- Dabigatran Etexilate
-

- $0.00 / 25Kg/Bag
-
2024-04-10
- CAS:211915-06-9
- Min. Order: 2Kg/Bag
- Purity: 99% up, High Density
- Supply Ability: 20 tons
- Dabigatran Etexilate
-

- $200.00 / 1kg
-
2023-06-26
- CAS:211915-06-9
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 1000kg/Months
- Dabigatran etexilate
-
- $0.00 / 1kg
-
2022-09-27
- CAS:211915-06-9
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 1000kg
|
| Product Name: | Dabigatran etexilate | | Synonyms: | Pradaxa (dabigatran);Dabigatran Etexilate (free base);Alanine, N-[[2-[[[4-[[[(hexyloxy)carbonyl]aMino]iMinoMethyl]phenyl]aMino]Methyl]-1-Methyl-1H-benziMidazol-5-yl]carbonyl]-N-2-pyridinyl-, ethyl ester;BIBR-1048(Dabigatran etexilate);BIBR 1048;Prazaxa;(E)-ethyl 3-(2-((4-(N'-(hexyloxycarbonyl)carbamimidoyl)phenylamino)methyl)-1-methyl-N-(pyridin-2-yl)-1H-benzo[d]imidazole-5-carboxamido)propanoate;BIBR-1048, >=99% | | CAS: | 211915-06-9 | | MF: | C34H41N7O5 | | MW: | 627.73 | | EINECS: | 606-722-8 | | Product Categories: | Pharmaceutical intermediate;API;Dabigatran etexilate;Other APIs | | Mol File: | 211915-06-9.mol | 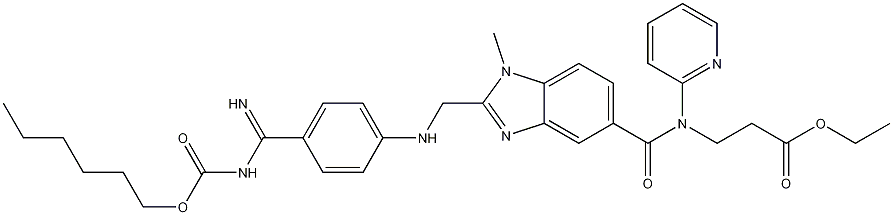 |
| | Dabigatran etexilate Chemical Properties |
| Melting point | 128-129° | | density | 1.24±0.1 g/cm3(Predicted) | | storage temp. | Sealed in dry,Store in freezer, under -20°C | | solubility | DMSO (Slightly), Methanol (Slightly) | | pka | 9.88±0.46(Predicted) | | form | Solid | | color | White to Pale Yellow | | Stability: | Hygroscopic |
| | Dabigatran etexilate Usage And Synthesis |
| Description | Dabigatran etexilate (Pradaxa, BIBR 1048, Boehringer Ingelheim) is the prodrug of dabigatran (originally known as BIBR 953), a potent, nonpeptidic small molecule that specifically and reversibly inhibits both free and clot-bound thrombin by binding to the active site of the thrombin molecule.
| | Indications and Usage | Dabigatran is the newest generation of oral anticoagulant drugs – direct thrombin inhibitors (DTIs), belonging to non-peptide thrombin inhibitors and instead specifically and selectively blocks the thrombin activity (free or bound) to prevent coagulation. Dabigatran targets the preventative and clinical treatment needs of acute and chronic thromboembolic disease.
| | Mechanisms of Action | It achieves anticoagulant effects through specific and selective inhibition of blood enzymes (free or bound). After oral ingestion and absorption through the digestive system, Dabigatran turns into Dabiga groups that have the direct anticoagulant abilities. By binding to thrombin in fibrin specific binding sites, Dabigatran stops fibrinogen from cleaving into fibrin, thus interrupting the blood clotting network and consequently thrombosis. Dabiga groups can also dissociate from the fibrin-thrombin coagulate and have an anticoagulating effect. Different from vitamin K antagonists, which affect different coagulants, Dabigatran can provide effective, predictable and stable anticoagulant effects. It will also have minimal reactions to other drugs or food, and does not require frequent monitoring or dosage adjustment.
| | Clinical Research | Compared to the control group treated with Warfarin, Dabigatran can significantly decrease the risk of stroke and embolic disease (including hemorrhagic stroke), greatly reduce the occurrence of bleeding (including fatal bleeding and intracranial hemorrhage), and greatly lower vascular mortality rate. Dabigatran provides effective and stable anticoagulant effects and does not require constant monitoring of anticoagulant functions or dosage adjustments.
| | Warnings and Precautions | 1. Dabigatran is not to be used by patients with severe kidney damage (CrCl<30ml/min).
2. Patients with high bleeding risk should also not use Dabigatran.
3. Patients must be tested and monitored for signs of lack of bleeding or anemia; if there is severe bleeding, cease use of Dabigatran and identify the bleeding area.
