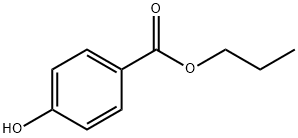
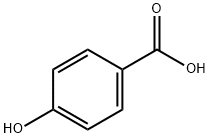
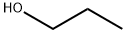Propylparaben
- CAS No.
- 94-13-3
- Chemical Name:
- Propylparaben
- Synonyms
- Propyl parahydroxybenzoate;Parabens;PROPYL 4-HYDROXYBENZOATE;Nipazol;PROPYL P-HYDROXYBENZOATE;Nipasol;Nipasol M;4-HYDROXYBENZOIC ACID PROPYL ESTER;PROPYLPARABENE;Propyl parahydroxybenzoate CRS
- CBNumber:
- CB0176673
- Molecular Formula:
- C10H12O3
- Molecular Weight:
- 180.2
- MDL Number:
- MFCD00002354
- MOL File:
- 94-13-3.mol
- MSDS File:
- SDS
| Melting point | 95-98 °C(lit.) |
|---|---|
| Boiling point | 133°C |
| Density | 1.0630 |
| vapor pressure | 0.67 hPa (122 °C) |
| refractive index | 1.5050 |
| FEMA | 2951 | PROPYL P-HYDROXYBENZOATE |
| Flash point | 180°(356°F) |
| storage temp. | Sealed in dry,Room Temperature |
| solubility | ethanol: soluble0.1M, clear, colorless |
| form | Crystalline Powder |
| pka | pKa 8.4 (Uncertain) |
| color | White |
| Specific Gravity | 0.789 (20/4℃) |
| Odor | at 100.00 %. sweet smoky burnt woody hawthorn |
| PH | 6-7 (H2O, 20°C) (saturated solution) |
| Odor Type | smoky |
| Water Solubility | <0.1 g/100 mL at 12 ºC |
| Merck | 14,7866 |
| BRN | 1103245 |
| Stability | Stable. Incompatible with strong oxidizing agents, strong bases. |
| InChIKey | QELSKZZBTMNZEB-UHFFFAOYSA-N |
| LogP | 2.8 at 20℃ |
| FDA 21 CFR | 184.1670; 582.3670; 172.515; 201.327; 310.545; 352.70 |
| Substances Added to Food (formerly EAFUS) | PROPYL P-HYDROXYBENZOATE |
| SCOGS (Select Committee on GRAS Substances) | Propyl Paraben |
| CAS DataBase Reference | 94-13-3(CAS DataBase Reference) |
| EWG's Food Scores | 5-7 |
| FDA UNII | Z8IX2SC1OH |
| NIST Chemistry Reference | N-propyl-p-hydroxy-benzoate(94-13-3) |
| EPA Substance Registry System | Propylparaben (94-13-3) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H315-H319-H317 | |||||||||
| Precautionary statements | P501-P261-P272-P264-P280-P302+P352-P337+P313-P305+P351+P338-P362+P364-P333+P313 | |||||||||
| Hazard Codes | Xi | |||||||||
| Risk Statements | 36/37/38 | |||||||||
| Safety Statements | 26-24/25-36 | |||||||||
| WGK Germany | 1 | |||||||||
| RTECS | DH2800000 | |||||||||
| Autoignition Temperature | >600 °C | |||||||||
| TSCA | Yes | |||||||||
| HS Code | 29182930 | |||||||||
| Toxicity | LD50 orally in Rabbit: > 5000 mg/kg | |||||||||
| NFPA 704 |
|
Propylparaben price More Price(40)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | 54789 | Propyl 4-hydroxybenzoate tested according to Ph. Eur. | 94-13-3 | 1kg | $203 | 2024-03-01 | Buy |
| Sigma-Aldrich | 16897 | Propylparaben certified reference material, TraceCERT | 94-13-3 | 100mg | $371 | 2024-03-01 | Buy |
| Sigma-Aldrich | 1.07427 | Propyl 4-hydroxybenzoate EMPROVE? ESSENTIAL Ph Eur,BP,JP,NF,FCC | 94-13-3 | 5kg | $404 | 2024-03-01 | Buy |
| Sigma-Aldrich | 1577008 | Propylparaben United States Pharmacopeia (USP) Reference Standard | 94-13-3 | 200mg | $474 | 2024-03-01 | Buy |
| Sigma-Aldrich | 1.07427 | Propyl 4-hydroxybenzoate EMPROVE? ESSENTIAL Ph Eur,BP,JP,NF,FCC | 94-13-3 | 25kg | $1269 | 2024-03-01 | Buy |
Propylparaben Chemical Properties,Uses,Production
Chemical properties
Propylparaben is a colorless and fine crystalline or white crystalline powder, almost odorless and with slightly astringent.Soluble in ethanol, ethyl ether, acetone and other organic solvents, slightly soluble in water.
