- Mycophenolate mofetil
-

- $0.00 / 1KG
-
2024-03-16
- CAS:115007-34-6
- Min. Order: 100g
- Purity: 98%+
- Supply Ability: 100kg
- Mycophenolate mofetil
-

- $50.00 / 1kg
-
2023-09-05
- CAS:115007-34-6
- Min. Order: 1kg
- Purity: 0.99
- Supply Ability: 20t
- Mycophenolate mofetil
-

- $150.00 / 1kg
-
2023-08-09
- CAS:115007-34-6
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 1000tons
|
| Product Name: | Mycophenolate mofetil | | Synonyms: | Mycophenolate mofetil###115007-34-6;MYCOPHENOLATE MOFETIL,98.0%~102%,ENTERPRISE STANDARD;AKOS 92025;4-HEXENOIC ACID,6-(1,3-DIHYDRO-4-HYDROXY-6-METHOXY-7-METHYL-3-OXO-5-ISOBENZOFURANYL)-4-METHYL-,2-(4-MORPHOLINYL)ETHYL ESTER;2-Morpholin-4-ylethyl (E)-6-(4-hydroxy-6-methoxy-7-methyl-3-oxo-1H-isobenzofuran-5-yl)-4-methyl-hex-4-enoate;Mycophenolate mofetil;Mycophenolate Mofetil(MMF);Matimaikaofenzhi | | CAS: | 115007-34-6 | | MF: | C23H31NO7 | | MW: | 433.49 | | EINECS: | 680-628-5 | | Product Categories: | Anti-cancer & immunity;API;115007-34-6 | | Mol File: | 115007-34-6.mol | 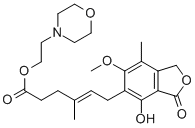 |
| | Mycophenolate mofetil Chemical Properties |
| solubility | Practically insoluble in water, freely soluble in acetone, sparingly soluble in anhydrous ethanol. | | BCS Class | 2 | | InChI | InChI=1S/C23H31NO7/c1-15(5-7-19(25)30-13-10-24-8-11-29-12-9-24)4-6-17-21(26)20-18(14-31-23(20)27)16(2)22(17)28-3/h4,26H,5-14H2,1-3H3/b15-4+ | | InChIKey | RTGDFNSFWBGLEC-SYZQJQIISA-N | | SMILES | C12COC(=O)C=1C(O)=C(C/C=C(\C)/CCC(=O)OCCN1CCOCC1)C(OC)=C2C | | CAS DataBase Reference | 115007-34-6(CAS DataBase Reference) |
| | Mycophenolate mofetil Usage And Synthesis |
| General description | Mycophenolate mofetil (MMF), whose trade name is “Cellcept”, was developed by the American Syntex Company. It is the semisynthetic derivative of mycophenocli acid (MPA) which is separated out from the glycolysis product of mold Penicillin glaucum. Mycophenolate mofetil is reusable cytostatics who have a history of clinical application being more than 10 years. It is mainly used for the treatment of psoriasis. At late 1980s, animal experiments have confirmed that it can significantly prolong the survival time of the allogeneic transplanted organs such as kidney, heart and islet of rodents. Sollinger et al has successfully applied MMF to the treatment of anti-rejection reaction upon kidney transplant for the first time with a remarkable effect. FDA has approved officially for its registration in 1995 under the trade name “Cellcept”, making clinical applications been added into a new and effective immunosuppressant for prevention and treatment of rejection reaction.
| | Pharmacology | After the oral administration of Mycophenolate mofetil, it has a rapid absorption rate in the upper gastrointestinal tract with the main absorption site being located in the stomach. At the same time, it is metabolized into pharmacologically active product of mycophenolic acid (the MPA) by the plasma lipase with the mean bioavailability being 94%. Plasma concentration immediately reaches peak one hour after orally taking the drug, followed by a rapid decline. MPA undergoes glucuronidation metabolism and be converted into stable, non-pharmacological active glucuronide (MPAG) in the liver, and further be excreted via urine. In the gut, the MPAG via biliary excretion enters into the small intestine with the glucosidase of gut microorganisms converting it into MPA, further being reabsorbed by the intestine for forming the enterohepatic circulation, thus reaching the second peak of plasma MPA (6 to 12 hours after administration). About 6% of MPAG is excreted out from the feces.
