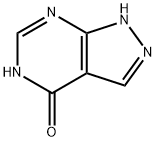
Allopurinol
- CAS No.
- 315-30-0
- Chemical Name:
- Allopurinol
- Synonyms
- HPP;ALO;1H-PYRAZOLO[3,4-D]PYRIMIDIN-4-OL;Zyloprim;1H-pyrazolo[4,3-d]pyriMidin-7-ol;Allopurinol CRS;4-HPP;Pural;Remid;Urbol
- CBNumber:
- CB1181254
- Molecular Formula:
- C5H4N4O
- Molecular Weight:
- 136.11
- MDL Number:
- MFCD00599413
- MOL File:
- 315-30-0.mol
- MSDS File:
- SDS
| Melting point | >300 °C (lit.) |
|---|---|
| Boiling point | 250.36°C (rough estimate) |
| Density | 1.4295 (rough estimate) |
| refractive index | 1.8500 (estimate) |
| storage temp. | 15-25°C |
| solubility | 1 M NaOH: soluble50mg/mL, clear to very slightly hazy, colorless to faintly yellow |
| form | Powder |
| pka | 10.2(at 25℃) |
| color | White or almost white |
| Water Solubility | 0.35 g/L (25 ºC) |
| Merck | 14,279 |
| BCS Class | 3,1 |
| InChIKey | OFCNXPDARWKPPY-UHFFFAOYSA-N |
| CAS DataBase Reference | 315-30-0(CAS DataBase Reference) |
| EWG's Food Scores | 1 |
| NCI Dictionary of Cancer Terms | allopurinol |
| FDA UNII | 63CZ7GJN5I |
| NCI Drug Dictionary | Lopurin |
| ATC code | M04AA01,M04AA51 |
| NIST Chemistry Reference | Allopurinol(315-30-0) |
| EPA Substance Registry System | Allopurinol (315-30-0) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS06 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H301-H317 | |||||||||
| Precautionary statements | P280-P301+P310+P330-P302+P352 | |||||||||
| Hazard Codes | T,Xi,Xn | |||||||||
| Risk Statements | 25-43-36/37/38-20/21/22 | |||||||||
| Safety Statements | 28-36/37-45-36/37/39-26-24-36 | |||||||||
| RIDADR | UN 2811 6.1/PG 3 | |||||||||
| WGK Germany | 2 | |||||||||
| RTECS | UR0785000 | |||||||||
| TSCA | Yes | |||||||||
| HazardClass | 6.1 | |||||||||
| PackingGroup | III | |||||||||
| HS Code | 29335990 | |||||||||
| Toxicity | LD50 oral in mouse: 78mg/kg | |||||||||
| NFPA 704 |
|
Allopurinol price More Price(55)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | A8003 | Allopurinol xanthine oxidase inhibitor | 315-30-0 | 5g | $80.5 | 2024-03-01 | Buy |
| Sigma-Aldrich | 1013002 | Allopurinol United States Pharmacopeia (USP) Reference Standard | 315-30-0 | 250mg | $436 | 2024-03-01 | Buy |
| Sigma-Aldrich | BP870 | Allopurinol British Pharmacopoeia (BP) Reference Standard | 315-30-0 | 200MG | $252 | 2023-06-20 | Buy |
| TCI Chemical | A0907 | Allopurinol >98.0%(T) | 315-30-0 | 25g | $31 | 2024-03-01 | Buy |
| TCI Chemical | A0907 | Allopurinol >98.0%(T) | 315-30-0 | 250g | $157 | 2021-12-16 | Buy |
Allopurinol Chemical Properties,Uses,Production
Chemical Properties
White to Off-White Solid
Originator
Zyloprim ,Burroughs-Wellcome ,US ,1966
Uses
Xanthine oxidase inhibitor; decreases uric acid production. Used in treatment of hyperuricemia and chronic gout. Antiurolithic
Uses
Allopurinol does not reduce serum uric acid levels by increasing renal uric acid excretion; instead it lowers plasma urate levels by inhibiting the final steps in uric acid biosynthesis.
