4253-81-0
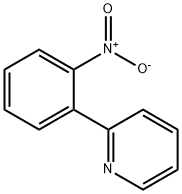 4253-81-0 结构式
4253-81-0 结构式
制备方法
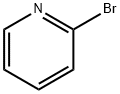
109-04-6
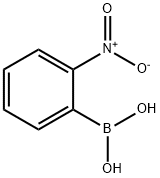
5570-19-4

4253-81-0
向含有K2CO3(4.65 g,33.6 mmol)、Pd(OAc)2(0.189 g,0.840 mmol)和PPh3(0.882 g,3.36 mmol)的170 mL DME/EtOH/H2O(4:1:1)悬浮液中,依次加入2-硝基苯硼酸(3.37 g,20.2 mmol)和2-溴吡啶(1.60 mL,16.8 mmol)。将反应混合物在70℃下搅拌过夜后,进行减压浓缩。向浓缩残余物中加入水(100 mL)和Et2O(100 mL),水层用Et2O(50 mL×2)萃取。合并的有机层用盐水(50 mL)洗涤,经Na2SO4干燥后蒸发溶剂。残余物通过硅胶柱色谱(洗脱剂:己烷/EtOAc = 6:1)纯化,得到2-(2-硝基苯基)吡啶(2.23 g,产率66%)为白色固体。产物的光谱数据与文献报道一致:Rf = 0.28(己烷/EtOAc = 2:1);1H NMR(400 MHz,CDCl3):δ 8.66(d,J = 5.0 Hz,1H),7.91(d,J = 8.2 Hz,1H),7.81(dd,J = 7.7, 7.7 Hz,1H),7.68(d,J = 7.2 Hz,1H),7.63(dd,J = 7.7, 7.7 Hz,1H),7.55(dd,J = 7.7, 7.2 Hz,1H),7.48(d,J = 8.2 Hz,1H),7.32(dd,J = 7.7, 5.0 Hz,1H)。
参考文献:
[1] Heterocycles, 2014, vol. 8, # 2, p. 1539 - 1551
[2] Patent: WO2009/58348, 2009, A1. Location in patent: Page/Page column 83
[3] Patent: CN108530490, 2018, A. Location in patent: Paragraph 0086; 0087; 0088; 0089