| Company Name: |
Rhawn Reagent
|
| Tel: |
400-400-1332688 18019345275 |
| Email: |
amy@rhawn.cn |
| Products Intro: |
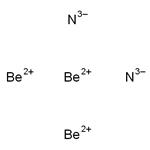
Product Name:triberyllium nitride
CAS:1304-54-7
|
- triberyllium nitride
-
- $8.00 / 1KG
-
2024-04-09
- CAS:1304-54-7
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: g-kg-tons, free sample is available
|
| | triberyllium nitride Basic information |
| | triberyllium nitride Chemical Properties |
| Melting point | 2200 ±40° | | Boiling point | volatile [MER06] | | density | 2.710 | | solubility | reacts with acid solutions, alkaline solutions | | form | gray refractory crystals | | color | gray refractory crystals, crystalline;
cubic | | Water Solubility | decomposes slowly in H2O, quickly in acids and alkalies to evolve NH3 [MER06] | | Crystal Structure | Cubic | | EPA Substance Registry System | Beryllium nitride (Be3N2) (1304-54-7) |
| RIDADR | 1566 | | HazardClass | 6.1(b) | | PackingGroup | III |
| | triberyllium nitride Usage And Synthesis |
| Description | Beryllium nitride has the molecular formula of Be3N2 and the molecular weight of 55.0652 g/mol. It can be prepared from the elements at high temperature (1100– 1500 °C). Be3N2 decomposes in vacuum into beryllium and nitrogen:
3Be +N2 → Be3N2
It is a white to yellow powder with a density of 2.71 g/cm3. Its melting point is 2208 °C and a boiling point of 2240 °C. Its CAS number is 1304-54-7. It is soluble in water and is readily hydrolyzed forming beryllium hydroxide and ammonia:
Be3N2 + 3H203→Be(OH)2 + 2NH3
It has two polymorphic forms: cubic a-Be3N2 with a defect anti-bixbyite structure (a = 8.1452 ), and hexagonal β-Be3N2. | | Chemical Properties | triberyllium nitride is hard, refractory, white crystals. Readily attached by strong alkali solutions, liberating ammonia. | | Physical properties | Hard white or grayish crystal.
Oxidizes in air above 600°C.
Slowly decomposes in water,
quickly in acids and alkalis with
evolution of NH3. | | Physical properties | Gray cubic crystal; hard and refractory; density 2.71 g/cm3; melts at 2,200°C; decomposes in acid or alkali; slowly reacts with water. | | Uses | triberyllium nitride productions of the radioactive carbon isotope1 4C for tracer uses. | | Preparation | Beryllium nitride may be prepared by heating beryllium metal powder with dry nitrogen in an oxygen-free atmosphere above 700°C.
3Be + N2→Be3N2 | | Hazard | Chronic inhalation of the powder can cause cancer and adverse reproductive effects. |
| | triberyllium nitride Preparation Products And Raw materials |
|