885618-33-7
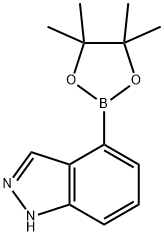 885618-33-7 结构式
885618-33-7 结构式
基本信息
1H-吲唑-4-硼酸频哪酯
1H-吲唑-4-硼酸频哪醇酯
4-(4,4,5,5-四甲基-1,3,2-二氧杂硼烷-2-基)-1H-吲唑
4-(4,4,5,5-四甲基-1,3,2-二氧杂硼烷-2-基)-1H-吲唑
4-(4,4,5,5-四甲基-1,3,2-二氧杂环戊硼烷-2-基)-1氢-吲唑
1H-吲唑, 4-(4,4,5,5-四甲基-1,3,2-二氧杂环己硼烷-2-基)-
4-(4,4,5,5-四甲基-1,3,2-二氧杂硼烷-2-基)-1H-吲唑,1H-吲唑-4-硼酸频哪醇酯
Indazole-4-boronic acid p...
2-dioxaborolan-2-yl)-1H-indazole
Pinacol ester indazole-4-boronic acid
1H-Indazole-4-boronic acid picol ester
1H-indazol-4-boronic acid pinacol ester
1H-Indazole-4-boronic acid pinacol ester
1H-Indazol-4-ylboronic Acid Pinacol Ester
4-(tetramethyl-1,3,2-dioxaborolan-2-yl)-1h-indazole
1H-Indazole, 4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-
物理化学性质
制备方法
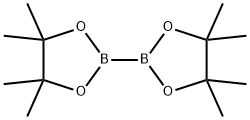
73183-34-3
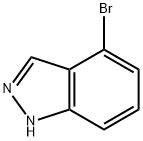
186407-74-9

885618-33-7
向4-溴-1H-吲唑(500 mg,2.54 mmol)和双(频哪醇合)二硼(1.5当量,3.81 mmol)的二甲基亚砜(DMSO,20 mL)溶液中加入干燥的乙酸钾(3.0当量,7.61 mmol,747 mg)和PdCl2(dppf)2(3 mol%,0.076 mmol,62 mg)。将反应混合物用氩气脱气,并在80℃下加热反应40小时。反应完成后,冷却至室温,将反应混合物在水(50 mL)和乙醚(3×50 mL)之间分配。合并有机层,用盐水(50 mL)洗涤,分离后用无水硫酸镁(MgSO4)干燥。通过柱色谱法纯化粗产物,使用30%-40%乙酸乙酯/石油醚作为洗脱剂,得到1H-吲唑-4-硼酸频哪醇酯(369 mg,60%)和未反应的4-溴-1H-吲唑(60 mg,20%)的不可分离的3:1混合物。该混合物为黄色胶状物,静置后固化为灰白色固体。1H NMR(400 MHz,DMSO-d6)数据如下:1H-吲唑-4-硼酸频哪醇酯(70):δ 1.41(12H,s),7.40(1H,dd,J = 8.4 Hz,6.9 Hz),7.59(1H,d,J = 8.4 Hz),7.67(1H,d,J = 6.9 Hz),10.00(1H,br s),8.45(1H,s);4-溴-1H-吲唑:δ 7.40(1H,m),7.18(1H,t,J = 7.9 Hz),7.50(1H,d,J = 9.1 Hz),7.77(1H,d,J = 7.9 Hz),8.09(1H,s)。杂质峰出现在δ 1.25。
参考文献:
[1] Patent: WO2007/42806, 2007, A1. Location in patent: Page/Page column 34-35
[2] Patent: WO2011/130628, 2011, A1. Location in patent: Page/Page column 128
[3] Patent: US2013/102595, 2013, A1. Location in patent: Paragraph 0173
[4] Patent: JP2015/187145, 2015, A. Location in patent: Paragraph 0171
[5] Bioorganic and Medicinal Chemistry, 2014, vol. 22, # 3, p. 1156 - 1162
