34371-14-7
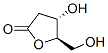 34371-14-7 结构式
34371-14-7 结构式
基本信息
D-(+)-核糖酸-GAMMA-内酯
(4S,5R)-4-羟基-5-(羟甲基)噁戊环-2-酮
(4S,5R)-4-羟基-5-(羟甲基)二氢呋喃-2(3H)-酮
2'-Deoxyribonolactone
2-deoxy-D-ribonolactone
2-Deoxy-D-ribono-1,4-lactone
2-Deoxy-D-ribonic-1,4-lactone
2-Deoxy-D-ribonic acid γ-lactone
2-Deoxy-D-ribonic acid-1,4-lactone
2-Deoxy-D-ribono-1,4-lactone >=95% (GC)
D-erythro-Pentonic acid, 2-deoxy-, γ-lactone
2,4,5-trihydroxypentanoic acid gamma-lactone
物理化学性质
制备方法
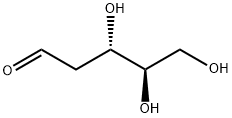
533-67-5
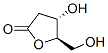
34371-14-7
实施例1A (4S,5R)-4-羟基-5-(羟甲基)二氢呋喃-2(3H)-酮的合成:在500 mL圆底烧瓶中加入2-脱氧-D-核糖(10 g,74.6 mmol)和水(60 mL),随后缓慢滴加溴(59.6 g,373 mmol)。反应混合物在室温下搅拌5天。反应完成后,将混合物用水(300 mL)稀释,并用叔丁基甲基醚(3×200 mL)进行萃取。水层用碳酸银中和至pH值为6,过滤除去生成的固体。滤液在真空下浓缩,得到残余物。将残余物溶解于乙酸乙酯(200 mL)中,加入硫酸镁干燥。静置过夜后,过滤除去固体,滤液浓缩后得到(4S,5R)-4-羟基-5-(羟甲基)二氢呋喃-2(3H)-酮,为油状物(7.72 g,78%)。1H NMR(400 MHz,CDCl3)δ 1.83(t,J = 5.9 Hz,1H),2.13(d,J = 4.3 Hz,1H),2.57(dd,J = 18.1, 4.0 Hz,1H),3.00(dd,J = 18.0, 7.2 Hz,1H),3.85(m,1H),3.97(m,1H),4.46(q,J = 3.2 Hz,1H),4.65(m,1H)。
参考文献:
[1] European Journal of Organic Chemistry, 2013, # 7, p. 1306 - 1317
[2] Patent: US2017/57981, 2017, A1. Location in patent: Paragraph 0268; 0269; 0320; 0321
[3] RSC Advances, 2014, vol. 4, # 22, p. 11023 - 11028
[4] Patent: WO2011/3018, 2011, A2. Location in patent: Page/Page column 40-41
[5] Patent: WO2015/81297, 2015, A1. Location in patent: Paragraph 00291