- Rosuvastatin
-

- $0.00 / 1kg
-
2023-11-27
- CAS:287714-41-4
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 5o0kg
- Rosuvastatin
-
- $0.00 / 1KG
-
2023-11-01
- CAS:287714-41-4
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 300
- Rosuvastatin
-

- $0.00 / 25KG
-
2023-01-31
- CAS:287714-41-4
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 50000KG/month
Question and answer - Q:What is rosuvastatin?
- A:Rosuvastatin is a prescription drug. It comes in the form of a tablet you take by mouth.Rosuvastatin is available as the brand....
- Oct 10,2019
|
| Product Name: | Rosuvastatin | | Synonyms: | 7-[4-(4-Fluorophenyl)-6-(1-methylethyl)-2-(methyl-methylsulfonyl-amino)-pyrimidin-5-yl]-3,5-dihydroxy-hept-6-enoic acid;ROSUVASTATIN-D3 SODIUM SALT;(3R,5S,6E)-7-[4-(4-fluorophenyl)-2-(N-MethylMethanesulfonaMido)-6-(propan-2-yl)pyriMidin-5-yl]-3,5-dihydroxyhept-6-enoic acid;(3R,5S,E)-7-(4-(4-Fluorophenyl)-6-isopropyl-2-(N-methylmethylsulfonamido)pyrimidin-5-yl)-3,5-dihydroxyhept-6-enoic acid;R-2: TERT-BUTYL-(+) 7-[4-(4-FLUOROPHENYL)-6-ISOPROPYL-2-(N-METHYLSULFONYLAMINO) PYRIMIDINE-5-YL]-(3R, 5S)-DIOXANE-(E)-6-HEPTENATE;Rosuvastatin;Rosuvastatin Impurity 21;CS-111 | | CAS: | 287714-41-4 | | MF: | C22H28FN3O6S | | MW: | 481.54 | | EINECS: | 689-191-5 | | Product Categories: | Isotopically Labeled Pharmaceutical Reference Standard;API | | Mol File: | 287714-41-4.mol | 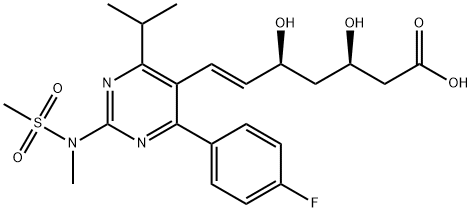 |
| | Rosuvastatin Chemical Properties |
| | Rosuvastatin Usage And Synthesis |
| Description | Known as an antilipemic agent, rosuvastatin belongs to the class of medications called statins, which is primarily used in the treatment of dyslipidemia, including high cholesterol and related conditions. It functions by blocking the enzyme that helps make cholesterol in the body, which is effective to improve cholesterol levels by reducing blood total cholesterol and triglyceride levels while raising the good cholesterol, HDL cholesterol levels in combination with a healthy diet and exercise program. It is also applied to treat people with certain inherited cholesterol disorders. Besides, since the high level of cholesterol is related to angiocardiopathy, rosuvastatin is beneficial to prevent people from cardiovascular diseases, which is used to reduce the risk of heart attacks, stroke, and angioplasty for people who have at least 2 risk factors for cardiovascular disease.
| | Indications and Usage | Rosuvastatin is a drug against hyperlipidemia, a HMG-CoA reductase inhibitor, successfully developed by British AstraZeneca. Applicable to treat a variety of lipid abnormalities, including primary hypercholesterolemia, mixed lipid abnormalities, simple hypertriglyceridemia, and senile coronary heart disease complicated by hyperlipidemia. Rosuvastatin is currently the statin with the strongest and most comprehensive lipid-lowering effect on the market, reducing LDL cholesterol and improve HDL function better than the world-recognized leader atorvastatin, and with better tolerability, fewer side effects, and unique pharmacokinetic effects. | | Mechanisms of Action | Rosuvastatin is a selective inhibitor of methylglutaryl coenzyme A reductase (HMG-CoA). The HMG-CoA reductase inhibitor is a rate-limiting enzyme which transforms 3-hydroxy-3-methylglutaryl coenzyme A into a methylpentate-cholesterol precursors. Its main site of action is the liver, reducing cholesterol in its target organ. It can competitively inhibit HMG-CoA in the liver to better reduce fat. It can also effectively promote the transfer of LDL into cells, thus enhancing the clearance of low-density lipoprotein, effectively reducing it. In addition, it can also inhibit platelet aggregation, reducing the body's inflammatory response, and protecting the function of endothelial cells, effectively stabilizing the plaque of coronary heart disease patients and reducing its effects. It increases the number of liver cell surface LDL receptors, promoting its absorption and catabolism, inhibiting hepatic synthesis of VLDL, thereby reducing the total count of VLDL and LDL particles.
