- Ribavirin
-

- $0.00 / 1kg
-
2024-04-13
- CAS:36791-04-5
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 20tons
- Ribavirin
-

- $0.00 / 1KG
-
2024-03-16
- CAS:36791-04-5
- Min. Order: 100g
- Purity: 98%+
- Supply Ability: 100kg
- RibavirinPowder
-

- $0.00 / 1KG
-
2024-03-16
- CAS:
- Min. Order: 100g
- Purity: 98%+
- Supply Ability: 100kg
|
| | Ribavirin Chemical Properties |
| Melting point | 174-176°C | | alpha | D25 -36.5° (c = 1 in water); D20 -38° (c = 1 in water) | | Boiling point | 387.12°C (rough estimate) | | density | 1.4287 (rough estimate) | | refractive index | -36 ° (C=1, H2O) | | storage temp. | 2-8°C | | solubility | Freely soluble in water, slightly soluble in ethanol (96 per cent), slightly soluble or very slightly soluble in methylene chloride. It shows polymorphism (5.9). | | form | White solid | | pka | 12.95±0.70(Predicted) | | color | White | | Water Solubility | >=10 g/100 mL at 19 ºC | | Merck | 14,8198 | | BCS Class | 3 | | Stability: | Stable for 1 year from date of purchase as supplied. Solutions in DMSO or distilled water may be stored at -20°C for up to 3 months. | | InChIKey | IWUCXVSUMQZMFG-AFCXAGJDSA-N | | LogP | -2.260 (est) | | CAS DataBase Reference | 36791-04-5 | | EPA Substance Registry System | Ribavirin (36791-04-5) |
| | Ribavirin Usage And Synthesis |
| Antiviral drugs | Ribavirin is a novel non-selective nucleoside class broad-spectrum antiviral drug, belonging to the inosine monophosphate (IMP) dehydrogenase inhibitor. It can participate in the guanine participate in the human body metabolism, interfere with the biosynthesis of guanine, prevent the replication of the virus and has inhibitory effect on a variety of DNA and RNA viruses. Ribavirin has a stronger antiviral effect than amantadine and vidarabine, etc with definite efficacy and small drug side effects. Rare adverse reactions include conjunctivitis and hypotension with the former exhibiting as intraocular foreign body sensation, photophobia, itching, swelling and hematoma and the latter manifested as blurred vision, dizziness, and fatigue. It is used for clinical treatment of viral upper respiratory tract infection, respiratory syncytial virus-induced viral pneumonia and bronchitis, influenza, para-influenza, epidemic encephalris, mumps, chickenpox, shingles, herpes zoster, autumn diarrhea, children early-stage adenovirus pneumonia, acute Lassa fever, rubella, viral pneumonia, genital herpes, herpes simplex virus keratitis, psoriasis, etc with special efficacy. It also have certain efficacy on treating epidemic hemorrhagic fever, hepatitis B, and hepatitis A. Applying this drug in early stage of epidemic hemorrhagic fever can shorten the period, reducing the damage of kidney and vascular and symptoms of poisoning.
| | Pharmacological effects | 1, Ribavirin can be subject to phosphorylation in red blood cells to generate ribavirin monophosphate, diphosphate and triphosphate, wherein the ribavirin monophosphate is the strong inhibitor of the inosine monophosphate dehydrogenase which can inhibit cellular guanylate synthesis, decrease the tri-phosphorylation of the intracellular guanylate triphosphate, and blocking the synthesis of viral nucleic acid.
2, ribavirin triphosphate can inhibit the influenza virus RNA polymerase and interfere with viral replication.
3, ribavirin triphosphate can inhibit the synthesis of viral mRNA 5 'end and transcriptase, thereby inhibiting the DNA and RNA synthesis. It has inhibitory effect on various kinds of viruses (including DNA viruses and RNA viruses). It also has prevention and treatment effect on the influenza, adenovirus pneumonia, hepatitis, herpes, and measles and is also effective on treating the epidemic hemorrhagic fever.
4, it is mainly excreted through urine in the prototype with also a small amount of de-sugarized ribavirin metabolites with a small amount discharged from the feces.