It undergoes in vivo metabolic activation via its hydrolysis and noncompetitively inhibits the activity of hypoxanthine monophosphate dehydrogenase, blocking the biosynthesis of guanine nucleotide, and thus playing its immunosuppressive effect on lymphocytes with both T lymphocytes and B lymphocytes being significantly affected.
| | Uses | It is mainly used for prevention the allograft rejection reaction upon organ transplant as mostly seen in renal transplant and also suitable in the heat, liver transplant especially for the refractory rejection reaction after the transplant. It can be combined together with cyclosporine and corticosteroids. It can also be applied to rheumatoid arthritis, systemic lupus erythematosus, primary glomerulonephritis, psoriasis and some other autoimmune diseases. Apply oral administration in kidney transplant; initial dose: 2~3mg/kg per day, maintenance dose: 1~2mg/ kg per day and take in 2 to 3 times. The dose can be adjusted according to specific condition. Rheumatoid arthritis: 300 mg daily.
| | synthetic route | Take mycophenolic acid (2) as the starting material, perform chlorination reaction through thionyl chloride, and then have esterification reaction with 4-(2-hydroxyethyl) morpholinem (4) to get mycophenolate mofetil (1).

Figure 1 the synthesis route of mycophenolate mofetil
Use (2) and (4) as a starting material, choose n-butyl ether or n-amyl ether as the solvent, directly prepare it by direct azeotropic dehydration (1).

Figure 2 The synthesis route of mycophenolate mofetil
For the above two processes, because of the use of n-butyl ether, n-amyl ether as a solvent, method two demands a higher production costs. Instead, although there are two steps of reactions of method one, acyl chloride product of the first step can be directly transferred into the next step without extra processing. The final product only need recrystallization before getting pure product.
The above information is edited by the Chemicalbook of Dai Xiongfeng.
| | Clinical application | 1. Organ Transplantation
Organ transplantation is transplanting the healthy organ of donor into another body for the rapid recovery of that function. The greatest threat of organ transplantation is the rejection reaction, mainly being presented as acute rejection reaction. Although currently there are relative mature protocols of immunosuppression such as the combined therapy of cyclosporine A, azathioprine, tacrolimus, cyclophosphamide, prednisone, and methylprednisolone, but there are still many cases that a considerable number of patients has got transplant rejection which ultimately lead to chronic rejection and transplant failure. In addition, adverse effects of immunosuppressive drugs also further restrict their clinical application. In 1995, people had successfully applied mycophenolate mofetil for organ transplant surgery to prevent and treat graft rejection. On May of the same year, FDA approved the application of mycophenolate mofetil in renal transplantation treatment. In 1996, it had been successfully applied for the first time in kidney transplant patients. Following researchers have found that mycophenolate mofetil in combination with cyclosporine and prednisone can reduce the incidence of acute rejection reaction after kidney transplantation, and also has good efficacy on refractory renal transplant rejection. Mycophenolate mofetil alone can be used as a kind of immunosuppressants for long-term maintenance therapy which can improve renal function and of special benefit for accepting elderly donors with relatively poor renal function.
2. Immune kidney disease
Immune kidney disease is a group of chronic glomerulus disease caused by variety triggering factors of same immune pathological characteristics. Immunological renal diseases includes anaphylactic purpura nephritis, refractory nephrotic syndrome, primary nephrotic syndrome, lupus nephritis, IgA nephropathy, etc., because of the dysfunction of patient's immune system, the resulting immune complex is deposited in the kidney which cause damage to the inherent cells of the kidney and cause inflammation reactions. This disrupts the normal function of kidney inherent cells, causing symptoms such as proteinuria, hematuria, and edema of the patients.
