- Dronedarone Impurities
-
- $0.00 / 10mg
-
2024-04-10
- CAS:141626-36-0
- Min. Order: 10mg
- Purity: 90%+
- Supply Ability: 10g
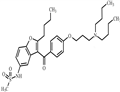
- Dronedarone
-

- $165.00 / 1KG
-
2023-03-27
- CAS:141626-36-0
- Min. Order: 1KG
- Purity: 0.99
- Supply Ability: 3t
- Dronedarone
-

- $0.00 / 20KG
-
2021-11-15
- CAS:141626-36-0
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 200000pcs
|
| | Dronedarone Basic information |
| Product Name: | Dronedarone | | Synonyms: | Dronedarone;N-(2-Butyl-3-(4-(3-(dibutylamino)propoxy)benzoyl)-5-benzofuranyl)methanesulfonamide;N-(2-butyl-3-(4-(3-(dibutylaMino)propoxy)benzoyl)benzofuran-5-yl)MethanesulfonaMide;MethanesulfonaMide,N-[2-butyl-3-[4-[3-(dibutylaMino)propoxy]benzoyl]-5-benzofuranyl]-;CS-294;Dronedarone
(SR-33589);N-[2-butyl-3-[[4-[3-(dibutylamino)propoxy]phenyl]-oxomethyl]-5-benzofuranyl]methanesulfonamide;DRONEDARONE BASE | | CAS: | 141626-36-0 | | MF: | C31H44N2O5S | | MW: | 556.76 | | EINECS: | | | Product Categories: | Cardiovascular APIs | | Mol File: | 141626-36-0.mol | 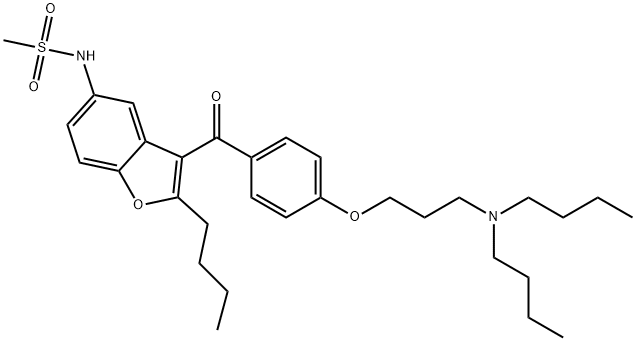 |
| | Dronedarone Chemical Properties |
| Melting point | 65.3° | | Boiling point | 683.9±65.0 °C(Predicted) | | density | 1.143±0.06 g/cm3(Predicted) | | storage temp. | 2-8°C | | solubility | ≥27.84 mg/mL in DMSO; insoluble in H2O; ≥49.8 mg/mL in EtOH | | form | solid | | pka | 7.40±0.30(Predicted) | | CAS DataBase Reference | 141626-36-0(CAS DataBase Reference) |
| | Dronedarone Usage And Synthesis |
| Description | AF is the most common form of sustained cardiac arrhythmia, with
an increasing prevalence in the aging population. AF accounts for
34.5% of arrhythmia-related hospital admissions in the United States.
The most significant consequences of AF include congestive heart failure, a 5-fold increased risk of stroke, and increased rate of mortality.
Although a 90% conversion rate from AF to normal sinus rhythm (NSR)
can be achieved with electrical cardioversion, up to 70% of these
patients require additional therapy with antiarrhythmic drugs in order
to maintain NSR.
Dronedarone, a close
analog of amiodarone, is structurally modified to provide improved
safety and pharmacokinetic profile. With the introduction of a sulfonamide group, dronedarone is less lipophilic, has lower tissue accumulation, and has a much shorter serum half-life (~24 h) compared with
amiodarone. Additionally, dronedarone lacks the iodine moieties that
are responsible for thyroid dysfunctions associated with amiodarone.
