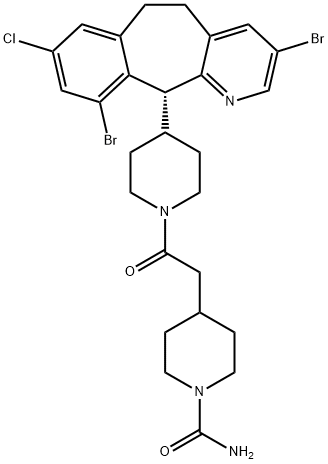
LONAFARNIB
- CAS No.
- 193275-84-2
- Chemical Name:
- LONAFARNIB
- Synonyms
- Sarasar;Sch66336;Sch 66336;EOS-61911;LONAFARNIB;LonafarMib;SCH-066336;Unii-iow153004f;Lonafarnib [usan];Valine Impurity 114
- CBNumber:
- CB31011743
- Molecular Formula:
- C27H31Br2ClN4O2
- Molecular Weight:
- 638.82
- MDL Number:
- MFCD06795138
- MOL File:
- 193275-84-2.mol
- MSDS File:
- SDS
| Melting point | 214.5-215.9° (monohydrate); mp 222-223° |
|---|---|
| alpha | D25 = +49.1° (c = 0.21 in methanol) |
| Boiling point | 710.4±70.0 °C(Predicted) |
| Density | 1.536 |
| storage temp. | -20°C |
| solubility | Chloroform (Slightly), Methanol (Slightly) |
| form | powder |
| pka | 15.76±0.40(Predicted) |
| color | white to beige |
| NCI Dictionary of Cancer Terms | lonafarnib; SCH 66336 |
| FDA UNII | IOW153004F |
| NCI Drug Dictionary | lonafarnib |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|---|---|
| Signal word | Warning |
| Hazard statements | H315-H319-H335 |
| Precautionary statements | P261-P264-P271-P280-P302+P352-P305+P351+P338 |
LONAFARNIB price More Price(36)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | SML1457 | Lonafarnib ≥98% (HPLC) | 193275-84-2 | 5MG | $134 | 2024-03-01 | Buy |
| Sigma-Aldrich | SML1457 | Lonafarnib ≥98% (HPLC) | 193275-84-2 | 25MG | $462 | 2024-03-01 | Buy |
| Cayman Chemical | 11746 | Lonafarnib ≥98% | 193275-84-2 | 1mg | $33 | 2024-03-01 | Buy |
| Cayman Chemical | 11746 | Lonafarnib ≥98% | 193275-84-2 | 5mg | $112 | 2024-03-01 | Buy |
| Cayman Chemical | 11746 | Lonafarnib ≥98% | 193275-84-2 | 10mg | $143 | 2024-03-01 | Buy |
LONAFARNIB Chemical Properties,Uses,Production
Uses
Lonafarnib is an orally bioavailable tricyclic inhibitor of farnesyl protein transferase. It inhibits Rheb farnesylation and mTOR signaling and enhances taxane and tamoxifen antitumor activity. Studies show that it induces CCAAT/enhancer-binding protein homologous protein-dependent expression of death receptor 5, leading to induction of apoptosis in human cancer cells
Uses
Chemotherapeutic (farnesyl transfer ase inhibitor).
Definition
ChEBI: A 4-{2-[4-(3,10-dibromo-8-chloro-6,11-dihydro-5H-benzo[5,6]cyclohepta[1,2-b]pyridin-11-yl)piperidin-1-yl]-2-oxoethyl}piperidine-1-carboxamide that has R configuration. It is used as oral farnesyltransferase nhibitor.
General Description
Lonafarnib (SCH66336) is a farnesyl transferase inhibitor (FTI). K- and N-Ras are substrates of farnesyl transferase.
Biological Activity
lonafarnib (sch66336, sarasar) is an potent, selective, orally, bioavailable tricyclic nonpeptidyl nonsulfhydry inhibitor of farnesyltransferase (ftase).[1] it is a small molecular with the formula of c27h31br2cln4o2 and molecular weight of 638.82. farnesylated ras proteins was found to regulate signal transduction pathways which drive cell proliferation, growth and survival and be required for its membrane localization.[1, 2] lonafarnib inhibits the post-translational farnesylcation of ras proteins, therefore blocking translocation of ras to the plasma membrane.[3][1] eric w, malcolm j. m, kim n. c, d. scott e, et al. a multinomial phase ii study of lonafarnib (sch 66336) in patients with refractory urothelial cancer. urologic oncology: seminars and original investigations. 2005, 23. 143-149.[2] gongjie l, stacey a. t, cindy h. m, yunsheng h, w. robert b, et al. continuous and intermittent dosing of lonafarnib potentiates the therapeutic efficacy of docetaxel on preclinical human prostate cancer models. int. j. cancer. 2009, 125. 2711–2720.[3] vasiliki a. n, alexander j. s, keith t. f, hensin t, et al. melanoma: new insights and new therapies. j invest dermatol. 2012, 132. 854–863.
Biochem/physiol Actions
Lonafarnib prevents the post-translational lipid modification of H-Ras and other farnesylated proteins. Lonafarnib treatment results in microtubule bundling, increased microtubule acetylation and stabilization and suppression of microtubule dynamics.
Mechanism of action
Lonafarnib is a protein farnesyltransferase inhibitor (FTI) that reversibly binds to the farnesyltransferase CAAX binding site9, thereby inhibiting progerin farnesylation and subsequent intercalation into the nuclear membrane.
Side effects
- vomiting
- diarrhea
- nausea
- stomach pain
- constipation
- gas
- decreased appetite
- decreased weight
target
FT
storage
Store at -20°C
LONAFARNIB Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| career henan chemical co | +86-0371-86658258 | sales@coreychem.com | China | 29914 | 58 |
| Biochempartner | 0086-13720134139 | candy@biochempartner.com | CHINA | 967 | 58 |
| Hubei Jusheng Technology Co.,Ltd. | 18871490254 | linda@hubeijusheng.com | CHINA | 28180 | 58 |
| BOC Sciences | +1-631-485-4226 | inquiry@bocsci.com | United States | 19553 | 58 |
| Chongqing Chemdad Co., Ltd | +86-023-61398051 +8613650506873 | sales@chemdad.com | China | 39916 | 58 |
| CONIER CHEM AND PHARMA LIMITED | +8618523575427 | sales@conier.com | China | 47465 | 58 |
| HANGZHOU CLAP TECHNOLOGY CO.,LTD | 86-571-88216897,88216896 13588875226 | sales@hzclap.com | CHINA | 6313 | 58 |
| Dideu Industries Group Limited | +86-29-89586680 +86-15129568250 | 1026@dideu.com | China | 29271 | 58 |
| Hefei Hirisun Pharmatech Co., Ltd | +8615056975894 | shawn@hirisunpharm.com | CHINA | 9923 | 58 |
| Zhejiang J&C Biological Technology Co.,Limited | +1-2135480471 +1-2135480471 | sales@sarms4muscle.com | China | 10523 | 58 |
Related articles
- How is Lonafarnib synthesised?
- Lonafarnib was synthesised starting from the bromination of chloroanthranilic acid and using the lonafarnib precursor tricycle....
- Dec 27,2023
View Lastest Price from LONAFARNIB manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2019-07-06 | LONAFARNIB
193275-84-2
|
US $2.00 / kg | 1kg | 99% | 100kg | Career Henan Chemical Co |
-

- LONAFARNIB
193275-84-2
- US $2.00 / kg
- 99%
- Career Henan Chemical Co