Uses
1. Propylparaben Used as preservatives and antioxidants, and also used in the pharmaceutical industry
2. Used as the antimicrobial preservative in pharmaceuticals and cosmetics
3. According to ethyl p-hydroxybenzoate (17007)
4. Used as antiseptic and antimicrobial.
5. Used as the preservatives of food, cosmetics and medicines.
content analysis
Same with Method 1 in "Butyl p-hydroxybenzoate (07002)".
In calculation, per mL of 1 mol/L sodium hydroxide corresponds to 180.2mg of this goods (C10Hl2O8).
Toxicity
Adl 0-10 mg/kg (FAO/WHO, 2001).
LD50 3.7g/kg (mouse, oral).
GRAS (FDA, § 184.1670, 2000).
HACSG listed in the restricted list.
usage limits
2760--2002 GB (calculate in p-hydroxybenzoic acid; g/kg):
fresh fruit and vegetable 0.012; vinegar 0.10; carbonated beverages 0.20; fruit juice (fruit flavor) type beverages, jam (excluding canned), soy sauces 0.25; pastry stuffing 0.5 (the total amount of single use or mixed use with ethyl 4-hydroxybenzoate); egg yolk filling 0.20.
Another provision that sodium methylparaben is also equivalent application.
FAO/WHO (1984): Jam and jelly, 1000 mg/kg
FDA § 184.1670 (2000): 0.1%.
EEC(1990, mg/kg): Frozen drinks 160; beet pickled vegetables, salad dressings, 250; fragrance, fruit tarts, purees, concentrated soft drinks, 800; Fruit canned, salted fish, 1000.
Japan (calculate in p-hydroxybenzoic acid, g/kg; this product, g):
soy sauce 0.25g/L (this product 0.32); vinegarsoysoy 0.1g/L (This product 0.13); soft drinks and syrup 0.1 (this product 0.13); fruit sauce 0.2 (this product 0.26); fruits and vegetables 0.012 (this product 0.015).
Production methods
This product can be derived from the esterification of p-hydroxybenzoic acid and n-propanol. First mix p-hydroxybenzoic acid with propanol and heat to dissolve. Then add sulfuric acid slowly and continue to heat for 8h of refluxion. After cooling, pour them into the 4% sodium carbonate solution for precipitation and crystallization. Filtrate and wash to neutral to obtain the crude product. After further ethanol recrystallization, the finished products are obtained. In the preparation, the cation exchange resin can be used in place of the sulfuric acid catalyst.
It can be derived from the esterification of p-hydroxybenzoic acid and n-propanol in the presence of sulfuric acid.
Add p-hydroxybenzoic acid and n-propanol in turn to the esterification reactor, and heat to dissolve. Add concentrated sulfuric acid slowly and heat for 8h of refluxion. Pour the reaction solution into 4% sodium carbonate solution before it is cooled. Constantly stir for precipitation and crystallization. Then the crude product can be obtained after centrifugal filtration and washed to neutral. Finally the finished product is acquired after activated carbon decolorization and ethanol recrystallization. The method of preparing ethyl p-hydroxybenzoate can also be used as a reference.
HOC6H4COOH + C3H7OH [H2SO4] → HOC6H4COOC3H7 + H2O
Hazards & Safety Information
Category: Toxic substances
Toxicity classification :Moderate toxicity
Acute Toxicity :Celiac-mouse LD50: 200 mg/kg
Flammable hazardous characteristics :Flammable; excrete acrid and pungent smoke from fire
Storage and transport characteristics :Stored in the low-temperature, well-ventilated and dry warehouse
Fire extinguishing agent :water, carbon dioxide, dry powder, sand
Chemical Properties
Propyl p-hydroxybenzoate is almost odorless.
