|
|
| | 1,3-Dimethylbarbituric acid Basic information |
| Product Name: | 1,3-Dimethylbarbituric acid | | Synonyms: | AKOS B029702;1,3-DIMETHYLBARBITURIC ACID;1,3-DIMETHYL-2,4,6(1H,3H,5H)-PYRIMIDINETRIONE;6(1h,3h,5h)-pyrimidinetrione,1,3-dimethyl-4;1,3-DiMethylbarbituric acid, 98% 100GR;1,3-DiMethylbarbituric acid, 98% 25GR;1,3-DiMethylbarbituric acid(for OseltaMivir phosphate);1,3- twoMethyl barbituric acid | | CAS: | 769-42-6 | | MF: | C6H8N2O3 | | MW: | 156.14 | | EINECS: | 212-211-7 | | Product Categories: | | | Mol File: | 769-42-6.mol | 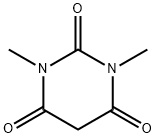 |
| | 1,3-Dimethylbarbituric acid Chemical Properties |
| Hazard Codes | Xn | | Risk Statements | 22-41 | | Safety Statements | 26-36/39 | | WGK Germany | 3 | | TSCA | Yes | | HS Code | 29339900 |
| | 1,3-Dimethylbarbituric acid Usage And Synthesis |
| Chemical Properties | Yellow or brownish powder | | Uses | 1,3-Dimethyl Barbituric Acid (Urapidil Impurity 4) is a derivative of Barbituric acid. All of the barbituric acid derivatives which have been reported to have pronounced hypnotic activity are disubstituted in the 5-position. | | Uses | 1,3-Dimethylbarbituric acid is used as a catalyst in the Knoevenagel condensation of a series of aromatic aldehydes. It is also used in the synthesis of 5-aryl-6-(alkyl- or aryl-amino)-1,3-dimethylfuro [2,3-d]pyrimidine derivatives and enantioselective synthesis of isochromene pyrimidinedione derivatives. | | General Description | 1,3-Dimethylbarbituric acid (1,3-Dimethyl-2,4,6(1H,3H,5H)-pyrimidinetrione) is an active methylene compound. It undergoes hollow Pd6 water-soluble cage, [{(tmen)Pd}6(timb)4](NO3)12 (tmen= N,N,N′,N′-tetramethylethylenediamine, timb=1,3,5-tris(1-imidazolyl)benzene)-catalyzed Knoevenagel condensation reaction with pyrene-1-carboxaldehyde. It undergoes self-sorted Pd7 molecular boat having an internal nanocavity (catalyst)-assisted Knoevenagel condensation reaction with various aromatic aldehydes. It has been synthesized by reacting 1,3-dimethylurea, malonic acid and acetic anhydride in acetic acid. It is widely used for the synthesis of various synthetic intermediates and heterocyclic compounds. | | Purification Methods | Crystallise the acid from water and sublime it in a vacuum. Also purify it by dissolving 10g in 100mL of boiling CCl4/CHCl3 (8:2) (1g charcoal), filtering and cooling to 25o. Dry it in vacuo [Kohn et al. Anal Chem 58 3184 1986]. [Beilstein 24 III/IV 1875.] |
| | 1,3-Dimethylbarbituric acid Preparation Products And Raw materials |
| Preparation Products | 6-[N-(2-Hydroxyethyl)amino]-1,3-dimethyl-2,4(1H,3H)-dione-->ISOCAFFEINE-->1,3-dimethyl-5-(thiophen-2-ylmethylene)pyrimidine-2,4,6(1H,3H,5H)-trione-->7-AMINO-1,3-DIMETHYL-2,4-DIOXO-1,2,3,4-TETRAHYDRO-5H-PYRANO[2,3-D]PYRIMIDINE-5,5,6-TRICARBONITRILE-->5-((1H-indol-3-yl)methylene)-1,3-dimethylpyrimidine-2,4,6(1H,3H,5H)-trione-->2-[(1,3-dimethyl-2,4,6-trioxo-1,3-diazinan-5-ylidene)methyl]phenyl acetate-->1,3-dimethyl-5-[(4-oxo-4H-chromen-3-yl)methylidene]-1,3-diazinane-2,4,6-trione-->1,3-DIMETHYL-5-([4-(METHYLSULFANYL)PHENYL]METHYLENE)-2,4,6(1H,3H,5H)-PYRIMIDINETRIONE-->6-chloro-1,3-dimethyl-5-nitro-pyrimidine-2,4-quinone-->1,3-DIMETHYL-5-[(DIMETHYLAMINO)METHYLENE]2,4,6-(1H,3H,5H)-TRIOXOPRYIMIDINE |
|