3. Digestive autoimmune diseases
(1) Autoimmune liver diseases Autoimmune liver diseases (ALD) is an autoimmune response mediated chronic liver disease. 20% of the ALD patients are not sensitive to the hormoneand azathioprine without appropriate treatment for long-term. Studies have shown that researchers believe that for ALD patients on whom traditional methods treatment has no effect, 88% of patients can alleviate the symptoms with applying mycophenolate mofetil. The application of mycophenolate mofetil can significantly reduce the amount of hormones.
(2) Autoimmune pancreatitis Autoimmune pancreatitis (AIP) is a pancreas inflammation lesion caused by the immune response triggered by the CD4+ adjuvant cells’ recognition to the antigen which is the own part of pancreas itself. Mycophenolate mofetil is used in patients of autoimmune pancreatitis mediated by hormone-dependent IgG4 and IgG4 mediated cholangitis. The related patient initially got symptoms alleviated by taking prednisone for 40mg/d. However, when the hormone concentration is reduced to 10mg/d, jaundice, hyperbilirubinemia, and elevated transaminase activities occurred. The patients were not able to tolerate the adverse reactions of adding azathioprine. Subsequent application of 750 mg mycophenolate mofetil for 2 times/d, being combined with 15 mg prednisone caused adverse reactions such as high blood sugar and other hormone-related adverse reactions after 3 months, thus gradually disabled prednisone and increase the dose of mycophenolate mofetil to the amount of 1g/times and 2 times/d. After 4 months, both the energy metabolism as well as blood sugar level of patients got alleviation without seeing any adverse reactions. Application of mycophenolate mofetil solves the problem that patients can’t use hormones. This demonstrates that mycophenolate mofetil or other immunosuppressants can be used to maintain or alleviate autoimmune pancreatitis.
4. Other diseases
(1) Refractory idiopathic thrombocytopenic purpura (ITP) is a common autoimmune bleeding disorder with a complicated pathogenesis and great difficulty of treatment. Traditional treatment usually adopt corticosteroids which although be effective on the majority of patients, however, still leaving some patients with poor results, called refractory idiopathic thrombocytopenic purpura (R-ITP). Mycophenolate mofetil as a new immunosuppressant with fewer adverse reactions, and is an effective approach of R-ITP therapy which has broad clinical application.
(2) Psoriasis is characterized by occurrence of varying sizes of papules skin, erythema with surface being covered with silvery white scales and also clear boundary on the surface of the skin. It mainly occurs in the scalp, extensor limbs and back. The etiology is not yet clear. In recent years, most scholars believed that it is related to heredity, infection, metabolic disorders, immune dysfunction, endocrine disorders and the environment. Studies have shown that there are no celar differences of safety and efficacy between methotrexate and Mycophenolate mofetil in the treatment of psoriasis, but mycophenolate mofetil has better security. Patients who can’t tolerate the side effects of methotrexate or with contraindications can choose mycophenolate mofetil.
(3) Myasthenia gravis (MG) is a chronic autoimmune disease caused by the dysfunction of conduction function between nerve conduction and neuromuscular. Immunosuppressant is important on the treatment of MG with its major mechanism of action of inhibiting the formation of auto-antibodies and reducing the related damage of cholinergic receptors or associated receptors in order to protect the smooth flow of neural excitation pathways, thereby improving the patient's muscle weakness. Compared with other immunosuppressant, mycophenolate mofetil has fewer side effects.
