1187932-25-7
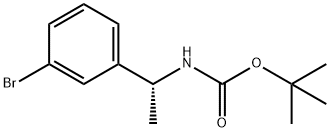 1187932-25-7 结构式
1187932-25-7 结构式
基本信息
(R)-1-(3-溴苯基)乙氨基甲酸叔丁酯
(R)-叔丁基1-(3-溴苯基)乙基氨基甲酸酯
(R)-[1-(3-溴苯基)乙基]氨基甲酸叔丁酯
N-[(1R)-1-(3-溴苯基)乙基]氨基甲酸叔丁酯
中文名:?7-溴-2-甲基喹唑啉英文名:?7-BROMO-2-METHYLQUINAZOLINECAS号:552331-87-0
tert-butyl (R)-1-(3-bromophenyl)ethylcarbamate
(R)-[1-(3-Bromo-phenyl)-ethyl]-carbamic acid tert-butyl ester
N-[(1R)-1-(3-Bromophenyl)ethyl]carbamic acid 1,1-dimethylethyl ester
Carbamic acid, N-[(1R)-1-(3-bromophenyl)ethyl]-, 1,1-dimethylethyl ester
制备方法
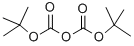
24424-99-5
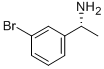
176707-77-0

1187932-25-7
以二碳酸二叔丁酯和(R)-1-(3-溴苯基)乙胺为原料合成(R)-1-(3-溴苯基)乙氨基甲酸叔丁酯的一般步骤:在实施例16中,化合物16a的合成如下:将(R)-1-(3-溴苯基)乙胺(1.023g,5.112mmol)溶于二氯甲烷(20mL)中,随后加入三乙胺(720μL,5.112mmol)和二碳酸二叔丁酯(1.784g,8.179mmol)。反应混合物在室温下搅拌过夜。反应完成后,通过减压蒸馏除去挥发性溶剂,残余物通过硅胶柱色谱法纯化,使用50g Isolute柱,以己烷/Et2O的连续梯度(1:0至4:1)洗脱,得到标题化合物(R)-1-(3-溴苯基)乙氨基甲酸叔丁酯(1.552g,100%),为白色固体。1H NMR(300MHz,CDCl3)δ 1.43(br s,12H),4.77(br s,2H),7.16-7.26(m,2H),7.39(dt,J = 2.0,7.1Hz,1H),7.46(s,1H)。
参考文献:
[1] Patent: WO2012/78915, 2012, A1. Location in patent: Page/Page column 90
[2] Patent: WO2013/48942, 2013, A1. Location in patent: Paragraph 00198
[3] Patent: WO2013/151923, 2013, A1. Location in patent: Paragraph 00186
[4] Patent: WO2013/185093, 2013, A1. Location in patent: Page/Page column 61; 62
[5] Patent: WO2013/185103, 2013, A1. Location in patent: Page/Page column 63