61160-18-7
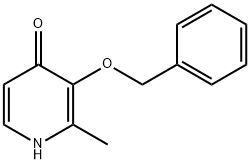 61160-18-7 结构式
61160-18-7 结构式
基本信息
2-甲基-3-苯基甲氧基-1H-吡啶-4-酮
3-(苄氧基)-2-甲基吡啶-4(1H)- 酮
3-BENZYLOXY-2-METHYL-4(1H)PYRIDONE
3-BENZYLOXY-2-METHYL-1H-PYRIDIN-4-ONE
3-(BENZYLOXY)-2-METHYL-4(1H)-PYRIDINONE
2-Methyl-3-phenylmethoxy-1H-pyridin-4-one
4(1H)-Pyridinone, 2-methyl-3-(phenylmethoxy)-
制备方法
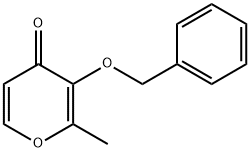
61049-69-2

61160-18-7
以3-(苄氧基)-2-甲基-4H-吡喃-4-酮为原料合成3-(苄氧基)-2-甲基吡啶-4(1H)-酮的一般步骤:向3-(苄氧基)-2-甲基-4H-吡喃-4-酮(13.8g,0.064mol)的乙醇(25mL)溶液中加入氨水溶液(50mL),回流反应过夜。反应完成后,减压蒸馏除去溶剂。将残余物溶于水,用浓盐酸调节pH至1。所得水相用乙酸乙酯(3×25mL)洗涤,随后用2M氢氧化钠溶液调节pH至10。水相再用氯仿(3×1L)萃取,合并有机相,用无水硫酸钠干燥,过滤,减压浓缩。粗产物用甲醇/乙醚混合溶剂重结晶,得到棕色立方晶体,熔点为162-164℃。产率为75%。产物经1H NMR(CDCl3)表征:δ 2.15(3H,s,CH3),5.03(2H,s,CH2Ph),6.35(1H,d,J = 6.9Hz,5-H),7.25-7.31(5H,m,Ph-H),7.39(1H,d,J = 6.9Hz,6-H)。分子式为C13H13NO2。
参考文献:
[1] Journal of Medicinal Chemistry, 1993, vol. 36, # 17, p. 2448 - 2458
[2] Journal of Heterocyclic Chemistry, 1992, vol. 29, # 4, p. 1017 - 1019
[3] Tetrahedron, 2001, vol. 57, # 16, p. 3479 - 3486
[4] Patent: WO2006/103463, 2006, A1. Location in patent: Page/Page column 9; Figure 2
[5] Bioorganic and Medicinal Chemistry, 2011, vol. 19, # 3, p. 1285 - 1297
