1132935-63-7
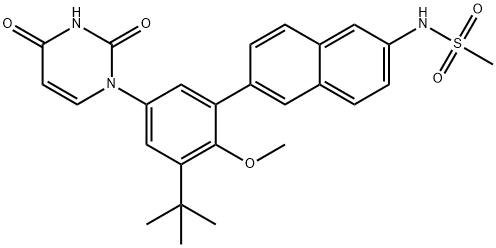 1132935-63-7 结构式
1132935-63-7 结构式
基本信息
达沙布韦
达塞布韦
达沙布韦 DASABUVIR
N-(6-(3-(叔丁基)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-2-甲氧基苯基)萘-2-基)甲磺酰胺
ABT-333
Dasabuvir
ABT333
ABT 333
ABT-333/Dasabuvir
Dasabuvir (ABT-333)
N-{6-[5-(2,4-Dioxo-3,4-dihydro-1(2H)-pyrimidinyl)-2-methoxy-3-(2-methyl-2-propanyl)phenyl]-2-naphthyl}methanesulfonamide
Methanesulfonamide, N-[6-[5-(3,4-dihydro-2,4-dioxo-1(2H)-pyrimidinyl)-3-(1,1-dimethylethyl)-2-methoxyphenyl]-2-naphthalenyl]-
N-[6-[3-tert-Butyl-5-(2,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)-2-methoxyphenyl]naphthalen-2-yl]methanesulfonamide Dasabuvir (ABT-333)
物理化学性质
制备方法
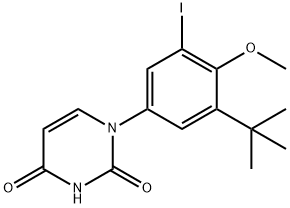
1132940-53-4
![N-[6-(4,4,5,5-四甲基-1,3,2-二氧硼杂环戊烷-2-基)萘-2-基]甲烷磺酰胺](/CAS/GIF/1132940-88-5.gif)
1132940-88-5

1132935-63-7
S3:在氮气保护下,向反应容器中加入1,4-二恶烷(141.4 L)、钯催化剂Pd(PPh3)4(3.9 kg)和水(70.68 L),随后加入N-(6-(4,4,5,5-四甲基-1,3,2-二氧杂硼杂环戊烷-2-基)萘-2-基)甲磺酰胺(23.56 kg)和1-(3-(叔丁基)-5-碘-4-甲氧基苯基)嘧啶-2,4(1H,3H)-二酮(27.13 kg),搅拌混合。接着加入碳酸钠(8.63 kg),将反应溶液保持4-6小时,期间通过TLC监测反应进度直至反应完成。反应完成后,将反应溶液冷却至40℃,然后用乙酸乙酯(235.6 L)稀释。合并的有机相用水(47.12 L)洗涤,随后在50℃下减压蒸馏浓缩有机相。粗产物中加入乙酸乙酯(94.24 L),在50-60℃下搅拌30分钟,最终得到粗产物(29.45 kg)。粗产物经过抽滤后,通过硅胶柱色谱法进行纯化。该取代反应为单一步骤,产率为76.5%。
参考文献:
[1] Patent: CN107266373, 2017, A. Location in patent: Paragraph 0057; 0058; 0059
[2] Patent: WO2009/39134, 2009, A1. Location in patent: Page/Page column 109
[3] Patent: WO2009/39127, 2009, A1. Location in patent: Page/Page column 168-169
[4] Patent: WO2009/39134, 2009, A1. Location in patent: Page/Page column 102-103; 109