Uric acid in humans is formed primarily by xanthine oxidase-catalyzed oxidation of hypoxanthine and xanthine to uric acid. Allopurinol (8) and its primary metabolite, alloxanthine (9) [CAS: 2465-59-0], are inhibitors of xanthine oxidase. Inhibition of the last two steps in uric acid biosynthesis by blocking xanthine oxidase reduces the plasma concentration and urinary excretion of uric acid and increases the plasma levels and renal excretion of the more soluble oxypurine precursors. Normally, in humans the urinary purine content is almost solely uric acid; treatment with allopurinol results in the urinary excretion of hypoxanthine, xanthine, and uric acid, each with its independent solubility. Lowering the uric acid concentration in plasma below its limit of solubility facilitates the dissolution of uric acid deposits. The effectiveness of allopurinol in the treatment of gout and hyperuricemia that results from hematogical disorders and antineoplastic therapy has been demonstrated.
Uses
antihyperuricemia, antigout, antiurolithic
Definition
ChEBI: Allopurinol is a bicyclic structure comprising a pyrazole ring fused to a hydroxy-substituted pyrimidine ring. It has a role as a radical scavenger, a gout suppressant, an antimetabolite and an EC 1.17.3.2 (xanthine oxidase) inhibitor. It is an organic heterobicyclic compound and a nucleobase analogue. It derives from a hydride of a 1H-pyrazolo[4,3-d]pyrimidine.
Indications
Allopurinol (Zyloprim) is the drug of choice in the treatment of chronic tophaceous gout and is especially useful in patients whose treatment is complicated by renal insufficiency.
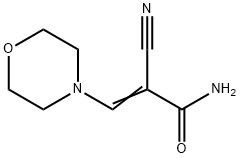
Manufacturing Process
3-Morpholino-2-cyanoacrylamide: A stirred mixture of cyanoacetamide (63 g),
triethylorthoformate (134 g), morpholine (82.5 g) and acetonitrile (37.5 ml)
was heated under reflux for 4 hours. The initial reflux temperature was 117°C
and the final reflux temperature was 82°C.
At the end of the reflux period the mixture was cooled to 30°C and the heavy crystalline precipitate was collected and washed with 2 x 75 ml of ethanol.
The product was dried in vacuum at 30°C. Wt = 111 g. Yield = 82%, MP 173-
175°C.
3-Aminopyrazole-4-carbxamide hemisulfate: To water (253 ml) at 60°C was
added 3-morpholino-2-cyanoacrylamide (63.4 g) and 85% technical hydrazine
hydrate (22.7 g). The mixture was rapidly heated to 95°C and the
temperature was maintained at >90°C for 20 minutes. The mixture was then
cooled to 60°C and the pH carefully adjusted to 1.5 by the addition of a
mixture of sulfuric acid (45.7 g) and ice. The acidified reaction was cooled to
5°C and the crystalline product collected and washed with cold water (2 x 100
ml) and acetone (2 x 50 ml). The product was dried in vacuum at 80°C. Wt
=5.8 g. Yield =95%, MP 237-239°C.
4-Hydroxypyrazolo[3,4-d]pyrimidine: A suspension of 3-aminopyrazole-4-
carboxamide hemisulfate (113 g) in formamide (325 g) was stirred and
heated to 145°C. The reaction was held at 145°C for 5 hours. The reaction
was then cooled to 30°C and the product collected and washed with
formamide (2 x 50 ml), water (2 x 150 ml) and acetone (2 x 100 ml). Wt of
crude product = 79 g. The crude product was recrystallized by dissolution in a
solution made from sodium hydroxide (25 g) in water (1,200 ml) with
treatment at 25°C with charcoal (8 g), followed by reprecipitation by the
addition of concentrated hydrochloric acid to pH 5. The product was collected
and washed with cold water (2 x 300 ml), acetone (2 x 200 ml) and dried in
vacuum at 60°C. Wt = 70 g. Yield = 80%.
brand name
Lopurin (Abbott); Lopurin (BASF); Zyloprim (Promethus).
Therapeutic Function
Xanthine oxidase inhibitor, Gout therapy
General Description
Odorless tasteless white microcrystalline powder.
Air & Water Reactions
Insoluble in water.
Reactivity Profile
Allopurinol is an aminoalcohol. Amines are chemical bases. They neutralize acids to form salts plus water. These acid-base reactions are exothermic. The amount of heat that is evolved per mole of amine in a neutralization is largely independent of the strength of the amine as a base. Amines may be incompatible with isocyanates, halogenated organics, peroxides, phenols (acidic), epoxides, anhydrides, and acid halides. Flammable gaseous hydrogen is generated by amines in combination with strong reducing agents, such as hydrides. Allopurinol darkens above 572° F, and at an indefinite high temperature, Allopurinol chars and decomposes. At 221° F, maximum stability occurs at pH 3.1- 3.4. Allopurinol decomposes in acidic and basic solutions.
