除虫菊素I
|
|
|
- CAS号:
- 121-21-1
- 英文名:
- PYRETHRIN 1
- 英文别名:
- piretrina1;pyrethrini;PYRETHRIN 1;Pyrethrin Ⅰ;pyrethrinei;pyrethrins I;piretrina1(portuguese);pyrethrins(pyrethrini);Spirodiclofen Impurity 14;openyl)cyclopropanecarboxylate
- 中文名:
- 除虫菊素I
- 中文别名:
- 除虫菊酯1;除虫菊素I;除虫菊酯I, 100ΜG /ΜL于环己烷;除虫菊酯 I (标准品)100 NG/UL
- CBNumber:
- CB4782827
- 分子式:
- C21H28O3
- 分子量:
- 328.45
- MOL File:
- 121-21-1.mol
|
|
|
除虫菊素I化学性质
-
比旋光度:
-
D20 -14° (isooctane)
-
|
-
沸点:
-
bp0.0005 146-150°
-
|
-
密度:
-
1.5192
-
|
-
折射率:
-
nD20 1.5242
-
|
-
储存条件:
-
0-6°C
-
|
-
溶解度:
-
Chloroform (Slightly), Ethyl Acetate (Slightly)
-
|
-
形态:
-
Oil
-
|
-
颜色:
-
Colourless to Pale Yellow
-
|
-
稳定性:
-
Light Sensitive, Temperature Sensitive
-
|
-
EPA化学物质信息:
-
Pyrethrin I (121-21-1)
-
|
除虫菊素I性质、用途与生产工艺
除虫菊素为天然除虫菊的提取物,具有杀虫药用价值,内含除虫菊酯Ⅰ、除虫菊酯Ⅱ、瓜菊酯和茉莉菊酯。除虫菊素的杀虫谱广,有较高的胃毒作用,对咀嚼式口器害虫有特效;又有强烈的触杀作用,主要用于防治刺吸式口器害虫;且击倒快,对哺乳动物安全,易降解,不污染环境。40年代中期,化学合成农药如有机氯、有机磷和氨基酸甲酯类杀虫剂尚未问世前,除虫菊制剂就被广泛地用于防治农业害虫,并取得了良好效果。从1947年起,人工首次合成拟除虫菊酯获得成功,目前已经人工合成的拟除虫菊酯类化合物有:烯丙菊酯(1947年)、胺菊酯(1963年),苄呋菊酯(1965年)、苯醚菊酯(1968年)、苄氯菊酯(1972年)、氯氰菊酯(1973年)、溴氰菊酯(1973年)以及非三碳环的杀灭菊酯等,其药效、光稳定性超过天然除虫菊酯及有机磷、有机氯、氨基甲酸三类常用合成杀虫剂。近年来,合成产物逐步代替了天然物,不仅用于室内灭虫,也用于农业害虫的防治。
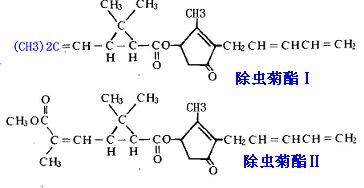 图1为除虫菊素主要成分除虫菊酯Ⅰ和除虫菊酯Ⅱ的化学结构式
图1为除虫菊素主要成分除虫菊酯Ⅰ和除虫菊酯Ⅱ的化学结构式
以上信息由Chemicalbook的晓楠编辑整理。
除虫菊素I
上下游产品信息
上游原料
下游产品
除虫菊素I
生产厂家
全球有 39家供应商
除虫菊素I国内生产厂家
121-21-1, 除虫菊素I 相关搜索:
- 有机氯杀虫剂
- 杀虫剂
- 除虫菊酯1
- 除虫菊酯 I (标准品)100 NG/UL
- 除虫菊酯I, 100ΜG /ΜL于环己烷
- 除虫菊素I
- 121-21-1
- Spirodiclofen Impurity 14
- Cyclopropanecarboxylicacid, 2,2-dimethyl-3-(2-methyl-1-propen-1-yl)-,(1S)-2-methyl-4-oxo-3-(2Z)-2,4-pentadien-1-yl-2-cyclopenten-1-yl ester,(1R,3R)-
- ydroxy-3-methyl-2-(2,4-pentadienyl)-2-cyclopenten-1-one
- pyrethrolone,chrysanthemummonocarboxylicacidester
- pyrethrins(pyrethrini)
- pyrethrins-(cinerini,cinerinii,pyrethrini,pyrethrinii)
- pyrethrini
- pyrethrinei
- piretrina1(portuguese)
- piretrina1
- oxy-3-methyl-2-(2,4-pentadienyl)-2-cyclopenten-1-one
- oxo-3-(2,4-pentadienyl)-2-cyclopenten-1-ylester,[1theta-[1alpha[s(z)],3bet
- openyl)cyclopropanecarboxylate
- chrysanthemummonocarboxylicacidpyrethroloneester
- 4-pentadienyl)-2-cyclopenten-1-ylester,(1r-(1alpha(s*(z)),3beta))-oxo-3-(
- -3-(2,4-pentadienyl)-2-cycloenten-1-ylester
- 2-methyl-4-oxo-3-(penta-2,4-dienyl)cyclopent-2-enylchrysanthemate
- 2-methyl-4-oxo-3-(2,4-pentadienyl)-2-cyclopenten-2,2-dimethyl-3-(2-methyl-1-pr
- 2-dimethyl-3-(2-methyl-1-propenyl)-2-methyl-4-cyclopropanecarboxylicaci
- 2,2-dimethyl-3-(2-methylpropenyl)-cyclopropanecarboxylicaciesterwith4-h
- 2,2-dimethyl-3-(2-methylpropenyl)-cyclopropaneacrylicaciesterwith4-hydr
- 2,2-dimethyl-3-(2-methyl-1-propenyl)cyclopropanecarboxylicacid2-methyl-4-oxo
- (+)-pyrethronyl(+)-trans-chrysanthemate
- Pyrethrin I, 100 μg /μL in cyclohexane
- Pyrethrin Ⅰ
- pyrethrins I
- (1R,3R)-2,2-Dimethyl-3-(2-methyl-1-propenyl)cyclopropanecarboxylic acid (S)-2-methyl-4-oxo-3-[(Z)-2,4-pentadienyl]-2-cyclopenten-1-yl
- (1R,3R)-2,2-Dimethyl-3-(2-methyl-1-propenyl)cyclopropane-1-carboxylic acid (S)-2-methyl-3-[(Z)-2,4-pentadienyl]-4-oxo-2-cyclopentene-1-yl ester
- 2-methyl-4-oxo-3-(penta-2,4-dienyl)cyclopent-2-enyl [1R-[1α[S*(Z)],3β]]-chrysanthemate pyrethrin I
- 2-Methyl-4-oxo-3-(penta-2,4-dienyl)cyclopent-2-enyl-[1R[a [S*(Z)], 3b ]]-chrysanthemat
- Cyclopropanecarboxylic acid, 2,2-dimethyl-3-(2-methyl-1-propenyl)-, (1S)-2-methyl-4-oxo-3-(2Z)-2,4-pentadienyl-2-cyclopenten-1-yl ester, (1R,3R)-
- 2-methyl-4-oxo-3-(penta-2,4-dienyl)cyclopent-2-enyl [1R-[1alpha[S*(Z)],3beta]]-chrysanthemate
- PYRETHRIN 1