|
|
| | Bis(benzonitrile)palladium chloride Basic information | | Reaction |
| | Bis(benzonitrile)palladium chloride Chemical Properties |
| Melting point | 131 °C(lit.) | | storage temp. | Inert atmosphere,2-8°C | | solubility | Soluble in acetone, chloroform | | form | Crystalline Powder | | color | Orange to brown | | Water Solubility | insoluble | | Hydrolytic Sensitivity | 4: no reaction with water under neutral conditions | | BRN | 3981730 | | Exposure limits | NIOSH: IDLH 25 mg/m3 | | InChI | InChI=1S/2C7H5N.2ClH.Pd/c2*8-6-7-4-2-1-3-5-7;;;/h2*1-5H;2*1H;/q;;;;+2/p-2 | | InChIKey | WXNOJTUTEXAZLD-UHFFFAOYSA-L | | SMILES | C1(C#N)=CC=CC=C1.C1(C#N)=CC=CC=C1.[Pd](Cl)Cl | | CAS DataBase Reference | 14220-64-5(CAS DataBase Reference) |
| | Bis(benzonitrile)palladium chloride Usage And Synthesis |
| Reaction |
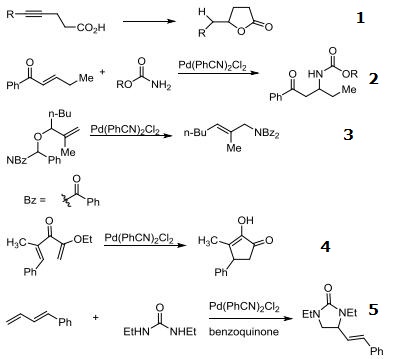
- Catalyst for the cyclization of δ-acetylenic carboxylic acids to butenolides.
- Catalyst for the aza-Michael reaction of carbamates with enones.
- Catalyst for the rearrangement of allylic imidates to allylic amides.
- Catalyst for the Nazarov cyclization of α-alkoxy dienones.
- Catalyst for the diamination of conjugated dienes.
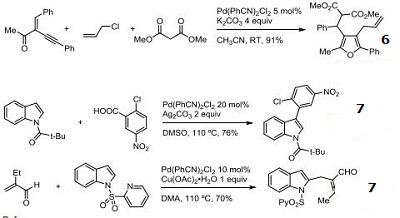
- Three component Michael addition, cyclization, cross-coupling reaction.
- C-H activation of indoles.
| | Chemical Properties | yellow powder | | Uses | Catalyst for greener amine synthesis from terminal olefins by Wacker oxidation followed by transfer hydrogenation of the resultant imine. | | Uses | suzuki reaction | | Uses | Bis(benzonitrile)palladium(II) chloride can be used as a catalyst:
- For greener amine synthesis from terminal olefins by Wacker oxidation, followed by transfer hydrogenation of the resultant imine.
- In cross-coupling reactions and α-O-glycosidation.
Formal anti-Markovnikov hydroamination of terminal olefins | | General Description | This product has been enhanced for catalytic efficiency. |
| | Bis(benzonitrile)palladium chloride Preparation Products And Raw materials |
|