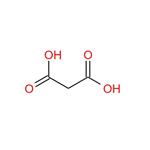
- Malonic acid
-

- $10.00/ kg
-
2024-04-24
- CAS:141-82-2
- Min. Order: 1kg
- Purity: 99.8%
- Supply Ability: 10000ton
- Malonic acid
-

- $0.00 / 25Kg/Drum
-
2024-04-24
- CAS:141-82-2
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 5000mt
- Malonic acid
-
- $0.00 / 1KG
-
2024-04-23
- CAS:141-82-2
- Min. Order: 1KG
- Purity: >99% HPLC
- Supply Ability: 1000KG
Related articles - What is Malonic acid?
- Malonic acid, also known as Propanedioic acid, Methane dicarbonic acid or Carboxyacetic acid, is an organic acid. Mainly used ....
- Feb 26,2020
|
| | Malonic acid Basic information |
| Product Name: | Malonic acid | | Synonyms: | Malonate, AR,99.5%;Malonic acid anhydrous, free-flowing, Redi-Dri(TM), ReagentPlus(R), 99%;Malonic acid ReagentPlus(R), 99%;Malonic acid sublimed grade, >=99.95% trace metals basis;Malonic acid Vetec(TM) reagent grade, 98%;Malonic acid, synthesis grade;Malonate standard fo;Propane-1,3-dioic acid | | CAS: | 141-82-2 | | MF: | C3H4O4 | | MW: | 104.06 | | EINECS: | 205-503-0 | | Product Categories: | Acids;Electronic Chemicals;Materials Science;Micro &;Micro/NanoElectronics;Nanoelectronics;New Products for Materials Research and Engineering;Intermediates;Aromatic Carboxylic Acids, Amides, Anilides, Anhydrides & Salts;alpha,omega-Alkanedicarboxylic Acids;alpha,omega-Bifunctional Alkanes;Monofunctional & alpha,omega-Bifunctional Alkanes;141-82-2 | | Mol File: | 141-82-2.mol |  |
| | Malonic acid Chemical Properties |
| Melting point | 132-135 °C (dec.) (lit.) | | Boiling point | 140℃(decomposition) | | density | 1.619 g/cm3 at 25 °C | | vapor pressure | 0-0.2Pa at 25℃ | | refractive index | 1.4780 | | Fp | 157°C | | storage temp. | Sealed in dry,Room Temperature | | solubility | 1 M NaOH: soluble100mg/mL, clear to slightly hazy, colorless to faintly yellow | | pka | 2.83(at 25℃) | | form | Liquid | | color | White | | PH | 3.17(1 mM solution);2.5(10 mM solution);1.94(100 mM solution) | | Water Solubility | 1400 g/L (20 ºC) | | Merck | 14,5710 | | BRN | 1751370 | | Stability: | Stable. Incompatible with oxidizing agents, reducing agents, bases. | | InChIKey | OFOBLEOULBTSOW-UHFFFAOYSA-N | | LogP | -0.81 | | CAS DataBase Reference | 141-82-2(CAS DataBase Reference) | | NIST Chemistry Reference | Malonic acid(141-82-2) | | EPA Substance Registry System | Propanedioic acid (141-82-2) |
| | Malonic acid Usage And Synthesis |
| Description | Malonic acid (MA), also known as propanedioic acid, is a dicarboxylic acid with structure CH2(COOH)2. It have three kinds of crystal forms, of which two are triclinic, and one is monoclinic. That crystallized from ethanol is white triclinic crystals.It decomposes to acetic acid and carbon dioxide at 140℃. It does not decompose at 1.067×103~1.333×103Pa vacuum, but directly sublimates. The ionised form of malonic acid, as well as its esters and salts, are known as malonates. For example, diethyl malonate is malonic acid's ethyl ester. The name originates from Latin malum, meaning apple. | | Chemical Properties | Malonic acid is a white crystalline solid that decomposes at approximately 135°C. It has high solubility in water and oxygenated solvents and exhibits greater acidity than acetic acid, which has a pK value of 4.75. The pKa values for the loss of its first and second protons are 2.83 and 5.69, respectively. It is slightly soluble in pyridine. It can decompose to formic acid and carbon dioxide in case of potassium permanganate. Since that malonic acid generates carbon dioxide and water after heated without pollution problems, it can be directly used as aluminum surface treatment agent. | | Uses | Malonic acid is used as an intermediate in the manufacture of barbiturates and other pharmaceuticals. It is a component used as a stabilizer in many high-end cosmetic and pharmaceutical products. Malonic acid is also used as building block in chemical synthesis, specifically to introduce the molecular group -CH2-COOH. It is used for the introduction of an acetic acid moiety under mild conditions by Knoevenagel condensation and subsequent decarboxylation. | | Application | Malonic acid is acts as a building block in organic synthesis. It is also useful as a precursor for polyesters and alkyd resins, which is used in coating applications, thereby protecting against UV light, corrosion and oxidation. It acts as a cross linker in the coating industry and surgical adhesive. It finds application in the production of specialty chemicals, flavors and fragrances, polymer cross linkers and pharmaceuticals. | | Definition | ChEBI: Malonic acid is an alpha,omega-dicarboxylic acid in which the two carboxy groups are separated by a single methylene group. It has a role as a human metabolite. It is a conjugate acid of a malonate(1-). | | Preparation | Malonic acid is usually produced from chloroacetic acid.
