| Company Name: |
Chemwill Asia Co.,Ltd.
|
| Tel: |
86-21-51086038 |
| Email: |
chemwill_asia@126.com |
| Products Intro: |
CAS:1705-60-8
Purity:99% Package:5KG;1KG;25KG
|
| Company Name: |
Meryer (Shanghai) Chemical Technology Co., Ltd.
|
| Tel: |
4006608290; 18621169109 |
| Email: |
market03@meryer.com |
| Products Intro: |
Product Name:2,2-Di(4,4-di(tert-butylperoxy)cyclohexyl)propane, 20 wt% solution in ethylbenzene
CAS:1705-60-8
Remarks:37859
|
|
| | 2,2-DI(4,4-DI(TERT-BUTYLPEROXY)CYCLOHEXYL)PROPANE, 20 WT% SOLUTION IN ETHYLBENZENE Basic information |
| Product Name: | 2,2-DI(4,4-DI(TERT-BUTYLPEROXY)CYCLOHEXYL)PROPANE, 20 WT% SOLUTION IN ETHYLBENZENE | | Synonyms: | 2,2-Bis(4,4-di-t-butylperoxy cyclohexyl) propane;PERKADOX 12-AE20;2,2-DI(4,4-DI(TERT-BUTYLPEROXY)CYCLOHEXYL)PROPANE, 20 WT% SOLUTION IN ETHYLBENZENE;2,2-DI(4,4-DI(TERT-BUTYLPEROXY)CYCLOHEXYL)PROPANE;2,2-bis(4,4-di-tert-Butyldioxycyclohexyl)propane;peroxide,[(1-methylethylidene)di-4-cyclohexanyl-1-ylidene]tetrakis[(1,1-dimeth;Peroxide,[(1-methylethylidene)di-4-cyclohexanyl-1-ylidene]tetrakis[(1,1-dimethylethyl);ylethyl) | | CAS: | 1705-60-8 | | MF: | C31H60O8 | | MW: | 560.8 | | EINECS: | 216-941-7 | | Product Categories: | | | Mol File: | 1705-60-8.mol | 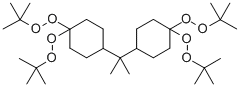 |
| | 2,2-DI(4,4-DI(TERT-BUTYLPEROXY)CYCLOHEXYL)PROPANE, 20 WT% SOLUTION IN ETHYLBENZENE Chemical Properties |
| Provider | Language |
|
ACROS
| English |
| | 2,2-DI(4,4-DI(TERT-BUTYLPEROXY)CYCLOHEXYL)PROPANE, 20 WT% SOLUTION IN ETHYLBENZENE Usage And Synthesis |
| Chemical Properties | Yellow liquid | | Reactivity Profile | Peroxides, such as 2,2-DI(4,4-DI(TERT-BUTYLPEROXY)CYCLOHEXYL)PROPANE, 20 WT% SOLUTION IN ETHYLBENZENE, are good oxidizing agents. Organic compounds can ignite on contact with concentrated peroxides. Strongly reduced material such as sulfides, nitrides, and hydrides may react explosively with peroxides. There are few chemical classes that do not at least produce heat when mixed with peroxides. Many produce explosions or generate gases (toxic and nontoxic). Generally, dilute solutions of peroxides (<70%) are safe, but the presence of a catalyst (often a transition metal such as cobalt, iron, manganese, nickel, or vanadium) as an impurity may even then cause rapid decomposition, a buildup of heat, and even an explosion. Solutions of peroxides often become explosive when evaporated to dryness or near-dryness. Danger of explosion when dry. May explode from heat, shock, friction or contamination. May ignite combustibles (wood, paper, oil, clothing, etc.). May be ignited by heat, sparks or flames. |
| | 2,2-DI(4,4-DI(TERT-BUTYLPEROXY)CYCLOHEXYL)PROPANE, 20 WT% SOLUTION IN ETHYLBENZENE Preparation Products And Raw materials |
|