1425045-01-7
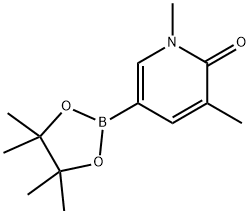 1425045-01-7 结构式
1425045-01-7 结构式
基本信息
1,5-二甲基-6-氧代-1,6-二氢吡啶-3-硼酸频哪醇酯
1,3-二甲基-5-(4,4,5,5-四甲基-1,3,2-二氧杂硼烷-2-基)吡啶-2(1H)-酮
1,3-二甲基-5-(4,4,5,5-四甲基-1,3,2-二氧杂硼杂环戊烷-2-基)-2(1H)-吡啶酮
1,3-DIMETHYL-5-(4,4,5,5-TETRAMETHYL-1,3,2-DI氧BENZALDEHYDE-2-YL)-1,2-DIHYDROPYRIDINE-2-ONE
1,3-Dimethyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-2(1H)-pyridinone
1,3-diMethyl-5-(4,4,5,5-tetraMethyl-1,3,2-dioxaborolan-2-yl)pyridin-2(1H)-one
1,3-dimethyl-5-(tetramethyl-1,3,2-dioxaborolan-2-yl)-1,2-dihydropyridin-2-one
1,3-Dimethyl-5-(4,4,5,5-tetramethyl-[1,3,2]dioxaborolan-2-yl)-1H-pyridin-2-one
2(1H)-Pyridinone, 1,3-dimethyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-
制备方法
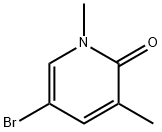
51417-13-1
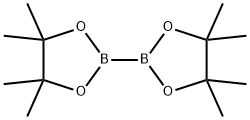
73183-34-3

1425045-01-7
以5-溴-1,3-二甲基吡啶-2(1H)-酮和联硼酸频那醇酯为原料合成1,3-二甲基吡啶-2(1H)-酮-5-频哪醇硼酸酯的一般步骤如下:首先,将氮气通入二恶烷中进行脱气处理。随后,将脱气后的二恶烷溶液(2.0 mL)加入到含有5-溴-1,3-二甲基吡啶-2(1H)-酮(75 mg,0.37 mmol)、双(频哪醇)二硼(113 mg,0.445 mmol)和KOAc(109 mg,1.11 mmol)的混合物中。接着,加入催化剂PdCl2(dppf)·CH2Cl2(30 mg,0.037 mmol)。将反应混合物在85℃下加热反应16小时。反应完成后,用EtOAc稀释反应混合物,并通过Celite垫过滤。最后,将滤液在真空下浓缩,得到粗产物1,3-二甲基吡啶-2(1H)-酮-5-频哪醇硼酸酯(169 mg,0.678 mmol,定量收率),为棕色油状物。
参考文献:
[1] Patent: WO2016/196644, 2016, A1. Location in patent: Paragraph 709; 710
[2] Journal of Medicinal Chemistry, 2016, vol. 59, # 10, p. 4462 - 4475
[3] Patent: WO2014/78323, 2014, A1. Location in patent: Paragraph 00584
[4] Patent: US2018/44335, 2018, A1. Location in patent: Paragraph 0459-0460
[5] Patent: WO2018/102452, 2018, A2. Location in patent: Paragraph 693; 694