- Lenvatinib
-

- $100.00 / 1kg
-
2024-04-23
- CAS:417716-92-8
- Min. Order: 1kg
- Purity: 99.99%
- Supply Ability: 100Tons
- Lenvatinib
-
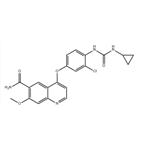
- $8905.00 / 1kg
-
2024-04-23
- CAS:417716-92-8
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 5000
- Lenvatinib
-

- $60.00 / 1kg
-
2024-01-02
- CAS:417716-92-8
- Min. Order: 10kg
- Purity: 0.99
- Supply Ability: 20tons
Related articles - Lenvatinib - pharmacodynamics
- Lenvatinib (LenvimaTM) is a multitargeted receptor kinase inhibitor that inhibits the kinase activities of vascular endothelia....
- Dec 13,2019
|
| Product Name: | Lenvatinib | | Synonyms: | 4-[3-Chloro-4-(cyclopropylaminocarbonyl)aminophenoxy]-7-methoxy-6-quinolinecarboxamide;E7080 (Lenvatinib);ER-203492-00;1-(4-(6-carbamoyl-7-methoxyquinolin-4-yloxy)-2-chlorophenyl)-3-cyclopropylurea;4-[3-Chloro-4-(cyclopropylaminocarbonyl)aminophenoxy]-7-methoxy-6-quinolinecarboxamide XEN445;E70801-(4-(6-carbamoyl-7-methoxyquinolin-4-yloxy)-2-chlorophenyl)-3-cyclopropylurea;4-(3-chloro-4-(cyclopropylaminocarbonyl)aminophenoxy)-7-methoxy-6-quinolinecarboxamide methanesulfonate;LENVATINIB | | CAS: | 417716-92-8 | | MF: | C21H19ClN4O4 | | MW: | 426.85 | | EINECS: | 1592732-453-0 | | Product Categories: | Inhibitors;FGF receptor antagonist;API;417716-92-8;Intermediate | | Mol File: | 417716-92-8.mol | 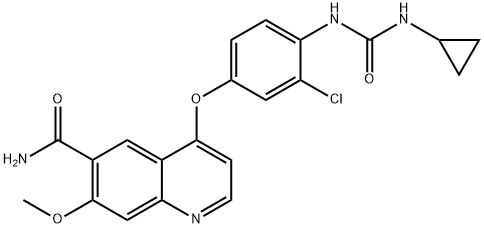 |
| | Lenvatinib Chemical Properties |
| Melting point | >216°C (dec.) | | Boiling point | 627.2±55.0 °C(Predicted) | | density | 1.46 | | storage temp. | -20°C | | solubility | Soluble in DMSO (up to 20 mg/ml) | | form | solid | | pka | 13.09±0.70(Predicted) | | color | White | | Stability: | Stable for 1 year from date of purchase as supplied. Solutions in DMSO may be stored at -20°C for up to 3 months. | | CAS DataBase Reference | 417716-92-8 |
| | Lenvatinib Usage And Synthesis |
| Thyroid cancer drugs |
Lenvatinib(417716-92-8) is a thyroid cancer drug developed by Eisai Corporation of Japan (Code: E7080), belonging to the inhibitor of oral multi-receptor tyrosine kinase (RTK) and can inhibit the kinase activity of the vascular endothelial growth factor (VEGF) Receptors VEGFR1 (FLT1), VEGFR2 (KDR), and VEGFR3 (FLT4). Lenvatinib can also inhibit the involvement of other RTKs in pathological angiogenesis, tumor growth, and cancer progression except for their normal cellular functions including fibroblast growth factor (FGF) receptors FGFR1, 2, 3, and 4; platelet-derived growth factor receptor (PDGFR [alpha]), KIT, and RET.
[Indications]: Lenvatinib is suitable for the treatment of patients of thyroid cancer of local recurrence or metastasis type, progressivity type and radioactive iodine-refractory differentiated type.
