). [Kahnt et al. Helv Chim Acta 38 1237 1955; Pappo J Am Chem Soc 81 1010 1959.] |
| | 18-HYDROXY-11-DEOXYCORTICOSTERONE Preparation Products And Raw materials |
|