The above information is edited by the Chemicalbook of Dai Xiongfeng.
| | Chemical Properties | This product is colorless or white crystalline powder and is odorless with no taste. It is stable in room temperature. It is easily soluble in water and slightly soluble in ethanol, chloroform and ether. It has its mp being 174-176 ℃ or 160-167 ℃ (refined products include two crystals).
| | Uses | It is a kind of broad-spectrum antiviral drugs for treating influenza (influenza A and B viruses), adenovirus pneumonia, hepatitis, herpes, measles with prevention and treatment effect; it is also effective on treating the epidemic hemorrhagic fever, particularly effective treatment of patients in their early stages. Oral administration can cause diarrhea without other significant toxicity but may cause neutropenia. Large dose can inhibit the maturation of hemoglobin and red blood cells, causing reversible anemia. There have been reports regarding to the teratogenicity and embryo toxicity in animal experiments. Pregnant women should be hanged.
Ribavirin is a broad-spectrum antiviral nucleoside drug used for treating influenza, adenovirus pneumonia of children, viral hepatitis, respiratory syncytial virus infection, acute keratitis, conjunctivitis, epidemic hemorrhagic fever and herpes zoster.
| | Production method | Take nucleotide or nucleoside as the starting material
First hydrolyze the guanosine and guanylate upon the action of glacial acetic acid and acetic anhydride to generate ribose-1-phosphate, which is then subject to the catalysis of double-para-nitro phenol to have reaction with triazide amide to generate condensate with aminolysis to obtain the product.
Guanosine (or guanylate) [acetic anhydride, glacial acetic acid]→[hydrolysis] ribose-1-phosphate [bis-(p-nitrophenol)-phosphate]→[triazide carboxamide] condensates [aminolysis]→Ribavirin
Enzymatic synthesis
First hydrolyze the guanosine and guanosine acid under the action of pyrimidine nucleoside phosphorylase to form ribose-1-phosphate which then, under the action of purine nucleoside phosphorylase, has reaction with triazide amide to directly generate triazole nucleosides.
Guanosine (or guanylate) [pyrimidine nucleoside phosphorylase] → ribose-1 nucleic acid [purine nucleotide cyclase] → Ribavirin.
| | Description | Ribavirin is a clinically useful antiviral medication for Hepatitis C, viral hemorrhagic fevers and other RNA and DNA viruses. The exact mechanism of its antiviral activity is uncertain. Various proposals include inhibition of viral polymerase1, RNA mutagenesis2, and inosine monophosphate dehydrogenase (IMPDH) inhibition3. Sensitizes tumor cells to anticancer agents 5-fluorouracil5 and doxorubicin6 via inhibition of eIF4E. Displays efficacy against atypical teratoid/rhabdoid tumors7 and inhibits glioma cell growth8. | | Chemical Properties | Colourless Solid | | Uses | Ribavirin is a guanosine analog with antiviral properties against DNA and RNA viruses, including respiratory syncytial virus, hepatitis C, and influenza. It acts as a prodrug that can be activated by either mono- or tri-phosphorylation by cellular kinases. These phosphorylated derivatives of ribavirin have diverse effects on both cellular and viral enzymes, resulting in suppression of viral replication. | | Uses | A broad spectrum antiviral nucleoside and inhibitor of inosine monophosphate dehydrogenase. | | Uses | anti-infective, anti-fungal | | Uses | Purine nucleoside analog; inhibits inosine monophosphate dehydrogenase (IMPDH). Used as an antiviral agent. | | Uses | aldosterone antagonist; antifibrogenic | | Indications | Ribavirin is a synthetic guanosine analogue that possesses
broad antiviral inhibitory activity against many
viruses, including influenza A and B, parainfluenza,RSV,
HCV, HIV-1, and various herpesviruses, arenaviruses,
and paramyxoviruses. Its exact mechanism of action has
not been fully elucidated; however, it appears to inhibit
the synthesis of viral mRNA through an effect on nucleotide
pools. Following absorption, host cell enzymes
convert ribavirin to its monophosphate, diphosphate,
and triphosphate forms. Ribavirin monophosphate inhibits the guanosine triphosphate (GTP) synthesis
pathway and subsequently inhibits many GTP-dependent
processes. Ribavirin triphosphate inhibits the 5 capping
of viral mRNA with GTP and specifically inhibits
influenza virus RNA polymerase. Ribavirin may also
act by increasing the mutation rate of RNA viruses, leading
to the production of nonviable progeny virions.
