1228014-35-4
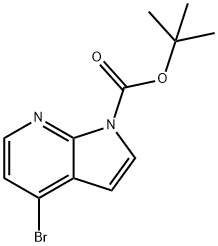 1228014-35-4 结构式
1228014-35-4 结构式
基本信息
4-Bromo-pyrrolo[2,3-b]pyridine-1-carboxylic acid tert-butyl ester
1H-Pyrrolo[2,3-b]pyridine-1-carboxylic acid, 4-bromo-, 1,1-dimethylethyl ester
物理化学性质
制备方法
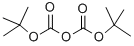
24424-99-5
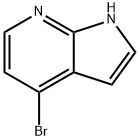
348640-06-2
![4-溴-1H-吡咯并[2,3-B]吡啶-1-羧酸叔丁酯](/CAS/20150408/GIF/1228014-35-4.gif)
1228014-35-4
以4-溴-1H-吡咯并[2,3-b]吡啶(XCI)(5g,25.4mmol,1当量)为原料,将其溶解于二氯甲烷(DCM)(100mL)中,加入4-二甲氨基吡啶(DMAP)(311mg,2.55mmol,0.1当量)和三乙胺(TEA)(5.3mL)。在0℃下,向反应混合物中缓慢加入二碳酸二叔丁酯(Boc2O)(7.2mL,31mmol,1.2当量)。将反应体系逐渐升温至室温并持续搅拌2小时。反应完成后,加入水(100mL)进行淬灭,并用二氯甲烷(DCM)萃取两次。合并有机相,减压浓缩除去溶剂,得到4-溴-1H-吡咯并[2,3-b]吡啶-1-羧酸叔丁酯(XCII)为无色油状物(6.95g,23.4mmol,收率92.1%)。产物经1H NMR(CDCl3, 400MHz)确认:δ 1.60(s, 9H), 6.50(d, J = 4Hz, 1H), 7.31(d, J = 5.2Hz, 1H), 7.63(d, J = 4Hz, 1H), 8.23(d, J = 5.2Hz, 1H);电喷雾质谱(ESIMS)分析显示分子离子峰m/z 297.1(M + H),符合预期分子式C12H13BrN2O2。
参考文献:
[1] Patent: WO2017/24021, 2017, A1. Location in patent: Paragraph 0694; 0695
[2] Patent: WO2017/24004, 2017, A1. Location in patent: Paragraph 0676
[3] Patent: WO2017/23980, 2017, A1. Location in patent: Paragraph 0676
[4] Patent: WO2017/23996, 2017, A1. Location in patent: Paragraph 0676
[5] Patent: WO2012/101654, 2012, A2. Location in patent: Page/Page column 59
