622867-52-1
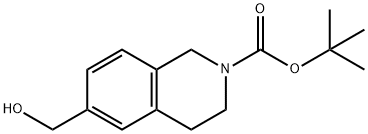 622867-52-1 结构式
622867-52-1 结构式
基本信息
6-羟基甲基-3,4-二氢-1H-异喹啉-2-羧酸叔丁酯
6-(羟基甲基)-3,4-二氢异喹啉-2(1H)-羧酸叔丁酯
叔-丁基 6-(羟甲基)-3,4-二氢异喹啉-2(1H)-甲酸基酯
tert-butyl 6-(hydroxymethyl)-3,4-dihydro-1H-isoquinoline-2-carboxylate
tert-butyl 6-(hydroxymethyl)-3,4-dihydroisoquinoline-2(1H)-carboxylate
6-Hydroxymethyl-3,4-dihydro-1H-isoquinoline-2-carboxylic acid tert-butyl ester
2(1H)-Isoquinolinecarboxylic acid, 3,4-dihydro-6-(hydroxymethyl)-, 1,1-dimethylethyl ester
物理化学性质
制备方法
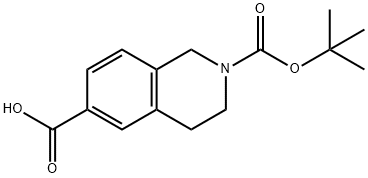
170097-67-3

622867-52-1
以2-(叔丁氧羰基)-1,2,3,4-四氢异喹啉-6-羧酸为原料合成6-(羟基甲基)-3,4-二氢异喹啉-2(1H)-羧酸叔丁酯的一般步骤:在实施例16中,中间体6-溴甲基-3,4-二氢-1H-异喹啉-2-羧酸叔丁酯(D-1)的制备如下:首先,在氮气保护下,将硼烷-THF络合物(99.17 mL,99.17 mmol)通过注射器缓慢加入至2-(叔丁氧羰基)-1,2,3,4-四氢异喹啉-6-羧酸(12.50 g,45.08 mmol)的无水THF(125.0 mL)溶液中,反应在25℃下持续16小时。随后,缓慢加入水(10.0 mL),接着加入2.0 M Na2CO3溶液(15.0 mL)。将混合物搅拌15分钟后,用EtOAc稀释,分离有机层。有机层用1M HCl洗涤,经MgSO4干燥,减压浓缩得到油状物。该油状物通过硅胶色谱法纯化,得到中间体D-1-1(11.8 g,99.3%收率),为白色固体。接着,将D-1-1(9.50 g,36.1 mmol)和N,N-二异丙基乙胺(9.43 mL,54.1 mmol)溶于二氯甲烷(200.0 mL)中,加入三苯基膦二溴化物(23.79 g,54.11 mmol),反应搅拌1小时后减压浓缩。所得残余物通过硅胶色谱法纯化,得到目标产物D-1(8.74 g,74%收率),为白色固体。
参考文献:
[1] Patent: US2016/24059, 2016, A1. Location in patent: Paragraph 0308-0309
[2] Patent: WO2012/122340, 2012, A1. Location in patent: Page/Page column 77
[3] Patent: US2014/73629, 2014, A1. Location in patent: Paragraph 0100; 0101
[4] Patent: WO2017/160632, 2017, A1. Location in patent: Page/Page column 78; 79
[5] Patent: WO2017/216726, 2017, A1. Location in patent: Page/Page column 643-644