Ribavirin resistance has not been documented in clinical
isolates. | | Definition | ChEBI: A 1-ribosyltriazole that is the 1-ribofuranosyl derivative of 1,2,4-triazole-3-carboxamide. An inhibitor of HCV polymerase. | | Brand name | Copegus (Roche); Rebetol (Schering); Virazole (Valeant). | | Acquired resistance | Development of resistant virus strains has not been
demonstrated. | | General Description | White powder. Exists in two polymorphic forms. | | General Description | Ribavirin is 1-β-D-ribofuranosyl-1,2,4-thiazole-3-carboxamide.The compound is a purine nucleoside analog with amodified base and a D-ribose sugar moiety.
Ribavirin inhibits the replication of a very wide variety ofRNA and DNA viruses,68 including orthomyxoviruses,paramyxoviruses, arenaviruses, bunyaviruses, herpesviruses,adenoviruses, poxvirus, vaccinia, influenza virus(types A and B), parainfluenza virus, and rhinovirus. In spiteof the broad spectrum of activity of ribavirin, the drug hasbeen approved for only one therapeutic indication—thetreatment of severe lower respiratory infections caused byRSV in carefully selected hospitalized infants and youngchildren. | | Air & Water Reactions | Water soluble. | | Reactivity Profile | Ribavirin may be sensitive to prolonged exposure to light. | | Hazard | Mildly toxic by ingestion. An experimental
teratogen. | | Fire Hazard | Flash point data for Ribavirin are not available; however, Ribavirin is probably combustible. | | Pharmaceutical Applications | A synthetic nucleoside. It is neither a classic pyrimidine nor a
purine, but stereochemical studies indicate that it is a guanosine
analog. It is usually formulated for administration by inhalation,
but oral and intravenous preparations are also used. | | Biochem/physiol Actions | Antiviral agent used against a wide variety of human viral infections, in particular, chronic hepatitis?C, HIV, and adenovirus. Its metabolite, ribavirin 5′-phosphate, is an inhibitor of inosine monophosphate (IMP) dehydrogenase, but many other mechanisms of action are also supported with experimental evidence. | | Mechanism of action | Ribavirin, a guanosine analogue, has broad-spectrum antiviral activity against both DNA and RNA viruses. It is phosphorylated by adenosine kinase to the triphosphate, resulting in the inhibition of viral
specific RNA polymerase, disrupting messenger RNA and nucleic acid synthesis. | | Pharmacology | Oral and intravenous ribavirin are associated with
additional adverse effects.When given via these routes,
ribavirin can produce hemolytic anemia that is reversible
following dosage reduction or cessation of therapy.
When given in combination with interferon- , ribavirin
increases the incidence of many of its side effects,
such as fatigue, nausea, insomnia, depression, and anemia,
and may cause fatal or nonfatal pancreatitis.
Ribavirin is mutagenic, teratogenic, and embryotoxic in
animals at doses below the therapeutic level in humans.