On February 13, 2015, the US FDA approved anticancer drug Lenvatinib for the treatment of thyroid cancer. Lenvatinib is a multi-target enzyme inhibitor, being capable of inhibiting the VEGFR2 and VEGFR3 (vascular endothelial growth factor receptor). The trade name of Lenvatinib is Lenvima.
On May 20, 2015, the European Medicines Agency (EMA) approved Lenvatinib for the treatment of invasive, locally advanced or metastatic differentiated (papillary, follicular, Hurthle type) thyroid cancer (DTC). In the trial, the median survival time for patients of radioactive iodine-refractory DTC treated with Lenvatinib was 18 months while the value for patients who take placebo is only 3 months.
In Europe, Lenvatinib will compete with Bayer's kinase inhibitor sorafenib (trade name: Nexavar), which had been respectively approved by EMA and the US Food and Drug Administration for the treatment of radioactive iodine refractory DTC in 2014 and 2013, respectively. At the time, Sorafenib had been reported to be the first targeted therapeutics to be marketed for the past 40 years in the treatment of refractory DTCs. The marketing applications of Lenvatinib in Switzerland, South Korea, Canada, Singapore, Russia, Australia and Brazil are also under review.
Information regarding the pharmacological effects, clinical evaluation, and indication of the thyroid cancer drug Lenvatinib were compiled and edited by Tongtong from Chemicalbook (2015-09-22).
| | Biological activity | Lenvatinib (E7080) is a multi-target inhibitor with the most potent effect on VEGFR2 (KDR)/VEGFR3 (Flt-4) with an IC50 of 4 nM/5.2 and weaker effect on VEGFR1/Flt-1. The selectivity of targeting on VEGFR 2/ 3 is about 10 times as high as that of targeting on FGFR1 and PDGFRα/β. Phase 3. | | In vitro study | E7080 can effectively inhibit the angiogenesis, also significantly inhibit the VEGF/KDR and SCF/KIT signaling pathway. Based on the in vitro receptor tyrosine and serine/threonine kinase assays, the IC50 of E7080 in inhibiting Flt-1, KDR, and Flt-4 were 22, 4.0 and 5.2 nM, respectively. In addition to these kinases, E7080 can also inhibit FGFR1 and PDGFR tyrosine kinases, acting on FGFR1, PDGFRα and PDGFRβ with IC50 values of 46, 51 and 100 nM, respectively. When E7080 acted respectively on HUVECs stimulated by VEGF and VEGF-C, respectively, and effectively inhibit VEGFR2 and VEGFR3 phosphorylation with IC50 of 0.83 nM and 0.36 nM, respectively. Recent studies have shown that treatment with 1 μM and 10 μM and E7080, through inhibiting the FGFR and PDGFR signaling pathway, can significantly inhibit cell migration and invasion. | | In vivo studies | Orally administration of E7080 at a dose of 30 mg/kg and 100 mg/kg for the treatment of H146 xenograft tumor model can inhibit the tumor growth in a dose-dependent manner. At 100 mg/kg, it can result in tumor regression. Moreover, treatment with a dose of 100 mg/kg of E7080, compared with the VEGF antibody and Imatinib treatment, can reduce the microvascular density more. | | Feature | E7080 is an orally active multi-target kinase inhibitor. | | Description | Lenvatinib is an inhibitor of the receptor tyrosine kinases VEGF receptor 2 (VEGFR2) and VEGFR3 (IC50s = 4 and 5.2 nM, respectively). It also inhibits the related kinases VEGFR1, FGFR1, PDGFRα, PDGFRβ and Kit (IC50s = 22, 46, 51, 39, and 100 nM, respectively). Lenvatinib (30 mg/kg, twice per day) reduces tumor growth in an H146 small cell lung cancer mouse xenograft model and induces tumor regression when administered at a dose of 100 mg/kg twice per day. Formulations containing lenvatinib have been used in the treatment of differentiated thyroid cancer, renal cell carcinoma, and hepatocellular carcinoma. | | Uses | Lenvatinib is an orally active inhibitor of multiple receptor tyrosine kinases including VEGF, FGF and SCF receptors. | | Uses | E7080 (Lenvatinib) is a multi-target inhibitor of VEGFR2 and VEGFR3 with IC50 of 4 nM and 5.2 nM, respectively. | | Definition | ChEBI: A member of the class of quinolines that is the carboxamide of 4-{3-chloro-4-[(cyclopropylcarbamoyl)amino]phenoxy}-7-methoxyquinoline-6-carboxylic acid. A multi-kinase inhibitor and orphan drug used (as its mesylate salt) for the treatment of various types
