147126-62-3
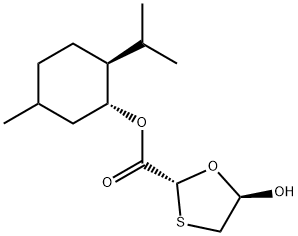 147126-62-3 结构式
147126-62-3 结构式
基本信息
拉米夫定杂质25
恩曲他滨中间体HME
拉米夫定中间体HME
(1R,2S,5R)-5-甲基-2-异丙基环己酯
(2R,5R)-5-羟基-(1,3)-氧硫杂环戊烷-2-羧酸孟酯(HME)
5R-羟基-1,3-氧硫杂环戊烷-2R-羧酸(1'R,2'S,5'R)-薄荷酯
(2R,5R)-5-羟基-1,3-氧硫杂环-2-羧酸(R,2S,5R)-5-甲基-2-异丙基环己酯
(2R,5R)-5-羟基-1,3-氧硫杂环-2-羧酸(1R,2S,5R)-5-甲基-2-异丙基环己酯
(2R,5R)-5-羟基-1,3-氧硫杂环-2-羧酸 (1R,2S,5R)-5-甲基-2-异丙基环己酯
Lamivudine Impurity 17
(2R,5R)-(1R,2S,5R)-2-isopropyl
(1R,2S,5R)-Methyl-5R-hydroxy-[1,3]-oxathiolane-2R-carboxylate
(1R,2S,5R)-Menthol-5R-hydroxy-[1,3]-oxathiolane-2R-carboxylate
(2R,5R)-5-HYDROXY-1,3-OXATHIOLANE-2-CARBOXYLIC ACID MENTHYL ESTE
(2R, 5R)-5-HYDROXY-1,3-OXATHIOLANE-2-CARBOXYLIC ACID METHYL ESTER
(2R, 5R)-5-HYDROXY-1,3-OXATHIOLANE-2-CARBOXYLIC ACID L METHYL ESTER
(2S,5R)-5-Hydroxy-[1,3]-oxathiolane-2-carboxylic acid menthyl ester
(2S,5R)-5-Hydroxy-[1,3]-oxathiolane-2-carboxylic acid menthyl ester (HME)
物理化学性质
制备方法
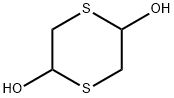
40018-26-6
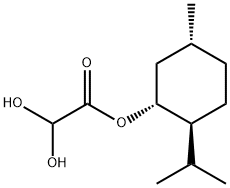
111969-64-3

147126-62-3
以L-薄荷基乙二酸酯一水合物(1.0 g,4.34 mmol)和1,4-二噻烷-2,5-二醇(396 mg,2.60 mmol)为原料,在丙酮(10 mL)中溶解,加入催化量的乙酸(0.5 mL)。将混合溶液以0.1 mL/min的总流速(其中L-薄荷基乙二酸酯溶液流速为0.05 mL/min)泵入流动反应器,反应条件设置为125°C和10 bar背压。反应完成后,通过薄层色谱(TLC)和气相色谱(GC)监测反应进程。随后,蒸发溶剂,将反应混合物冷却至0°C-5°C,缓慢滴加1%三乙胺的庚烷或己烷溶液,并在0°C下搅拌2-3小时以促进沉淀形成。通过过滤收集固体产物,用正己烷洗涤,得到(2R,5R)-((1R,2S,5R)-2-异丙基-5-甲基环己基)5-羟基-1,3-氧硫杂环戊烷-2-羧酸盐(1.1 g,产率88%),为白色固体。产物结构通过以下方法确认:1H-NMR(400 MHz, CDCl3): δ 5.96 (s, 1H), 5.57 (d, 1H), 4.74 (s, 1H), 3.32-3.29 (m, 1H), 3.17-3.08 (dd, 1H), 2.02 (d, 2H), 1.70 (d, 1H), 1.51-1.42 (m, 2H), 1.09-1.00 (m, 2H), 0.91 (d, 6H), 0.78 (d, 3H); 13C-NMR(100 MHz, CDCl3): δ 16.27, 20.69, 23.30, 26.16, 31.42, 34.11, 38.46, 40.35, 46.86, 46.07, 80.20, 101.22, 103.20, 172.18; FT-IR(Neat): 3456, 2956, 2864, 1731, 1457, 1387, 1288, 1196, 1041, 986 cm-1。反应条件:流速0.1 mL/min,停留时间20 min,反应温度50°C至125°C,结晶条件为1%三乙胺的庚烷/己烷溶液。
参考文献:
[1] Patent: WO2017/216709, 2017, A2. Location in patent: Page/Page column 12; 13; 14; 15; 17; 18
[2] Tetrahedron Letters, 2005, vol. 46, # 49, p. 8535 - 8538
[3] Patent: WO2013/21290, 2013, A1. Location in patent: Page/Page column 17; 18