| | Pharmacokinetics | MMF is absorbed rapidly after oral administration with the intestinal wall, liver and other tissues being de-esterified and being quickly converted into active role of MPA. The average bioavailability of oral administration is about 94% of that of the intravenous injection (based on area under the curve of MPA). MMF can’t be detected in normal blood cycle after oral administration. Split off from the original drug, MPA formed inactive mycophenolic acid glucuronide by the metabolism of glucuronide transferase enzyme in the liver. The MPA concentration reaches peak 1h after oral administration, and reaches a second peak due to the enterohepatic circulation effect after 6~12h of medication. It has a biological half-life of 16~18h; at clinically effective concentration, 97% of MPA bound to plasma proteins. There is a very small amount of MMF (<1%) is excreted from the urine as MPA prototype from the urine with the majority (87%) being in the form of mycophenolic acid glucuronide excreted from urine, 6% being excreted from feces. MMF absorption is closely related to the liver function with smaller absorption with poorer liver function.
| | Side effects | Mainly include hypertension, atrial fibrillation, orthostatic hypotension, tachycardia, thrombosis, vasodilatation, headache, dizziness, insomnia, anxiety, high cholesterol, blood sugar changes, potassium and calcium metabolism disorders, acidosis, increased alkaline phosphatase, increased creatinine, hyperlipidemia, parathyroid dysfunction, nausea, vomiting, indigestion, and liver function abnormalities. Bone marrow suppression is commonly seen. Disable it upon severe neutropenia. The incidences of fungal dermatitis, skin rashes, Amblyopia and cataract are about 3% to 10%.
| | Precautions | 1. Contraindications: People who are allergic to this drug or mycophenolic acid (MPA); pregnant women and lactating women should be disabled.
2. Take with caution: Patients of severe active digestive diseases, bone marrow suppression, being accompanied by hypoxanthine-guanine phosphoribosyltransferase ribose kinase deficiency, severe heart, and liver dysfunction.
| | Chemical Properties | White or almost white, crystalline powder. | | Uses | Inhibitor of nucleic acid synthesis; immunomodulator. | | Uses | Mycophenolate Mofetil is an intermediate in the synthesis of (4Z)-Mycophenolate Mofetil (M831455), a degradation product of Mycophenolate mofetil, an immunosuppressant. | | Definition | ChEBI: Mycophenolate mofetil is a carboxylic ester resulting from the formal condensation between the carboxylic acid group of mycophenolic acid and the hydroxy group of 2-(morpholin-4-yl)ethanol. In the liver, it is metabolised to mycophenolic acid, an immunosuppressant for which it is a prodrug. It is widely used to prevent tissue rejection following organ transplants as well as for the treatment of certain autoimmune diseases. It has a role as an immunosuppressive agent, a prodrug, an EC 1.1.1.205 (IMP dehydrogenase) inhibitor and an anticoronaviral agent. It is a gamma-lactone, a member of phenols, an ether, a carboxylic ester and a tertiary amino compound. It is functionally related to a mycophenolic acid and a 2-(morpholin-4-yl)ethanol. | | Indications | Mycophenolate mofetil (CellCept), in conjunction with
cyclosporine and corticosteroids, has clinical applications
in the prevention of organ rejection in patients receiving
allogeneic renal and cardiac transplants. By effectively
inhibiting de novo purine synthesis, it can
impair the proliferation of both T and B lymphocytes.
Following oral administration, mycophenolate mofetil
is almost completely absorbed from the GI tract, metabolized
in the liver first to the active compound mycophenolic
acid, and then further metabolized to an inactive
glucuronide.
Early clinical trials indicate that mycophenolate
mofetil in conjunction with cyclosporine and corticosteroids
is a more effective regimen than azathioprine
in preventing the acute rejection of transplanted organs.
GI side effects are most common. | | Brand name | Cellcept (Roche). | | Side effects | The main side effects include an
increased incidence of herpes zoster, neutropenia, gastrointestinal symptoms,
and opportunistic infections. In the transplant literature, 1% to 2%
of patients will develop a lymphoproliferative malignancy at the doses
recommended for psoriasis. This is a teratogen and should be avoided in
women of childbearing age. |
| | Mycophenolate mofetil Preparation Products And Raw materials |
|