123320-44-5
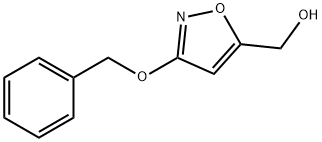 123320-44-5 结构式
123320-44-5 结构式
基本信息
(3-(苄氧基)异噁唑-5-基)甲醇
(3-(苄氧基)异恶唑-5-基)甲醇
3-(苄氧基)-1,2-F唑-5-基]甲醇
EOS-61969
[3-(benzyloxy)-1,2-oxazol-5-yl]Methano
5-Isoxazolemethanol, 3-(phenylmethoxy)-
[3-(benzyloxy)-1,2-oxazol-5-yl]Methanol
(3-phenylmethoxy-1,2-oxazol-5-yl)methanol
物理化学性质
制备方法
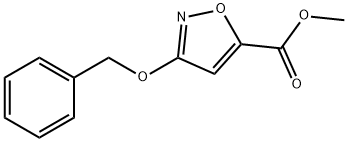
205115-22-6

123320-44-5
以3-(苄氧基)异恶唑-5-甲酸甲酯为原料合成(3-(苄氧基)异恶唑-5-基)甲醇的一般步骤:向3-(苄氧基)异恶唑-5-甲酸甲酯(2.33 g,9.99 mmol,1.00当量)的甲醇(50 mL)溶液中分批加入硼氢化钠(756 mg,19.98 mmol,2.00当量)。将反应混合物在15℃下搅拌3小时。通过薄层色谱(TLC,展开剂比例为石油醚:乙酸乙酯=5:1)监测反应进程,确认反应完成。将反应混合物缓慢倒入0.2 M盐酸溶液(200 mL)中,并用乙酸乙酯(150 mL×2)进行萃取。合并有机相,用饱和食盐水(200 mL×2)洗涤,无水硫酸钠干燥,过滤后减压浓缩,得到(3-(苄氧基)异恶唑-5-基)甲醇(1.85 g,9.02 mmol,收率90%),为无色油状物。LC-MS(ESI)m/z:206.1 [M+H]+。
参考文献:
[1] Patent: WO2018/140809, 2018, A1. Location in patent: Paragraph 00443; 00551; 00554; 00555
[2] European Journal of Organic Chemistry, 1998, # 3, p. 473 - 479
[3] Patent: WO2012/87833, 2012, A1. Location in patent: Page/Page column 133
[4] Journal of Medicinal Chemistry, 2015, vol. 58, # 23, p. 9133 - 9153
[5] Patent: WO2008/127361, 2008, A2. Location in patent: Page/Page column 89