Fire Hazard
Flash point data for Allopurinol are not available; however, Allopurinol is probably combustible.
Biochem/physiol Actions
Inhibitor of xanthine oxidase and de novo pyrimidine biosynthesis. A classical agent in treatment of hyperuricemia and gout.
Mechanism of action
Allopurinol, in contrast to the uricosuric drugs, reduces
serum urate levels through a competitive inhibition of
uric acid synthesis rather than by impairing renal urate
reabsorption. This action is accomplished by inhibiting
xanthine oxidase, the enzyme involved in the metabolism
of hypoxanthine and xanthine to uric acid. After
enzyme inhibition, the urinary and blood concentrations
of uric acid are greatly reduced and there is a simultaneous
increase in the excretion of the more soluble
uric acid precursors, xanthine and hypoxanthine.
Allopurinol itself is metabolized by xanthine oxidase
to form the active metabolite oxypurinol, which
tends to accumulate after chronic administration of the
parent drug.This phenomenon contributes to the therapeutic
effectiveness of allopurinol in long-term use.
Oxypurinol is probably responsible for the antigout effects
of allopurinol. Oxypurinol itself is not administered
because it is not well absorbed orally.
Pharmacokinetics
Allopurinol was synthesized in 1956 as part of a study of purine antagonists. It is well absorbed on oral administration, with peak plasma concentrations appearing within 1 hour. Decreases of uric acid can be observed within 24 to 48 hours. Excretion of allopurinol and its metabolite occurs primarily in the urine, with approximately 20% of a dose being excreted in the feces.
Clinical Use
Allopurinol is especially indicated in the treatment of
chronic tophaceous gout, since patients receiving it show
a pronounced decrease in their serum and urinary uric
acid levels. Because it does not depend on renal mechanisms
for its efficacy, allopurinol is particularly beneficial
for patients who already have developed renal uric acid
stones, patients with excessively high urate excretion
(e.g., above 1,200 mg in 24 hours), patients with a variety
of blood disorders (e.g., leukemia, polycythemia vera),
patients with excessive tophus deposition, and patients
who fail to respond well to the uricosuric drugs.
Allopurinol also inhibits reperfusion injury. This injury
occurs when organs that either have been transplanted
or have had their usual blood perfusion blocked
are reperfused with blood or an appropriate buffer solution.
The cause of this injury is local formation of free
radicals, such as the superoxide anion, the hydroxyl free
radical, or peroxynitrite. These substances are strong
oxidants and are quite damaging to tissues.
Side effects
Common toxicities associated with allopurinol administration include a variety of skin rashes, gastrointestinal upset, hepatotoxicity, and fever. These reactions are often sufficiently severe to dictate termination of drug therapy. It is advised that therapy not be initiated during an acute attack of gouty arthritis. As with the uricosuric drugs, therapy with allopurinol should be accompanied both by a sufficient increase in fluid intake to ensure water diuresis and by alkalinization of the urine. Prophylactic use of colchicine also helps to prevent acute attacks of gout that may be brought on during the initial period of allopurinol ingestion.
Safety Profile
Human poison by ingestion. Poison experimentally by intraperitoneal and subcutaneous routes. An experimental teratogen. Human systemic effects by ingestion: blood leukopenia, dermatitis, jaundice, muscle weakness, thrombocytopenia. When heated to decomposition it emits toxic fumes of NOx. An FDA proprietary drug used as a xanthine oxidase inhibitor.
Veterinary Drugs and Treatments
The principle veterinary uses for allopurinol are for the prophylactic
treatment of recurrent uric acid uroliths and hyperuricosuric calcium
oxalate uroliths in small animals. It has also been used in an
attempt to treat gout in pet birds and reptiles.
Allopurinol has been recommended as an alternative treatment
for canine Leishmaniasis. Although
it appears to have clinical efficacy,
it does not apparently clear the parasite in most dogs at usual dosages.
Allopurinol may also be useful for American Trypanosomiasis.
Drug interactions
Potentially hazardous interactions with other drugs
ACE inhibitors: increased risk of toxicity with
captopril.
Antivirals: concentration of didanosine increased -
avoid.
