3056-18-6
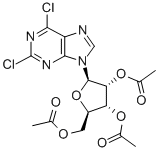 3056-18-6 结构式
3056-18-6 结构式
基本信息
2,3,5-三乙酰-2,6-二氯嘌呤核苷
2,3,5-4三乙酰基-2,6二氯嘌呤核苷
2,6-二氯嘌呤-2',3',5'-三乙酰
2,6-二氯-2',3',5'-三乙酰基嘌呤核糖苷
9- '2,3,5-三-O-乙酰基-Β-D-呋喃核糖基 -2,6-二氯
2,6-二氯-9-(2',3',5'-三-O-乙酰-Β-D-呋喃核糖基)嘌呤
2,6-二氯-9-(2',3',5'-三-O-乙酰基-ΒD-呋喃核糖基)嘌呤
2,6-二氯-9-(2',3',5'-三-O-乙酰基-BD-呋喃核糖基)嘌呤
9-[2,3,5-三-O-乙酰-BETA-D-呋喃核糖基]-2,6-二氯嘌呤
NSC 76763
2',3',5'-Tri-O-Acetyl-2,6-Dichloropurine
2,6-Dichloro-2',3',5'-triacetyl-purine riboside
9-[2,3,5-tri-O-acetyl--D-ribofuranosyl]-2,6-dichloropurine
9-[2,3,5-TRI-O-ACETYL-SS-D-RIBOFURANOSYL]-2,6-DICHLOROPURINE
9-[2,3,5-TRI-O-ACETYL-BETA-D-RIBOFURANOSYL]-2,6-DICHLOROPURINE
2,6-Dichloro-9-(2’,3’,5’-tri-O-acetyl-β-D-ribofuranosyl)purine
2,6-DICHLORO-9-(2,3,5-TRI-O-ACETYL-B-D-RIBOFURANOSYL)-9H-PURINE
9H-Purine,2,6-dichloro-9-(2,3,5-tri-O-acetyl-b-D-ribofuranosyl)-
物理化学性质
制备方法
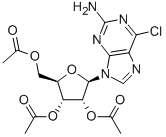
16321-99-6

3056-18-6
以(2R,3R,4R,5R)-2-(乙酰氧基甲基)-5-(2-氨基-6-氯-9H-嘌呤-9-基)四氢呋喃-3,4-二基二乙酸酯为原料,合成(2R,3R,4R,5R)-2-(乙酰氧基甲基)-5-(2,6-二氯-9H-嘌呤-9-基)四氢呋喃-3,4-二乙酸二酯的一般步骤如下:将亚硝酸异戊酯(165 g,1.41 mol)溶解于二氯甲烷(500 mL)中,并在0至5°C下搅拌。随后加入三甲基氯硅烷(76 g,0.70 mol),接着加入化合物IV(100 g,0.234 mol)的二氯甲烷(500 mL)溶液。将混合液滴加至上述反应母液中,滴加完毕后,继续在相同温度下搅拌反应1小时。反应完成后,加入饱和亚硫酸钠溶液进行淬灭,分液。分离得到的有机相依次用碳酸氢钠水溶液和饱和盐水洗涤,然后用无水硫酸钠干燥,过滤。将滤液减压浓缩至无馏分蒸出。残余物通过加入二氯甲烷(100 mL)和正庚烷(350 mL)进行重结晶,得到黄色固体化合物V,即2,6-二氯-9-(2',3',5'-三-O-乙酰基-β-D-呋喃核糖基)嘌呤(86 g,产率82%)。
参考文献:
[1] Journal of Organic Chemistry, 2003, vol. 68, # 2, p. 666 - 669
[2] Canadian Journal of Chemistry, 1981, vol. 59, # 17, p. 2608 - 2611
[3] Patent: CN106397516, 2017, A. Location in patent: Paragraph 0029; 0030; 0080; 0081; 0082; 0083
[4] Journal of Medicinal Chemistry, 1992, vol. 35, # 24, p. 4557 - 4561
[5] Journal of Organic Chemistry, 2002, vol. 67, # 19, p. 6788 - 6796
常见问题列表
| 报价日期 | 产品编号 | 产品名称 | CAS号 | 包装 | 价格 |
| 2025/12/22 | HY-131847 | 2,3,5-三乙酰-2,6-二氯嘌呤核苷 2,6-Dichloro-2',3',5'-triacetyl-purine riboside | 3056-18-6 | 100 mg | 100元 |
| 2025/12/22 | HY-131847 | 2,3,5-三乙酰-2,6-二氯嘌呤核苷 2,6-Dichloro-2',3',5'-triacetyl-purine riboside | 3056-18-6 | 10 mM * 1 mLin DMSO | 110元 |
| 2025/12/22 | XW02305618604 | 2,6-二氯-2'',3'',5''-三乙酰基嘌呤核糖苷 2,6-Dichloro-2',3',5'-triacetyl-purine riboside | 3056-18-6 | 5g | 585元 |