It is contraindicated in pregnant women and in the male
partners of pregnant women. Women of childbearing
potential and male partners of these women must use
two effective forms of contraception during ribavirin
treatment and for 6 months post therapy. Pregnant
women should not directly care for patients receiving
ribavirin. | | Pharmacokinetics | Oral absorption: 36–46%
Cmax 3 mg/kg oral: 4.1–8.2 μmol/L after 1–1.5 h
600 mg intravenous: 43.6 μmol/L end infusion
Plasma half-life: c. 24 h
Volume of distribution: 647 L
Plasma protein binding: <10%
Absorption
It is rapidly absorbed after oral administration. Mean peak concentrations after 1 week of oral doses of 200, 400 and 800 mg every 8 h were 5.0, 11.1 and 20.9 μmol/L, respectively. Trough levels 9–12 h after the end of 2 weeks’ therapy were 5.1, 13.2 and 18.4 μmol/L, respectively, indicating continued accumulation of the drug. Drug was still detectable 4 weeks later. Mean peak plasma concentrations after intravenous doses of 600, 1200 and 2400 mg were 43.6, 72.3 and 160.8 μmol/L, respectively; at 8 h the mean plasma concentrations were 2.1, 5.6 and 10.2 μmol/L. Aerosolized doses (6 g in 300 mL distilled water) are generally administered at a rate of 12–15 mL/h using a Collison jet nebulizer, the estimated dosage being 1.8 mg/kg per h for infants and 0.9 mg/kg per h for adults. When administered by small particle aerosol for 2.5–8 h, plasma concentrations ranged from 0.44 to 8.7 μmol/L.
Metabolism and excretion
It is rapidly degraded by deribosylation or amide hydrolysis, and together with its metabolites is slowly eliminated by the kidney. About 50% of the drug or its metabolites appear in the urine within 72 h and 15% is excreted in the stools. The remainder seems to be retained in body tissues, principally in red blood cells, which concentrate the drug or metabolites to a peak at 4 days, with a half-life of around 40 days. After intravenous administration 19.4% of the dose was eliminated during the first 24 h (compared with 7.3% after an oral dose), the difference reflecting the bioavailability. | | Clinical Use | Ribavirin aerosol (Virazole) is indicated in the treatment
of high-risk infants and young children with severe
bronchiolitis or pneumonia due to RSV infection.
Treatment is most effective if begun within 3 days of the
onset of symptoms.
Although ribavirin monotherapy is ineffective
against HCV, oral ribavirin in combination with interferon-α
(Rebatron) is approved for this indication and is
effective in patients resistant to interferon therapy
alone. Intravenous ribavirin may be useful in the therapy
of Hantaan virus infection, Crimean or Congo virus
hemorrhagic fever, Lassa fever, and severe adenovirus
infection. | | Clinical Use | RSV infections in infants (by nebulizer) in emergency situations
(i.e. transplant recipients)
Lassa fever
Hepatitis C (in combination with interferon-α)
Use in RSV pneumonia in infants is no longer routine. It
reduces mortality from Hantaan virus, the agent responsible
for hemorrhagic fever with renal syndrome. | | Side effects | Most adverse effects associated with aerosol ribavirin
are local. Pulmonary function may decline if aerosol ribavirin
is used in adults with chronic obstructive lung disease
or asthma. Deterioration of pulmonary and cardiovascular
function has also been seen in severely ill
infants given this preparation. Rash, conjunctivitis, and
rare cases of anemia have been reported. Health care
workers exposed to aerosol ribavirin during its administration
have reported adverse effects including headache,
conjunctivitis, rash, and rarely, bronchospasm. | | Side effects | It is generally well tolerated, though adverse reactions appear
to be related to dose and duration of therapy. Minor adverse
reactions include metallic taste, dry mouth sensation and
increased thirst, flatulence, fatigue and CNS complaints,
including headache, irritability and insomnia. Daily doses of
1 g may cause unconjugated bilirubin levels to double and
the reticulocyte count to increase. Hemoglobin concentrations
may decrease with treatment or higher dosages; with
doses of 3.9–12.6 g per day, a drop in hemoglobin was noted
by days 7–13 of treatment, which was generally ‘rapidly’
reversible
on withdrawal of the drug, but in some instances
necessitated blood transfusion.
Aerosol administration of about 2 g in 36 or 39 h during
3 days is well tolerated, does not affect results of pulmonary
function tests, and seems non-toxic.
