3951-95-9
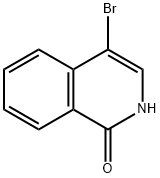 3951-95-9 结构式
3951-95-9 结构式
基本信息
1-羟基-4-溴异喹啉
4-溴-1-羟基-异喹啉
4-溴-1(2H)-异喹啉酮
1-羟基-4-溴异喹啉(4-溴异喹啉-1(2H)-酮)
4-Bromoisoquinolin-1-ol
4-BROMO-1(2H)-ISOQUINOLONE
4-BROMO-2H-ISOQUINOLIN-1-ONE
4-broMoisoquinolin-1(2H)-one
4-Bromo-1(2H)-isoquinolinone
4-BROMO-1-HYDROXYISOQUINOLINE
1(2H)-Isoquinolinone, 4-bromo-
4-bromo-1,2-dihydroisoquinolin-1-one
4-BROMO-1(2H)-ISOQUINOLONE ISO 9001:2015 REACH
物理化学性质
制备方法
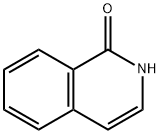
491-30-5

3951-95-9
以1-羟基异喹啉为原料合成4-溴-1(2H)-异喹啉酮的一般步骤:向含有异喹啉-1(2H)-酮(105 mg,0.723 mmol)的DMF(2 mL)溶液中加入N-溴代琥珀酰亚胺(NBS,142 mg,0.796 mmol)。将反应混合物在室温下搅拌2小时。反应完成后,将混合物浓缩以去除溶剂。粗产物通过制备型高效液相色谱(HPLC)进行纯化,得到中间体6 4-溴异喹啉-1(2H)-酮(110 mg,产率68%)。[C] MS(ESI)m/z:223.9(M + H)+。1H NMR(500 MHz,DMSO-d6)δ11.57(br.s,1H),8.24(dd,J = 8.0, 0.8 Hz,1H),7.88-7.83(m,1H),7.79-7.75(m,1H),7.61(ddd,J = 8.0, 7.1, 1.1 Hz,1H),7.55(s,1H)。
参考文献:
[1] Journal of Medicinal Chemistry, 2014, vol. 57, # 4, p. 1299 - 1322
[2] Angewandte Chemie - International Edition, 2011, vol. 50, # 36, p. 8416 - 8419
[3] Synthetic Communications, 2007, vol. 37, # 23, p. 4199 - 4208
[4] Patent: US2014/206686, 2014, A1. Location in patent: Paragraph 0497-0499
[5] Patent: WO2016/69976, 2016, A1. Location in patent: Page/Page column 91