of thyroid cancer that do not respond to radioiodine. | | Mechanism of action | Lenvatinib exerts its mechanism of action via inhibition of multiple receptors of tyrosine kinases: VEGFR-1 (FLT1), VEGFR-2( KDR), VEGFR-3 (FLT4), FGFR-1, FGFR-2, FGFR-3, FGFR-4, PDGFRa, RET, and c-KIT. Tumor growth is dependent on the development and proliferation of new blood vessels (neovascularization). Tumor growth and angiogenesis occur when the ligands bind to their respective tyrosine kinase receptors in the cellular membrane, initiating an intracellular signal transduction phosphorylation cascade promoting angiogenesis and cell proliferation. The inhibition of the VEGF receptors prevents tumor angiogenesis, and the inhibition of FGFR, RET, PDGFRα, and KIT prevents the further proliferation of malignant cells. The concurrent inhibition of both receptor pathways results in the inhibition of nuclear signal transduction and concomitant suppression of the activity of factors involved in tumor growth[5]. It inhibits vascular endothelial growth factor receptor family , fibroblast growth factor receptor family (FGFR1–4), platelet-derived growth factor receptor–alpha (PDGFRα), tyrosine-kinase receptor (KIT) and rearranged during transfection receptor (RET).
| | Side effects | The most common side effects of LENVIMA (lenvatinib) in people treated for thyroid cancer include tiredness; joint and muscle pain; decreased appetite; weight loss; nausea; mouth sores; headache; vomiting; rash, redness, itching, or peeling of your skin on your hands and feet; stomach (abdomen) pain; and hoarseness.
The most common side effects of LENVIMA when given with everolimus in people treated for kidney cancer include tiredness; joint and muscle pain; decreased appetite; vomiting; nausea; mouth sores; swelling in your arms and legs; cough; stomach (abdomen) pain; trouble breathing; rash; weight loss; and bleeding.
The most common side effects of LENVIMA in people treated for liver cancer include tiredness; decreased appetite; joint and muscle pain; weight loss; stomach (abdomen) pain; rash, redness, itching, or peeling of your skin on your hands and feet; hoarseness; bleeding; change in thyroid hormone levels; nausea. | | target | VEGFR-1 | | References | 1) Matsui et al. (2008), E7080, a novel inhibitor that targets multiple kinases, has potent antitumor activities against stem cell factor producing human small cell lung cancer H146, based on angiogenesis inhibition; Int. J. Cancer, 122 664
2) Matsui et al. (2008), Multi-kinase inhibitor E7080 suppresses lymph node and lung metastases of human mammary breast tumor MDA-MB-231 via inhibition of vascular endothelial growth factor-receptor (VEGF-R)2 and VEGF-R3 kinase; Clin. Cancer Res., 14 5459
3) Glen et al. (2011), E7080, a multi-targeted tyrosine kinase inhibitor suppresses tumor cell migration and invasion; BMC Cancer, 11 309
4) Yamamoto et al. (2014), Lenvatinib, an angiogenesis inhibitor targeting VEGFR/FGFR, shows broad antitumor activity in human tumor xenograft models associated with microvessel density and pericyte coverage; Vasc. Cell, 6 18
[5] Capozzi M, et al. Lenvatinib, a molecule with versatile application: from preclinical evidence to future development in anti-cancer treatment. Cancer Management and Research, 2019; 11: 3847–3860. |
| | Lenvatinib Preparation Products And Raw materials |
|