224824-22-0
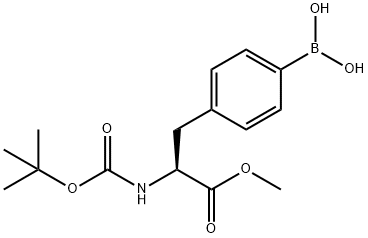 224824-22-0 结构式
224824-22-0 结构式
基本信息
N-叔丁氧羰基-4-(二羟基硼基)-L-苯丙氨酸甲酯
(S)-4-[2-(BOC-氨基)-3-甲氧基-3-氧代丙基]苯硼酸
(S)-(4-(2-((叔丁氧羰基)氨基)-3-甲氧基-3-酮)丙基)苯硼酸
(S)-4-[2-(Boc-amino)-3-methoxy-3-oxopropyl]phenylboronic Acid
(S)-4-(2-(tert-butoxycarbonylamino)-3-methoxy-3-oxopropyl)ph...
4-Borono-N-[(1,1-dimethylethoxy)carbonyl]-L-phenylalanine methyl ester
N-(tert-Butoxycarbonyl)-4-(dihydroxyboryl)-L-phenylalanine methyl ester
L-Phenylalanine, 4-borono-N-[(1,1-dimethylethoxy)carbonyl]-, 1-methyl ester
(S)-(4-(2-((tert-Butoxycarbonyl)amno)-3-methoxy-3-oxopropyl)phenyl)boronic acid
(S)-(4-(2-((tert-Butoxycarbonyl)aMino)-3-Methoxy-3-oxopropyl)phenyl)boronic acid
[4-[(2S)-3-methoxy-2-[(2-methylpropan-2-yl)oxycarbonylamino]-3-oxopropyl]phenyl]boronic acid
物理化学性质
制备方法
![(2S)-2-[(叔丁氧羰基)氨基]-3-[4-(4,4,5,5-四甲基-1,3,2-二氧杂环戊硼烷-2-基)苯基]丙酸甲酯](/CAS2/GIF/220587-29-1.gif)
220587-29-1

224824-22-0
以(S)-2-((叔丁氧基羰基)氨基)-3-(4-(4,4,5,5-四甲基-1,3,2-二氧硼杂环戊烷-2-基)苯基)丙酸甲酯(67.0 g,165 mmol)为原料,将其溶解于丙酮(700 mL)中。向该溶液中依次加入高碘酸钠(71.0 g,330 mmol)、乙酸铵(25.0 g,330 mmol)和水(300 mL),室温下搅拌反应55小时。反应完成后,过滤反应混合物,滤液经减压浓缩后,加入乙酸乙酯进行萃取。有机相依次用水和饱和氯化钠水溶液洗涤,无水硫酸钠干燥。减压除去溶剂,所得固体用石油醚/乙酸乙酯(1:10,v/v)混合溶剂洗涤并干燥,得到目标产物(S)-(4-(2-((叔丁氧羰基)氨基)-3-甲氧基-3-氧代丙基)苯硼酸(29 g,收率55%)。1H NMR (CDCl3, 400 MHz) δ: 7.71-7.56 (m, 2H), 7.25-7.16 (m, 2H), 4.39-4.37 (m, 1H), 3.71 (s, 3H), 3.14-3.10 (m, 1H), 2.96-2.90 (m, 1H), 1.40 (s, 9H). MS (ESI) m/z 224 (M + H - Boc)+.
参考文献:
[1] Angewandte Chemie - International Edition, 2014, vol. 53, # 6, p. 1669 - 1673
[2] Angew. Chem., 2014, vol. 126, # 6, p. 1695 - 1699,5
[3] Patent: US2015/51395, 2015, A1. Location in patent: Paragraph 0134-0135
[4] Patent: US2016/244451, 2016, A1. Location in patent: Paragraph 0144; 0145; 0146
[5] Patent: JP2016/37467, 2016, A. Location in patent: Paragraph 0060