For patients with homozygous and heterozygous familial and nonfamilial hypercholesterolemia or mixed dyslipidemia, it can reduce total cholesterol, LDL-C, ApoB, and non-HDL-C levels. It can also reduce TG levels and increase HDL-C levels. For patients with simple hypertriglyceridemia, Rosuvastatin can reduce total cholesterol, LDL-C, VLDL-C, ApoB, non HDL-C, and TG levels, and increase HDL-C levels.
| | Pharmacokinetics | Large liver uptake of orally administered Rosuvastatin, distribution volume around 134 L, and serum concentration peaks after 3-5 hours. Absolute bioavailability is 20%. Plasma protein binding rate (mainly albumin) is about 90%. Approximately 90% of the dose is excreted from feces (including absorbed and unabsorbed active substance) in the original form, with the rest discharged through urine. About 5% in the urine is in original form. Plasma clearance half-life is approximately 19 hours. Clearance half-life does not increase with dosage. The geometric mean of plasma clearance is about 50L/hour (coefficient of variation is 21.7%). As with other HMG-CoA reductase inhibitors, liver uptake of Rosuvastatin involves membrane transporter OATP-C, which is important for the removal of Rosuvastatin from the liver.
| | Drug Interactions |
- Cyclosporine: In combined usage, the AUC of Rosuvastatin is 7 times higher than that seen in healthy volunteers (using the same dosage). Co-administration does not affect cyclosporine plasma concentration.
- Vitamin K antagonists: As with other HMG-CoA reductase inhibitors, starting Rosuvastatin or gradually increasing dosage may increase International Normalized Ratio (INR) in patients simultaneously using vitamin K antagonists (such as Warfarin). Discontinuing use or gradually reducing dosage reduces INR. In this case, proper testing of INR is required.
- Combined administration of iferrate, fenofibrate, other fibrates, and lipid lowering dosages (≥1g/日) of niacin with HMG-CoA reductase inhibitors increases the risk of myopathy, possibly because their individual administration can also cause myopathy.
- Antacids: Combined administration with antacids containing aluminum magnesium hydroxide can reduce Rosuvastatin plasma levels by about 50%. This affect may be alleviated if the antacid is administered 2 hours later. The clinical significance of this drug interaction has not yet been studied.
- Erythromycin: Combined administration reduces AUC (0-t) of Rosuvastatin by 20%, and Cmax by 30%. This interaction may be caused by an increase in gastrointestinal motility caused by erythromycin.
- Oral contraceptives/Hormone Replacement Therapy (HRT): Administration combined with oral contraceptives increased ethinyl estradiol and norethindrone AUC by 26% and 34% respectively. These plasma concentrations should be considered when choosing dosages of oral contraceptives. There is no pharmacokinetic data for subjects who use this product with HRT, so the presence of similar interactions cannot be ruled out. In clinical trials, however, this combination is widespread and well tolerated.
| | Adverse Effects |
- Adverse reactions are usually mild and transient. Headaches, dizziness, constipation, nausea, abdominal pain, myalgia, and weakness are frequently observed.
- Dosage-related elevation of creatine kinase (CK) was observed in patients taking Rosuvastatin; most cases were mild, asymptomatic, and transient. If CK levels increase (>5×ULN), treatment should be discontinued.
- Effects on the liver: a dose-dependent increase of transaminase was observed in a small number of patients taking Rosuvastatin; most cases were mild, asymptomatic, and transient.