4. Dabigatran is not suitable for patients with significant manifestations of rheumatic valvar heart disease or patients with artificial heart valves.
| | Description | Dabigatran etexilate is an orally administered pro-drug of dabigatran,
which is a direct inhibitor of thrombin and a potent anticoagulant. The serine protease thrombin is the final mediator in the coagulation cascade
that leads to the production of fibrin, the main protein component of
blood clots. Thrombin is also a potent activator of platelets. Consequently, thrombin inhibitors have found utility as anticoagulants in
treating arterial and venous thrombosis.
Warfarin, although orally available, has a
narrow therapeutic index and requires frequent monitoring and dosage
adjustment. Low-molecular-weight heparins provide better safety profile
and less inter-patient variability; however, these agents are only available
for parenteral administration. Direct thrombin inhibitors (DTIs) are a
new class of anticoagulants that act by directly binding to thrombin at its
catalytic or fibrinogen-binding sites, or both. Unlike the heparins, DTIs do not require the activation of secondary factors such as antithrombin to derive their activity, which makes their action more predictable. In addition, their ability to inhibit both free and clot-bound thrombin predisposes them for enhanced anticoagulation effect. | | Originator | Boehringer Ingelheim (Germany) | | Uses | BIBR-1048 (Dabigatran) is an anticoagulant from the class of the direct thrombin inhibitors. It is being studied for various clinical indications and in many cases it offers an alternative to warfarin as the preferred orally administered blood thinner sin | | Uses | Dabigatran Etexilate is an oral anticoagulant and direct thrombin inhibitor. | | Definition | ChEBI: An aromatic amide obtained by formal condensation of the carboxy group of 2-{[(4-{N'-[(hexyloxy)carbonyl]carbamimidoyl}phenyl)amino]methyl}-1-methyl-1H-benzimidazole-5-carboxylic acid with the secondary amino group of
thyl N-pyridin-2-yl-beta-alaninate. A prodrug for dabigatran, a thrombin inhibitor and anticoagulant which is used for the prevention of stroke and systemic embolism. | | Brand name | Pradaxa | | Synthesis | The chemical synthesis of
dabigatran etexilate starts with the acylation of ethyl 3-(2-pyridylamino)propionate
with 4-(methylamino)-3-nitrobenzoyl chloride to produce
the corresponding amide. Subsequent reduction of the nitro group by
catalytic hydrogenation, and cyclization of the resultant phenylenediamine
with N-(4-cyanophenyl)glycine leads to a benzimidazole intermediate.
The cyano group is then transformed into an amidine by
employing the Pinner reaction. Finally, acylation of the amidino group
with hexyl chloroformate gives rise to dabigatran etexilate. | | in vitro | dabigatran showed anticoagulant effects in a concentration-dependent manner. it doubled the activated partrial thromboplastin time, prothrombin time and ecarin clotting in human ppp. [1] | | Drug interactions | Potentially hazardous interactions with other drugs
Analgesics: possible increased risk of bleeding with
NSAID’s; increased risk of haemorrhage with
ketorolac or IV diclofenac - avoid.
Anti-arrhythmics: concentration increased
by amiodarone, reduce dose of dabigatran;
concentration increased by dronedarone - avoid.
Antibacterials: concentration reduced by rifampicin
- avoid; possibly increased risk of bleeding with
clarithromycin.
Anticoagulants: increased risk of haemorrhage with
other anticoagulants - avoid.
Antidepressants: possible increased risk of bleeding
with SSRIs; concentration possibly reduced by St
John’s wort - avoid.
Antifungals: concentration increased by ketoconazole
and possibly itraconazole - avoid
Ciclosporin: concentration possibly increased by
ciclosporin - avoid.
Sulfinpyrazone: possible increased risk of bleeding.
Tacolimus: concentration possibly increased by
tacrolimus - avoid.
Ticagrelor: concentration of dabigatran increased.
Verapamil: reduce dose of dabigatran to 150 mg
daily, 75 mg in GFR=30-50mL/min. | | Metabolism | Dabigatran etexilate is a prodrug which does not exhibit
any pharmacological activity. After oral administration,
dabigatran etexilate is rapidly absorbed and converted to
dabigatran by esterase-catalysed hydrolysis in plasma and
in the liver. Dabigatran is a potent, competitive, reversible
direct thrombin inhibitor and is the main active principle
in plasma.
Mainly excreted in the urine (85%) and 6% via the faeces. | | references | 1. wienen w, stassen jm, priepke h et al. in-vitro profile and ex-vivo anticoagulant activity of the direct thrombin inhibitor dabigatran and its orally active prodrug, dabigatran etexilate. thromb haemost. 2007 jul;98(1):155-62.2. connolly sj, ezekowitz md, yusuf s et al. dabigatran versus warfarin in patients with atrialfibrillation. n engl j med. 2009 sep 17;361(12):1139-51. |
| | Dabigatran etexilate Preparation Products And Raw materials |
|