Chemical Properties
White or almost white, crystalline powder.
Chemical Properties
Propylparaben occurs as a white, crystalline, odorless, and tasteless powder.
Uses
An antimicrobial
Uses
propylparaben is one of the most frequently used preservatives against bacteria and mold. It has a low sensitizing and low toxicity factor, is reputed to be very safe, and considered to be a noncomedogenic raw material.
Uses
Pharmaceutic aid (antifungal). Antimicrobial preservative in foods and cosmetics.
Definition
ChEBI: The benzoate ester that is the propyl ester of 4-hydroxybenzoic acid. Preservative typically found in many water-based cosmetics, such as creams, lotions, shampoos and bath products. Also used as a food additive.
Production Methods
Propylparaben is prepared by the esterification of p-hydroxybenzoic acid with n-propanol.
Preparation
Produced by esterfying p-hydroxybenzoic acid with n-propanol, using an acid catalyst such as sulfuric acid and an excess of propanol. The materials are heated in a glass-lined reactor under reflux. The acid is then neutralized with caustic soda and the product is crystallized by cooling. The crystallized product is centrifuged, washed, dried under vacuum, milled and blended, all in corrosion-resistant equipment to avoid metallic contamination.
Aroma threshold values
Detection: 20 ppb
General Description
Colorless crystals or white powder or chunky white solid. Melting point 95-98°C. Odorless or faint aromatic odor. Low toxicity, Tasteless (numbs the tongue). pH: 6.5-7.0 (slightly acidic) in solution.
Air & Water Reactions
Water soluble [Hawley].
Reactivity Profile
Maximum stability of Propylparaben occurs at a pH of 4 to 5. Incompatible with alkalis and iron salts. Also incompatible with strong oxidizing agents and strong acids .
Fire Hazard
Flash point data for Propylparaben are not available; however, Propylparaben is probably combustible.
Flammability and Explosibility
Non flammable
Pharmaceutical Applications
Propylparaben is widely used as an antimicrobial preservative in
cosmetics, food products, and pharmaceutical formulations.
It may be used alone, in combination with other paraben esters,
or with other antimicrobial agents. It is one of the most frequently
used preservatives in cosmetics.
The parabens are effective over a wide pH range and have a
broad spectrum of antimicrobial activity, although they are most
effective against yeasts and molds.
Owing to the poor solubility of the parabens, the paraben salts,
particularly the sodium salt, are frequently used in formulations.
This may cause the pH of poorly buffered formulations to become
more alkaline.
Propylparaben (0.02% w/v) together with methylparaben
(0.18% w/v) has been used for the preservation of various
parenteral pharmaceutical formulations.
Contact allergens
This substance is one of the parabens family. Parabens are esters formed by p-hydroxybenzoic acid and an alcohol. They are largely used as biocides in cosmetics and toiletries, medicaments, or food. They have synergistic power with other biocides. Parabens can induce allergic contact dermatitis, mainly in chronic dermatitis and wounded skin.
Safety
Propylparaben and other parabens are widely used as antimicrobial
preservatives in cosmetics, food products, and oral and topical
pharmaceutical formulations.
Propylparaben and methylparaben have been used as preservatives
in injections and ophthalmic preparations; however, they are
now generally regarded as being unsuitable for these types of
formulations owing to the irritant potential of the parabens.
Systemically, no adverse reactions to parabens have been
reported, although they have been associated with hypersensitivity
reactions. The WHO has set an estimated acceptable total daily
intake for methyl, ethyl, and propyl parabens at up to 10 mg/kg
body-weight.
LD50 (mouse, IP): 0.2 g/kg
LD50 (mouse, oral): 6.33 g/kg
LD50 (mouse, SC): 1.65 g/kg
storage
Aqueous propylparaben solutions at pH 3–6 can be sterilized by autoclaving, without decomposition.At pH 3–6, aqueous solutions are stable (less than 10% decomposition) for up to about 4 years at room temperature, while solutions at pH 8 or above are subject to rapid hydrolysis (10% or more after about 60 days at room temperature).