| | References | https://en.wikipedia.org/wiki/Rosuvastatin
http://www.medicinenet.com/rosuvastatin/article.htm
http://bodyandhealth.canada.com/drug/getdrug/apo-rosuvastatin
https://www.drugbank.ca/drugs/DB01098
| | Description | Rosuvastatin is the most recent FDA-approved statin (doses 5–40 mg) for the treatment of dyslipidemia. Rosuvastatin, which has a structure similar to other synthetic statins, has a long halflife (20–24 hours) similar to atorvastatin and hydrophilicity comparable to pravastatin. Consequently, rosuvastatin is minimally metabolized and has no significant cytochrome P450 drug interactions. The efficacy of rosuvastatin is superior to that of other statins. Compared to atorvastatin, rosuvastatin provides approximately an 8% additional lowering of LDL-C on an equivalent dose level. This provides modestly more efficacy than a doubling of the dose of atorvastatin. Rosuvastatin also increases HDL-C slightly more than atorvastatin, especially at the highest doses. | | Uses | Rosuvastatin Calcium is a competitive inhibitor of HMG-CoA reductase with IC50 of 11 nM. | | Definition | ChEBI: Rosuvastatin is a dihydroxy monocarboxylic acid that is (6E)-7-{4-(4-fluorophenyl)-2-[methyl(methylsulfonyl)amino]-6-(propan-2-yl)pyrimidin-5-yl} hept-6-enoic acid carrying two hydroxy substituents at positions 3 and 5 (the 3R,5S-diastereomer). It has a role as an antilipemic drug, an anti-inflammatory agent, a CETP inhibitor, a cardioprotective agent, a xenobiotic and an environmental contaminant. It is a member of pyrimidines, a sulfonamide, a dihydroxy monocarboxylic acid, a statin (synthetic) and a member of monofluorobenzenes. It derives from a hept-6-enoic acid. It is a conjugate acid of a rosuvastatin(1-). | | Preparation | Rosuvastatin synthesis: Reduction to the 5-hydroxymethyl derivative proceeds smoothly with diisobutylaluminum hydride (DIBALH) in toluene at -10 °C. The hydroxymethyl group is then converted to the bromo derivative, which upon reaction with triphenylphosphine affords the Wittig reagent. The latter is treated with tert-butyl 2-[(4R,6S)-6-formyl-2,2-dimethyl-1,3-dioxan-4-yl]acetate and provides the protected rosuvastatin ester. Removal of the dioxane protecting group by HCl, ester hydrolysis with NaOH and precipitation with CaCl2 gives rosuvastatin.
Kleemann A; Cardiovascular Drugs. Ullmann's Encyclopedia of Industrial Chemistry 7th ed. (1999-2017). NY, NY: John Wiley & Sons. Online Posting Date: January 15, 2008 | | Brand name | Crestor (AstraZeneca). | | General Description | Rosuvastatin, 7-[4-(4-fluorophenyl)-6-(1-methylethyl)-2-(methyl-methylsulfonyl-amino)-pyrimidin-5-yl]-3,5-dihydroxy-hept-6-enoic acid (Crestor), is oneof the more recently introduced statins in the United States.As with all statins, there is a concern of rhabdomyolysis andas such, the FDA has mandated that a warning about thisside effect, as well as a kidney toxicity warning, be added tothe product label (http://www.fda.gov/CDER/Drug/advisory/crestor_3_2005.htm). This should not come as a surprisebecause of the relationship in the chemical architectureto cerivastatin, which was withdrawn from the market as aresult of its adverse side effects. | | Clinical Use | HMG CoA reductase inhibitor:
Hyperlipidaemia | | Drug interactions | Potentially hazardous interactions with other drugs
Anti-arrhythmics: concentration possibly increased
by dronedarone - reduce dose of rosuvastatin.
Antibacterials: erythromycin reduces concentration
of rosuvastatin; increased risk of myopathy with
daptomycin and fusidic acid - avoid.
Anticoagulants: effect of coumarins and phenindione
enhanced.
Antifungals: concentration increased by itraconazole
- reduce dose of rosuvastatin.
Antivirals: increased risk of myopathy with
atazanavir, darunavir, dasabuvir, fosamprenavir,
indinavir, ledipasvir, lopinavir, paritaprevir,
ritonavir, saquinavir and tipranavir - reduce dose
of rosuvastatin, avoid with fosamprenavir, indinavir,
ledipasvir, ritonavir and saquinavir.
Ciclosporin: increased risk of myopathy - avoid.
Clopidogrel: concentration of rosuvastatin increased,
maximum rosuvastatin dose is 20 mg in normal renal
function.
Colchicine: possible increased risk of myopathy.
Cytotoxics: concentration increased by eltrombopag
- reduce dose of rosuvastatin.
Lipid-lowering agents: increased risk of myopathy
with ezetimibe, fibrates, gemfibrozil (avoid) and
nicotinic acid - reduce dose of rosuvastatin.
Teriflunomide: concentration increased by
teriflunomide - reduce dose of rosuvastatin. | | Metabolism | Rosuvastatin undergoes limited metabolism in the liver
mainly by the cytochrome P450 isoenzyme CYP2C9
(approximately 10%).
Approximately 90% of the rosuvastatin dose is excreted
unchanged in the faeces (consisting of absorbed and
non-absorbed active substance) and the remaining part is
excreted in urine. |
| | Rosuvastatin Preparation Products And Raw materials |
|