Dronedarone is specifically indicated to reduce the risk of cardiovascular hospitalization in patients with paroxysmal or persistent AF or AFL,
with a recent episode of AF/AFL and associated cardiovascular risk
factors, who are in sinus rhythm or who will be cardioverted. Similar to
amiodarone, dronedarone is a potent blocker of multiple ion currents
(including the rapidly activating delayed-rectifier potassium current,
the slowly activating delayed-rectifier potassium current, the inward rectifier potassium current, the acetylcholine-activated potassium current, peak sodium current, and L-type calcium current) and exhibits
antiadrenergic effects. Overall, dronedarone was well tolerated. The
most common side effects were gastrointestinal in nature and
included nausea, vomiting, and diarrhea. | | Originator | Sanofi-Aventis (US) | | Uses | Dronedarone-d9 is an isotopic labeled form of Dronedarone(D679445). Dronedarone is a drug used for the treatment of atrial fibrillation and atrial flutter in patients who have suffered cardiac arrhythmias. | | Definition | ChEBI: A member of the class of 1-benzofurans used for the treatment of cardiac arrhythmias. | | Brand name | Multaq | | Clinical Use | Anti-arrhythmic:
Maintenance of sinus rhythm after successful
cardioversion in adult clinically stable patients with
paroxysmal or persistent atrial fibrillation | | target | Calcium Channel | Sodium Channel | Potassium Channel | | Drug interactions | Potentially hazardous interactions with other drugs
Anti-arrhythmics: increased risk of myocardial
depression with other anti-arrhythmics; increased
risk of ventricular arrhythmias with amiodarone or
disopyramide - avoid.
Antibacterials: increased risk of ventricular
arrhythmias with clarithromycin, telithromycin and
erythromycin; concentration reduced by rifampicin
- avoid
Anticoagulants: increased anti-coagulant effect with
coumarins and phenindione; increased dabigatran
concentration - avoid; avoid with rivaroxaban;
concentration of edoxaban increased - reduce dose
of edoxaban.
Antidepressants: concentration possibly reduced by
St John’s wort - avoid; increased risk of ventricular
arrhythmias with tricyclic antidepressants,
citalopram and escitalopram - avoid.
Antiepileptics: concentration possibly reduced
by fosphenytoin, phenytoin, carbamazepine,
phenobarbital and primidone - avoid.
Antifungals: concentration increased by ketoconazole
- avoid; avoid with itraconazole, posaconazole and
voriconazole.
Antipsychotics: increased risk of ventricular
arrhythmias with antipsychotics that prolong the QT
interval; increased risk of ventricular arrhythmias
with phenothiazines - avoid.
Antivirals: avoid with ritonavir; increased risk of
ventricular arrhythmias with saquinavir - avoid.
Beta-blockers: increased risk of myocardial
depression; concentration of metoprolol and
propranolol possibly increased; increased risk of
ventricular arrhythmias with sotalol - avoid.
Calcium channel blockers: concentration increased
by nifedipine; increased risk of bradycardia and
myocardial depression with diltiazem and verapamil.
Cytotoxics: possibly increases bosutinib
concentration - avoid or consider reducing bosutinib
dose; possibly increases ibrutinib concentration -
reduce ibrutinib dose
Digoxin: increased concentration (halve digoxin
maintenance dose).
Fingolimod: possibly increased risk of bradycardia.
Grapefruit juice: concentration of dronedarone
increased - avoid.
Lipid-lowering drugs: concentration of atorvastatin
and rosuvastatin possibly increased; increased risk of myopathy with simvastatin; concentration of
lomitapide possibly increased - avoid.
Tacrolimus: manufacturer advises use with caution. | | Metabolism | Dronedarone is extensively metabolised in the liver,
mainly by the cytochrome P450 isoenzyme CYP3A4 to
a less active N-debutyl metabolite, and several inactive
metabolites
About 6% of an oral dose is excreted in the urine (entirely
metabolites) and 84% in the faeces (metabolites and
unchanged drug). |
| | Dronedarone Preparation Products And Raw materials |
|