153747-97-8
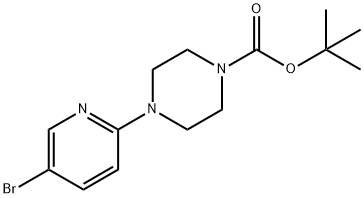 153747-97-8 结构式
153747-97-8 结构式
基本信息
帕布昔利布杂质6
帕布昔利布杂质54
哌泊塞克雷布杂质C
4-BOC-1-(5-溴-2-吡啶基)哌嗪
2-(N-BOC-哌嗪-1-基)-5-溴吡啶
4-(5-溴吡啶-2-基)哌嗪-1-甲酸叔丁酯
4-叔丁氧羰基-1-(5-溴-2-吡啶基)哌嗪
4-(5-溴-吡啶-2-基)-哌嗪-1-羧酸叔丁酯
4-(5-溴-吡啶-2-基)-哌嗪-1-羧酸叔丁基酯
Palbociclib impurity C
2-(4-BOC-piperazino)-5-bromopyridine
1-Boc-4-(5-bromo-2-pyridinyl)-piperazine
4-Boc-1-(5-bromo-2-pyridyl)piperazine 97%
5-Bromo-2-[4-(N-Boc)piperazin-1-yl]pyridine
Palbociclib Impurity C : Trifluoroacetic acid salt
t-Butyl 4-(5-bromopyridin-2-yl)piperazine-1-carboxylate
ert-butyl4-(5-bromopyridin-2-yl)piperazine-1-carboxylate
4-(5-Bromo-pyridin-2-yl)-piperazine-1-carboxylic acid tert-butyl ester
物理化学性质
安全数据
toxic hazardous materials or hazardous materials causing chronic effects
Eye Irrit. 2
Skin Irrit. 2
STOT SE 3
制备方法
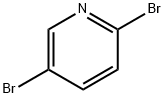
624-28-2
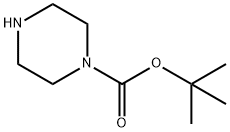
57260-71-6

153747-97-8
以2,5-二溴吡啶和N-BOC-哌嗪为原料合成4-Boc-1-(5-溴-2-吡啶基)哌嗪的一般步骤:在氩气保护下,向装有搅拌子的密封管中加入2,5-二溴吡啶(2g,10.7mmol)、叔丁醇钠(1.6g,16.6mmol)、xantphos(400mg,0.7mmol)及甲苯(100mL),氩气吹扫5分钟。随后,向反应体系中加入叔丁基哌嗪-1-羧酸酯(3.4g,14.30mmol)和Pd2(dba)3(200mg,0.21mmol),于80℃下加热反应4小时(TLC监测显示原料完全消耗)。反应完成后,将反应混合物用EtOAc(200mL)稀释,加入水(100mL),通过硅藻土床过滤,并用EtOAc(2×30mL)洗涤滤床。合并有机相,用盐水(50mL)洗涤,无水Na2SO4干燥,减压浓缩得粗产物。粗产物经硅胶柱色谱(100-200目,50g,10-20% EtOAc-己烷梯度洗脱)纯化,得到4-(5-溴-2-吡啶基)哌嗪-1-羧酸叔丁酯(3g,82%),为黄色固体。LCMS(ESI+):m/z:342.57 [M+H]+。
参考文献:
[1] Organic Letters, 2003, vol. 5, # 24, p. 4611 - 4614
[2] Patent: WO2018/125961, 2018, A1. Location in patent: Page/Page column 123
[3] Patent: WO2007/20888, 2007, A1. Location in patent: Page/Page column 45-46
[4] Patent: US2006/293310, 2006, A1. Location in patent: Page/Page column 5
| 报价日期 | 产品编号 | 产品名称 | CAS号 | 包装 | 价格 |
| 2026/03/03 | H55311 | 4-Boc-1-(5-溴-2-吡啶基)哌嗪, 97% 4-Boc-1-(5-bromo-2-pyridyl)piperazine, 97% | 153747-97-8 | 1g | 266元 |
| 2025/05/22 | H55311 | 4-Boc-1-(5-溴-2-吡啶基)哌嗪, 97% 4-Boc-1-(5-bromo-2-pyridyl)piperazine, 97% | 153747-97-8 | 5g | 1512元 |
| 2025/05/22 | H55311 | 4-Boc-1-(5-溴-2-吡啶基)哌嗪, 97% 4-Boc-1-(5-bromo-2-pyridyl)piperazine, 97% | 153747-97-8 | 25g | 7560元 |
