Scandium trifluoromethanesulfonate Chemische Eigenschaften,Einsatz,Produktion Methoden
R-Sätze Betriebsanweisung:
R36/37/38:Reizt die Augen, die Atmungsorgane und die Haut.
S-Sätze Betriebsanweisung:
S26:Bei Berührung mit den Augen sofort gründlich mit Wasser abspülen und Arzt konsultieren.
S36:DE: Bei der Arbeit geeignete Schutzkleidung tragen.
Beschreibung
Scandium trifluoromethanesulfonate, commonly called Scandium(III) triflate, is a chemical compound with formula Sc(SO3CF3)3, a salt consisting of scandium cations Sc3+ and triflate SO3CF3? anions.
Scandium(III) triflate is an extremely active, efficient, recoverable and reusable acylation catalyst. Its an important catalyst for the Friedel-Crafts acylation, Diels-Alder reactions and other carbon-carbon bond-forming reactions. It also stereochemically catalyzes the radical polymerization of acrylates. Scandium(III) triflate complex of (4′S,5′S)-2,6-bis[4′-(triisopropylsilyl)oxymethyl-5′-phenyl-1′,3′-oxazolin-2′-yl]pyridine has been employed as catalyst for the asymmetric Friedel-Crafts reaction between substituted indoles and methyl (E)-2-oxo-4-aryl-3-butenoates.
Chemische Eigenschaften
White powder
Verwenden
Scandium(III) trifluoromethanesulfonate is widely used as a catalyst in hydrothiolation, selective two-electron reduction of oxygen by ferrocene derivatives and vinylogous Fridel-crafts alkylation of indoles and pyrrole in water. It is involved in the Mukaiyama aldol addition and stereochemically catalyzes the radical polymerization of acrylates. It acts as a Lewis acid catalyst and used in the synthesis of bullvalone via a stabilized sulfur ylide.
Application
Scandium(III) triflate was used as a catalyst in:
Hydrothiolation reaction of aromatic and aliphatic thiols.
Selective two-electron reduction of O2 by ferrocene derivatives.
Vinylogous Friedel-Crafts alkylation of indoles and pyrroles in water.
Synthesis of β-cyanoketones.
Combination with triethylsilane to reductively open functionalized pyranoside rings.
The key steps of synthesis of bullvalone via a stabilized sulfur ylide.
Reaktionen
-
-
Water tolerant Lewis acid.
-
-
Commonly used in a range of Lewis acid catalyzed reactions.
-
-
Efficient metal source for Lewis acid catalyzed asymmetric reactions.
-
-
Catalyzes Friedel-Crafts alkylation, acylation and related reactions.
-
-
Catalyzes various domino- and multi-component processes.
-
-
Catalyzes electrophilic additions of alpha-diazoesters with ketones.
-
-
Catalyzes carbon insertion reactions.
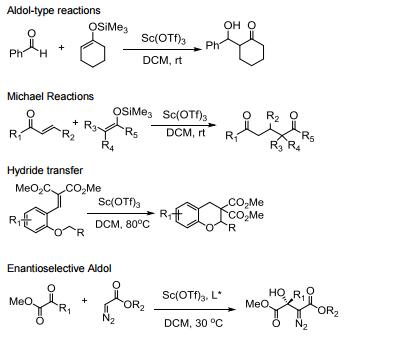
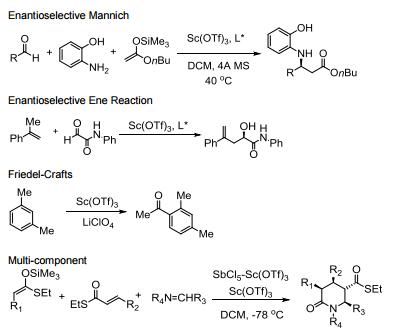
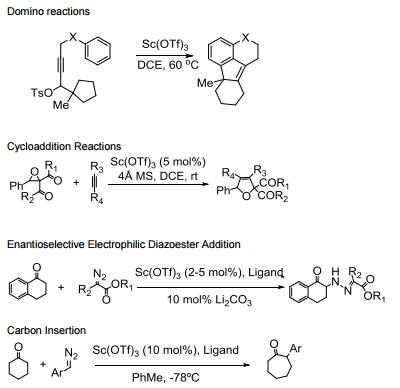
Allgemeine Beschreibung
Scandium(III) triflate is an extremely active, efficient, recoverable and reusable acylation catalyst. Its an important catalyst for the Friedel-Crafts acylation, Diels-Alder reactions and other carbon-carbon bond-forming reactions. It also stereochemically catalyzes the radical polymerization of acrylates. Scandium(III) triflate complex of (4′
S,5′
S)-2,6-bis[4′-(triisopropylsilyl)oxymethyl-5′-phenyl-1′,3′-oxazolin-2′-yl]pyridine has been employed as catalyst for the asymmetric Friedel-Crafts reaction between substituted indoles and methyl (
E)-2-oxo-4-aryl-3-butenoates.
Scandium trifluoromethanesulfonate Upstream-Materialien And Downstream Produkte
Upstream-Materialien
Downstream Produkte

