- Entecavir
-

- $0.00 / 25kg
-
2024-04-12
- CAS:142217-69-4
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 2000ton
- Entecavir
-

- $0.00 / 1kg
-
2024-04-11
- CAS:142217-69-4
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 20tons
- Entecavir
-

- $0.00 / 25Kg/Bag
-
2024-04-10
- CAS:142217-69-4
- Min. Order: 2Kg/Bag
- Purity: 96% up
- Supply Ability: 20 tons
|
| | Entecavir Chemical Properties |
| Melting point | 249-252°C | | Boiling point | 661.4±65.0 °C(Predicted) | | density | 1.81±0.1 g/cm3(Predicted) | | storage temp. | -20°C | | solubility | DMSO (Slightly), Methanol (Sparingly) | | pka | 14.22±0.60(Predicted) | | form | powder | | color | white to beige | | optical activity | [α]/D +25 to +40°, c = 0.2 in H2O | | Stability: | Hygroscopic | | CAS DataBase Reference | 142217-69-4(CAS DataBase Reference) |
| | Entecavir Usage And Synthesis |
| Pharmacodynamics | Entecavir is a guanosine nucleoside analogue with selective activity against hepatitis B virus (HBV). It is designed to selectively inhibit the Hepatitis B virus, blocking all three steps in the replication process. Entecavir is more efficient than an older Hepatitis B drug, lamivudine.
| | Description | Entecavir is a cyclopentyl guanosine analog launched for the once-daily oral treatment of chronic hepatitis B virus (HBV) infection, and it is the third nucleoside or nucleotide analog to be marketed for this indication. Lamivudine, a deoxythiacytosine analog, and adefovir dipivoxil, a nucleotide analog, have been marketed since 1998 and 2002, respectively. Entecavir and adefovir are specifically indicated for HBV, whereas lamivudine is indicated for both HBV and HIV infections. | | Chemical Properties | White to Off-White/Yellow Crystalline Powder | | Originator | BMS (US) | | Uses | Entecavir is a new generation of guanine nucleoside analogues oral medicine for treatment of hepatitis B virus infection in, mainly for the treatment of adult patients with viral replication activity and serum transaminase continued to increase, or liver tissue for pathological activity of chronic hepatitis B, is currently down virus the fastest and the most powerful, the mutation rate lowest nucleoside analogues. | | Definition | ChEBI: Guanine substituted at the 9 position by a 4-hydroxy-3-(hydroxymethyl)-2-methylidenecyclopentyl group. A synthetic analogue of 2'-deoxyguanosine, it is a nucleoside reverse transcriptase inhibitor with selective antiviral activity against hepatitis B virus
Entecavir is phosphorylated intracellularly to the active triphosphate form, which competes with deoxyguanosine triphosphate, the natural substrate of hepatitis B virus reverse transcriptase, inhibiting every stage of the enzyme's activity, although it ha
no activity against HIV. It is used for the treatment of chronic hepatitis B. | | Trade name | Baraclude (BMS)
| | Mechanism of action | Entecavir is a nucleoside analog, or more specifically, a deoxyguanosine analogue that belongs to a class of carbocyclic nucleosides and inhibits reverse transcription, DNA replication and transcription in the viral replication process. | | Pharmacokinetics | Entecavir had a mean terminal half-life ranging from 128 to 149 hours and an effective half-life of approximately 24 hours. Elimination was predominantly through renal excretion, with mean urinary recovery ranging from 62% to 73%. | | Clinical Use | Treatment of chronic hepatitis B virus infection in patients >16 years of age.
Entecavir comes as a tablet and solution (liquid) to take by mouth. It is usually taken once a day on an empty stomach, at least 2 hours after a meal and at least 2 hours before the next meal. Take entecavir at around the same time every day. | | Side effects | The most common side effects of entecavir: the increase of ALT, fatigue, dizziness, nausea, abdominal pain, abdominal discomfort, abdominal discomfort, liver, muscle, insomnia, rubella and indigestion, also be found in neutrophils decreased slightly.
These adverse reactions were mild to moderate. It also found that, as the same type of antiviral drugs, entecavir and the first generation of antiviral drugs have similar side effects, such as acid poisoning, hepatomegaly, liver fatty degeneration in the withdrawal will appear rebound phenomenon. | | Synthesis | Entecavir is synthesized from 4-trimethylsilyl-3-butyn-2-one and acrolein. The key features of its preparation are: (1) a stereoselective boron–aldol reaction to afford the acyclic carbon skeleton of the methylenecylopentane moiety; (2) its cyclization by a Cp2TiCl-catalyzed intramolecular radical addition of an epoxide to an alkyne; and (3) the coupling with a purine derivative by a Mitsunobu reaction.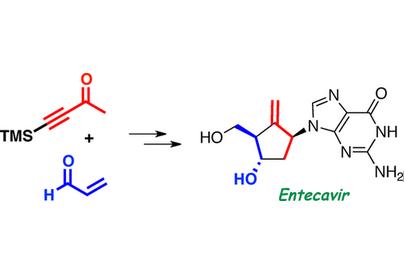 | | storage | Store at -20°C |
| | Entecavir Preparation Products And Raw materials |
|