|
|
| | Fosteabine sodium Basic information |
| Product Name: | Fosteabine sodium | | Synonyms: | 2(1H)-Pyrimidinone, 4-amino-1-[5-O-[hydroxy(octadecyloxy)phosphinyl]-β-D-arabinofuranosyl]-, monosodium salt;Cytarabine ocfosfate;cytarabine ocphosfate;Fosteabine sodium;1-[5-O-[Stearyloxy(sodiooxy)phosphinyl]-β-D-arabinofuranosyl]cytosine;4-Amino-1-[5-O-[(sodiooxy)(octadecyloxy)phosphinyl]-β-D-arabinofuranosyl]-1,2-dihydropyrimidin-2-one;Cytarabin ocfosfate;Cytarabine ocphosphate | | CAS: | 65093-40-5 | | MF: | C27H49N3O8P.Na | | MW: | 597.661 | | EINECS: | | | Product Categories: | | | Mol File: | 65093-40-5.mol | 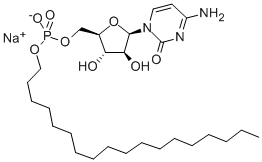 |
| | Fosteabine sodium Chemical Properties |
| | Fosteabine sodium Usage And Synthesis |
| Description | Cytarabine ocfosfate is the first orally active stearate derivative of cytarabine
introduced for the treatment of certain types of malignancies including myelodysplastic
syndromes that eventually evolve into acute leukemia. Cytarabine is one of the most effective
drugs for treating acute myeloid leukemia but suffers from poor bioavailability. As a monophosphorylated
analog of cytarabine, the title compound exhibits potent antitumor activity and
slowly converts to cytarabine in the liver, releasing cytarabine into the bloodstream over a
prolonged period. Cytarabine ocfosfate is effective by both parenteral and oral administration
and, therefore, has an advantage over the parenteral form of cytarabine or its analogs. It
should be noted that cytarabine ocfosfate has major side effects such as myelosuppression
and gastrointestinal toxicity. | | Originator | Yamasa (Japan) | | Definition | ChEBI: Cytarabine ocfosphate is a pyrimidine ribonucleoside monophosphate. | | Brand name | Starasid |
| | Fosteabine sodium Preparation Products And Raw materials |
|