Ciclosporin: isolated reports of raised ciclosporin
levels (risk of nephrotoxicity).
Cytotoxics: effects of azathioprine and
mercaptopurine enhanced with increased toxicity;
avoid with capecitabine and ideally azathioprine.
Metabolism
Allopurinol is rapidly metabolized via oxidation and the formation of numerous ribonucleoside derivatives. The major oxidation metabolite, alloxanthine or oxypurinol, has a much longer half-life (18–30 hours versus 2–3 hours) than the parent drug and is an effective, although less potent, inhibitor of xanthine oxidase. The longer plasma half-life of alloxanthine results in an accumulation in the body during chronic administration, thus contributing significantly to the overall therapeutic effects of allopurinol.
Precautions
Since allopurinol is metabolized by the hepatic microsomaldrug-metabolizing enzymes, coadministration ofdrugs also metabolized by this system should be donewith caution. Because allopurinol inhibits the oxidationof mercaptopurine and azathioprine, their individualadministered doses must be decreased by as much as75% when they are given together with allopurinol.Allopurinol may also increase the toxicity of other cytotoxicdrugs (e.g., vidarabine). The actions of allopurinolare not antagonized by the coadministration of salicylates.
Allopurinol Preparation Products And Raw materials
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| HAINAN POLY PHARMCEUTICAL CO. LTD | +86-0571-89385087 +8613616530509 | ff@hnpoly.com | China | 118 | 58 |
| Hebei Mojin Biotechnology Co., Ltd | +8613288715578 | sales@hbmojin.com | China | 12453 | 58 |
| Sinoway Industrial co., ltd. | 0592-5800732; +8613806035118 | xie@china-sinoway.com | China | 992 | 58 |
| Shaanxi TNJONE Pharmaceutical Co., Ltd | +86-13474506593 +86-13474506593 | sarah@tnjone.com | China | 775 | 58 |
| Capot Chemical Co.,Ltd. | 571-85586718 +8613336195806 | sales@capotchem.com | China | 29797 | 60 |
| Shanghai Daken Advanced Materials Co.,Ltd | +86-371-66670886 | info@dakenam.com | China | 15371 | 58 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21695 | 55 |
| Shanghai Time Chemicals CO., Ltd. | +86-021-57951555 +8617317452075 | jack.li@time-chemicals.com | China | 1807 | 55 |
| ATK CHEMICAL COMPANY LIMITED | +undefined-21-51877795 | ivan@atkchemical.com | China | 32480 | 60 |
| career henan chemical co | +86-0371-86658258 | sales@coreychem.com | China | 29914 | 58 |
Related articles
- Allopurinol: the treatment of elevated uric acid levels
- Allopurinol, a key gout treatment, inhibits uric acid, explored for cardiovascular benefits, with optimized safety in synthesi....
- Jan 2,2024
- Allopurinol: mechanism of action and clinical efficacy
- Allopurinol is a medication commonly used to treat gout. However, recent studies have shown that Allopurinol may be beneficial....
- Jun 20,2023
- Allopurinol - Reaction / Application on synthetic works
- Allopurinol is xanthine oxidase-activated prodrug of thymidine phosphorylase inhibitor. It is also an important organic interm....
- Nov 19,2019
View Lastest Price from Allopurinol manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2024-04-10 | Allopurinol
315-30-0
|
US $0.00 / Kg/Bag | 2Kg/Bag | 99% up / USP/BP/EP | 20 tons | Sinoway Industrial co., ltd. | |
 |
2024-04-05 | Allopurinol
315-30-0
|
US $0.00 / kg | 1kg | 99% | 20tons | Shaanxi TNJONE Pharmaceutical Co., Ltd | |
 |
2024-03-16 | Allopurinol
315-30-0
|
US $0.00 / KG | 100g | 98%+ | 100kg | WUHAN CIRCLE POWDER TECHNOLOGY CO.,LTD |
-

- Allopurinol
315-30-0
- US $0.00 / Kg/Bag
- 99% up / USP/BP/EP
- Sinoway Industrial co., ltd.
-

- Allopurinol
315-30-0
- US $0.00 / kg
- 99%
- Shaanxi TNJONE Pharmaceutical Co., Ltd
-

- Allopurinol
315-30-0
- US $0.00 / KG
- 98%+
- WUHAN CIRCLE POWDER TECHNOLOGY CO.,LTD