Incompatibilities
The antimicrobial activity of propylparaben is reduced considerably
in the presence of nonionic surfactants as a result of micellization.
Absorption of propylparaben by plastics has been reported, with
the amount absorbed dependent upon the type of plastic and the
vehicle. Magnesium aluminum silicate, magnesium trisilicate,
yellow iron oxide, and ultramarine blue have also been reported to
absorb propylparaben, thereby reducing preservative efficacy.
Propylparaben is discolored in the presence of iron and is subject
to hydrolysis by weak alkalis and strong acids.
Regulatory Status
Propylparaben and methylparaben are affirmed GRAS direct food
substances in the USA at levels up to 0.1%. All esters except the
benzyl ester are allowed for injection in Japan.
In cosmetics, the EU and Brazil allow use of each paraben at
0.4%, but the total of all parabens may not exceed 0.8%. The upper
limit in Japan is 1.0%.
Accepted as a food additive in Europe. Included in the FDA
Inactive Ingredients Database (IM, IV, and SC injections; inhalations;
ophthalmic preparations; oral capsules, solutions, suspensions,
and tablets; otic, rectal, topical, and vaginal preparations).
Included in parenteral and nonparenteral medicines licensed in the
UK. Included in the Canadian List of Acceptable Non-medicinal
Ingredients.
Propylparaben Preparation Products And Raw materials
Raw materials
1of2
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Jiangxi Alpha Hi-tech Pharmaceutical Co., Ltd | 510-85010237 | overseamarketing@alphahi-tech.com | CHINA | 41 | 58 |
| Hefei TNJ Chemical Industry Co.,Ltd. | 0551-65418671 | sales@tnjchem.com | China | 34572 | 58 |
| Hebei Yibangte Import and Export Co. , Ltd. | +86-0311-85030958 +8615532196582 | lisa@yibangte.com | China | 2988 | 58 |
| Nanjing Deda New Material Technology Co., Ltd | +8613223293093 | bella@njdeda.com | China | 81 | 58 |
| Hebei Jingbo New Material Technology Co., Ltd | +8619931165850 | hbjbtech@163.com | China | 1000 | 58 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21695 | 55 |
| Hangzhou FandaChem Co.,Ltd. | 008657128800458; +8615858145714 | fandachem@gmail.com | China | 9352 | 55 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418679 +86-18949832763 | info@tnjchem.com | China | 2989 | 55 |
| Tianjin Zhongxin Chemtech Co., Ltd. | +86-022-66880623 +8618622897568 | sales@tjzxchem.com | China | 559 | 58 |
| Shanghai Zheyan Biotech Co., Ltd. | 18017610038 | zheyansh@163.com | CHINA | 3620 | 58 |
Related articles
- Propylparaben: A Common Antimicrobial and Preservative Chemical Found in Personal Care Products and Food
- Propylparaben, a paraben compound, is widely used for its antimicrobial and preservative properties in various consumer produc....
- Dec 20,2023
- Propylparaben in Skincare
- The passage talks about how Propylparaben works in skincare.
- Nov 29,2022
View Lastest Price from Propylparaben manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2024-04-19 | Propyl paraben
94-13-3
|
US $20.00 / KG | 20KG | 99% | 1000kg | Hebei Yime New Material Technology Co., Ltd. | |
 |
2024-04-19 | Propylparaben
94-13-3
|
US $10.00-5.00 / kg | 1kg | 99.5 | 10 ton per month | Nanjing Deda New Material Technology Co., Ltd | |
 |
2024-04-19 | Propylparaben
94-13-3
|
US $10.00-5.00 / kg | 1kg | 99.5 | 10 ton per month | Nanjing Deda New Material Technology Co., Ltd |
-

- Propyl paraben
94-13-3
- US $20.00 / KG
- 99%
- Hebei Yime New Material Technology Co., Ltd.
-

- Propylparaben
94-13-3
- US $10.00-5.00 / kg
- 99.5
- Nanjing Deda New Material Technology Co., Ltd
-

- Propylparaben
94-13-3
- US $10.00-5.00 / kg
- 99.5
- Nanjing Deda New Material Technology Co., Ltd