218451-34-4
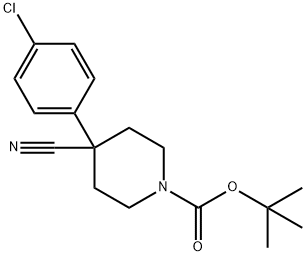 218451-34-4 结构式
218451-34-4 结构式
基本信息
4-(4-氯苯基)-4-氰基哌啶-1-羧酸叔丁酯
1-Boc-4-(4-chlorophenyl)piperidine-4-carbonitrile
TERT-BUTYL 4-(4-CHLOROPHENYL)-4-CYANOPIPERIDINE-1-CARBOXYLATE
1-Piperidinecarboxylic acid, 4-(4-chlorophenyl)-4-cyano-, 1,1-dimethylethyl ester
制备方法
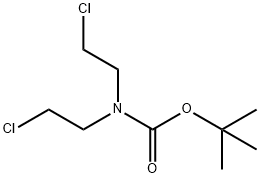
118753-70-1
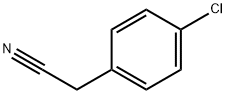
140-53-4

218451-34-4
步骤1. 在氮气保护下,将2-(4-氯苯基)乙腈(20.1g, 132.59mmol, 1.00当量)和N,N-双(2-氯乙基)氨基甲酸叔丁酯(35.4g, 146.19mmol, 1.10当量)溶于无水DMF(200mL)中,冷却至0℃。在2小时内分批加入氢化钠(27g, 60%矿物油分散, 666.73mmol, 3.00当量)。反应混合物在60℃下搅拌1.5小时,随后室温搅拌过夜。反应完成后,小心加入250mL饱和NaHCO3水溶液淬灭反应。用二氯甲烷(3×200mL)萃取反应混合物,合并有机相,用盐水(2×300mL)洗涤,无水硫酸钠干燥,减压浓缩。粗产物通过硅胶柱层析纯化,洗脱剂为二氯甲烷/石油醚(1:1),得到4-(4-氯苯基)-4-氰基哌啶-1-羧酸叔丁酯(30g, 71%),为黄色固体。TLC检测:Rf = 0.15(展开剂:乙酸乙酯/石油醚=1/5)。
参考文献:
[1] Patent: WO2015/32286, 2015, A1. Location in patent: Page/Page column 310; 311
[2] Patent: CN107428750, 2017, A. Location in patent: Paragraph 0548; 0551; 0552; 0553; 0554
[3] Journal of Medicinal Chemistry, 2008, vol. 51, # 7, p. 2147 - 2157
[4] Patent: WO2006/46024, 2006, A1. Location in patent: Page/Page column 122; 146
[5] Patent: WO2007/125321, 2007, A2. Location in patent: Page/Page column 120; 141-142