It is both teratogenic and embryotoxic in laboratory animals,
so precautions must be observed in women of childbearing
age. | | Synthesis | Ribavirin, 1-|?-D-ribofuranosyl-1H-1,2,4-triazol-3-carboxamide (36.1.28), is
synthesized by reacting methyl ester of 1,2,4-triazol-3-carboxylic acid with O-1,2,3,
5-tetraacetyl-|?-D-ribofuranose to make methyl ester of 1-O-2,3,5-tetraacetyl-|?-D-ribofura�nosyl-1,2,4-triazol-3-carboxylic acid (36.1.27), which is treated with an ammonia solution
of methanol to simultaneously dezacylate the carbohydrate part and amidation of the car�boxyl part of the product to give ribavirin. 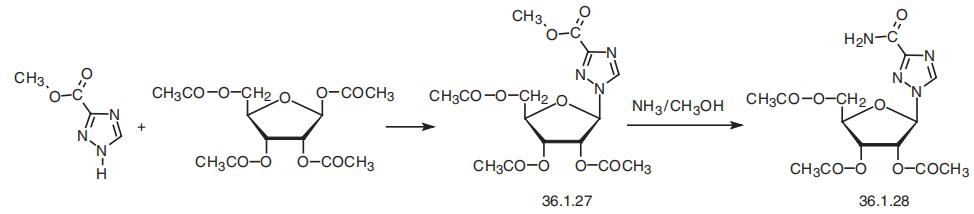
| | Drug interactions | Potentially hazardous interactions with other drugs
Antivirals: effects possibly reduced by abacavir;
increased risk of toxicity with stavudine; increased
side effects with didanosine - avoid; increased risk of
anaemia with zidovudine - avoid.
Azathioprine: possibly enhances myelosuppressive
effects of azathioprine. | | Metabolism | Ribavirin is metabolised by reversible phosphorylation
and a degradative pathway involving deribosylation and
amide hydrolysis to produce an active triazole carboxyacid
metabolite.
Ribavirin is mainly excreted in the urine as unchanged
drug and metabolites. | | storage | +4°C | | References | 1) Bougie and Bisaillon (2003), Initial binding of the broad spectrum antiviral nucleoside ribavirin to the hepatitis C virus RNA polymerase; J. Biol. Chem. 278 52471
2) Crotty et al. (2000), The broad-spectrum antiviral ribonucleoside ribavirin is an RNA virus mutagen; Nat. Med. 6 1375
3) Zhou et al. (2003), The effect of ribavirin and IMPDH inhibitors on hepatitis C virus subgenomic replicon RNA; Virology 310 333
4) Couee and Tipton (1990), Inhibition of ox brain glutamate by perphenazine; Biochem. Pharmacol. 39 1167
5) Hu et al. (2019), Ribavirin sensitizes nasopharyngeal carcinoma to 5-fluorouracil through suppressing 5-fluorouracil-induced ERK-dependent-elF4E activation; Biochem. Biophys. Res. Commun. 513 862
6) Tan et al. (2018), Ribavirin augments doxorubicin’s efficacy in human hepatocellular carcinoma through inhibiting doxorubicin-induced elF4E activation; J. Biochem. Mol. Toxicol. 32(1) e22007
7) Casaos et al. (2018), Ribavirin as a potential therapeutic for atypical teratoid/rhabdoid tumors; Oncotarget; 9 8054
8) Volpin et al. (2017) Use of an anti-viral drug, Ribavirin, as an anti-glioblastoma therapeutic; Oncogene, 36 3037 |
| | Ribavirin Preparation Products And Raw materials |
| Raw materials | SULPHOSUCCINIC ACID ESTER-->Ribonucleic acid-->Inosine-->5'-Guanylic acid-->Guanosine-->D-Ribose-->L-DIHYDROOROTIC ACID-->PHOSPHORYLASE B FROM RABBIT MUSCLE-->NUCLEOSIDE PHOSPHORYLASE BACTERIAL | | Preparation Products | 6-benzylaminopurine hydrochloride-->ribavirin 2',3',5'-triacetate-->1-[3,5-O-[1,1,3,3-Tetrakis(1-Methylethyl)-1,3-disiloxanediyl]-β-D-ribofuranosyl]-1H-1,2,4-triazole-3-carboxaMide-->2',3'-Isopropylidene Ribavirin |
|