- 5-Aminofluorescein
-

- $1.00 / 1KG
-
2024-04-24
- CAS:3326-34-9
- Min. Order: 1KG
- Purity: 99.91%
- Supply Ability: 200000
- 5-Aminofluorescein
-

- $0.00 / 25KG
-
2023-06-30
- CAS:3326-34-9
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 50000KG/month
- 5-Aminofluorescein
-

- $20.00 / 1kg
-
2023-03-01
- CAS:3326-34-9
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 5000 Ton
|
| | 5-Aminofluorescein Basic information |
| Product Name: | 5-Aminofluorescein | | Synonyms: | 5-Amino-3',6'-dihydroxyspiro[isobenzofuran-1(3H),9'-(9H)xanthen]-3-one
Fluoresceinamine;5-amino-3',6'-dihydroxy-3H-spiro[isobenzofuran-1,9'-xanthen]-3-one;FLUORESCEINAMINE ISOMER I;FLUORESCEINAMINE, ISOMER 1;5-amino-2-(3-hydroxy-6-oxoxanthen-9-yl)benzoic acid;5-AMINO-3',6'-DIHYDROXYSPIRO[ISOBENZOFURAN-1(3H),9'-(9H)XANTHEN]-3-ONE;5-AMINOFLUORESCEIN ISOMER I;5-AMINOFLUORESCEIN | | CAS: | 3326-34-9 | | MF: | C20H13NO5 | | MW: | 347.32 | | EINECS: | 222-043-6 | | Product Categories: | marker;Fluorescent Labels and Indicators;Fluorescent Labels & Indicators | | Mol File: | 3326-34-9.mol | 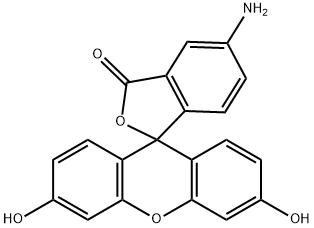 |
| | 5-Aminofluorescein Chemical Properties |
| Hazard Codes | Xn | | Risk Statements | 22-36/37/38 | | Safety Statements | 26 | | WGK Germany | 3 | | F | 10 | | TSCA | Yes | | HS Code | 32049090 |
| | 5-Aminofluorescein Usage And Synthesis |
| Chemical Properties | Dark Red Solid | | Uses | 5-aminofluorescein is also used to prepare FITC Isomer I (CDX-F0011, CDX-F0020). It is a chemical used as molecular probe and important as fluorescent dye and for derivatives. Fluorescent labeling reagent for proteins. Used in the fluorescent antibody technique for rapid identification of pathogens. | | Uses | Glycosaminoglycuronans react with 5-aminofluorescein to yield fluorescent derivatives. 5-aminofluorescein is also used to prepare FITC Isomer I (F-011, F-020). | | Definition | ChEBI: 5-aminofluorescein is a primary amino compound that is fluorescein carrying an amino substituent at C-5. Building block/intermediate for the synthesis of the fluorescent dye flourescein; also used to produce N-(fluorescein-5-yl)maleamic acid. It derives from a fluorescein (lactone form). | | Preparation |
The preparation of 5-aminofluorescein is carried out either by first saponification of 5-nitrofluorescein acetates with alcohol alkali, followed by reduction of the obtained 5-nitrofluorescein in a solution of alcohol alkali with hydrazine hydrate on a nickel catalyst to obtain 5-aminofluorescein , Or by boiling in an aqueous solution of sodium sulfide. The restoration of the nitro group and the saponification of acetates in the second case occurs in one stage.
| | General Description | Fluoresceinamine, isomer I belongs to the class of derivatized fluoresceins. |
| | 5-Aminofluorescein Preparation Products And Raw materials |
|