Reaction: The chloroacetic acid is added to the reaction kettle by adding sodium carbonate aqueous solution to generate sodium chloroacetate aqueous solution, and then 30% sodium cyanide solution is slowly added dropwise, and the reaction is carried out at a predetermined temperature to generate sodium cyanoacetate. After the cyanation reaction is completed, add sodium hydroxide for heating and hydrolysis to generate sodium malonate solution, concentrate, then dropwise add sulfuric acid for acidification to generate malonic acid, filter and dry to obtain the product.
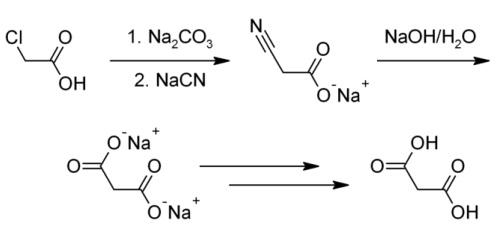
This method often does not produce a pure enough product or the pure product has an extremely low yield. Industrially, malonic acid is also produced by hydrolyzing dimethyl malonate or diethyl malonate. This manufacturing method is able to bring about a higher yield and purity, but the organic synthesis of malonic acid through these processes is extremely costly and environmentally hazardous. | | Reactions | In a well - known reaction, malonic acid condenses with urea to form barbituric acid. Malonic acid is also frequently used as an enolate in Knoevenagel condensations or condensed with acetone to form Meldrum's acid. The esters of malonic acid are also used as a - CH2COOH synthon in the malonic ester synthesis. | | Biological Functions | Malonic acid is the classic example of a competitive inhibitor of the enzyme succinate dehydrogenase (complex II), in the respiratory electron transport chain.It binds to the active site of the enzyme without reacting, competing with the usual substrate succinate but lacking the ?CH2CH2? group required for dehydrogenation. This observation was used to deduce the structure of the active site in succinate dehydrogenase. | | General Description | White crystals or crystalline powder. Sublimes in vacuum. | | Air & Water Reactions | Water soluble. | | Reactivity Profile | Malonic acid is a carboxylic acid. Carboxylic acids donate hydrogen ions if a base is present to accept them. They react in this way with all bases, both organic (for example, the amines) and inorganic. Their reactions with bases, called "neutralizations", are accompanied by the evolution of substantial amounts of heat. Neutralization between an acid and a base produces water plus a salt. Carboxylic acids with six or fewer carbon atoms are freely or moderately soluble in water; those with more than six carbons are slightly soluble in water. Soluble carboxylic acid dissociate to an extent in water to yield hydrogen ions. The pH of solutions of carboxylic acids is therefore less than 7.0. Many insoluble carboxylic acids react rapidly with aqueous solutions containing a chemical base and dissolve as the neutralization generates a soluble salt. Carboxylic acids in aqueous solution and liquid or molten carboxylic acids can react with active metals to form gaseous hydrogen and a metal salt. Such reactions occur in principle for solid carboxylic acids as well, but are slow if the solid acid remains dry. Even "insoluble" carboxylic acids may absorb enough water from the air and dissolve sufficiently in Malonic acid to corrode or dissolve iron, steel, and aluminum parts and containers. Carboxylic acids, like other acids, react with cyanide salts to generate gaseous hydrogen cyanide. The reaction is slower for dry, solid carboxylic acids. Insoluble carboxylic acids react with solutions of cyanides to cause the release of gaseous hydrogen cyanide. Flammable and/or toxic gases and heat are generated by the reaction of carboxylic acids with diazo compounds, dithiocarbamates, isocyanates, mercaptans, nitrides, and sulfides. Carboxylic acids, especially in aqueous solution, also react with sulfites, nitrites, thiosulfates (to give H2S and SO3), dithionites (SO2), to generate flammable and/or toxic gases and heat. Their reaction with carbonates and bicarbonates generates a harmless gas (carbon dioxide) but still heat. Like other organic compounds, carboxylic acids can be oxidized by strong oxidizing agents and reduced by strong reducing agents. These reactions generate heat. A wide variety of products is possible. Like other acids, carboxylic acids may initiate polymerization reactions; like other acids, they often catalyze (increase the rate of) chemical reactions Malonic acid is incompatible with strong oxidizers. Malonic acid is also incompatible with bases and reducing agents. | | Hazard | Strong irritant. | | Fire Hazard | Flash point data for Malonic acid are not available; however, Malonic acid is probably combustible. | | Flammability and Explosibility | Not classified | | Biotechnological Applications | The calcium salt of malonic acid occurs in high concentrations in beetroot. It exists in its normal state as white crystals. Malonic acid is the classic example of a competitive inhibitor: It acts against succinate dehydrogenase (complex II) in the respiratory electron transport chain. | | Purification Methods | Crystallise malonic acid from *benzene/diethyl ether (1:1) containing 5% of pet ether (b 60-80o), wash with diethyl ether, then recrystallise it from H2O or acetone. Dry it under vacuum over conc H2SO4. [Beilstein 2 IV 1874.] |
| | Malonic acid Preparation Products And Raw materials |
|