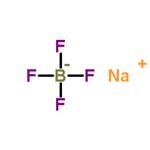
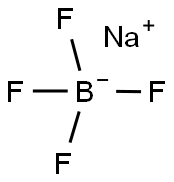Sodium tetrafluoroborate
- CAS No.
- 13755-29-8
- Chemical Name:
- Sodium tetrafluoroborate
- Synonyms
- SODIUM FLUOROBORATE;SODIUM FLUOBORATE;apretonr;SODIUM BOROFLUORIDE;Sodium boron f;Sodium tetrafL;Sodium fluborate;Sodium fluoborater;Sodium fluoroberate;sodium tetrafluoride
- CBNumber:
- CB4775517
- Molecular Formula:
- BF4Na
- Molecular Weight:
- 109.79
- MDL Number:
- MFCD00003515
- MOL File:
- 13755-29-8.mol
- MSDS File:
- SDS
| Melting point | 384 °C |
|---|---|
| Density | 2.47 g/mL at 25 °C(lit.) |
| storage temp. | Inert atmosphere,Room Temperature |
| solubility | water: soluble(lit.) |
| form | Powder, Crystals or Needles |
| color | White to yellow to beige to brown |
| Specific Gravity | 2.47 |
| Water Solubility | 108 g/100 mL (26 ºC) |
| Sensitive | Hygroscopic |
| Merck | 14,8617 |
| Exposure limits |
ACGIH: TWA 2.5 mg/m3 NIOSH: IDLH 250 mg/m3; TWA 2.5 mg/m3 |
| Stability | Moisture sensitive; do not store in glass (though the completely dry material does not etch glass). Incompatible with strong oxidizing agents. |
| CAS DataBase Reference | 13755-29-8(CAS DataBase Reference) |
| FDA UNII | CFC805A5WR |
| EPA Substance Registry System | Borate(1-), tetrafluoro-, sodium (13755-29-8) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS05 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H314 | |||||||||
| Precautionary statements | P260-P280-P303+P361+P353-P304+P340+P310-P305+P351+P338-P363 | |||||||||
| Hazard Codes | C,Xi | |||||||||
| Risk Statements | 34 | |||||||||
| Safety Statements | 45-36/37/39-25-26 | |||||||||
| RIDADR | 3260 | |||||||||
| WGK Germany | 1 | |||||||||
| RTECS | ED2975000 | |||||||||
| F | 21 | |||||||||
| Hazard Note | Irritant | |||||||||
| TSCA | Yes | |||||||||
| HS Code | 2826 90 80 | |||||||||
| NFPA 704 |
|
Sodium tetrafluoroborate price More Price(36)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | 8.43877 | Sodium tetrafluoroborate for synthesis | 13755-29-8 | 250g | $55.8 | 2024-03-01 | Buy |
| Sigma-Aldrich | 202215 | Sodium tetrafluoroborate 98% | 13755-29-8 | 25g | $59.9 | 2024-03-01 | Buy |
| Sigma-Aldrich | 8.43877 | Sodium tetrafluoroborate for synthesis | 13755-29-8 | 1kg | $158 | 2024-03-01 | Buy |
| Sigma-Aldrich | 202215 | Sodium tetrafluoroborate 98% | 13755-29-8 | 10kg | $292.8 | 2024-03-01 | Buy |
| TCI Chemical | O0494 | Sodium Tetrafluoroborate >98.0%(T) | 13755-29-8 | 25g | $34 | 2024-03-01 | Buy |
Sodium tetrafluoroborate Chemical Properties,Uses,Production
Description
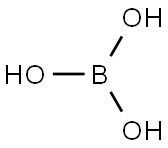
Sodium tetrafluoroborate is an organic compound that has the molecular formula NaBF4. The compound is a salt that forms white or colorless water-soluble rhombic crystals. It is less soluble in organic solvents and readily soluble in water.
Preparation
Sodium tetrafluoroborate can be prepared by reacting tetrafluoroboric with sodium carbonate or hydroxide.
NaOH + HBF4 → NaBF4 + H2O
Na2CO3 + 2 HBF4 → 2 NaBF4 + H2O + CO2
Applications
Sodium tetrafluoroborate can be used as a catalyst for the synthesis of bis(indolyl)methanes via electrophilic substitution reaction of indoles with ketones and aldehydes. In the synthesis of ionic liquids such as trihexyl(tetradecyl)phosphonium tetrafluoroborat and 1-butyl-3-methylimidazolium tetrafluoroborate.
In the laboratory, sodium tetrafluoroborate may be used for the synthesis of boron fluoride. It may also be used in the synthesis of fluoroheterocyclic and fluoro-nucleic acids. The compound is also used in metal finishing, fluxes, and plating circuits.
Chemical Properties
white crystals or powder
Uses
Sodium tetrafluoroborate may be used for the laboratory synthesis of boron fluoride. It may be used for the synthesis of fluoro-nucleic acids and fluoroheterocyclic compounds.
Uses
Sodium Tetrafluoroborate is used in various metal finishing and plating circuits.
Uses
Fluorinating agent, see Lawton, Levy, J. Am. Chem. Soc. 77, 6083 (1955).
General Description
Sodium tetrafluoroborate (NaBF4) is colorless sodium salt and its crystals belong to the rhombic crystal system. It can be synthesized by reacting tetrafluoroboric acid with sodium carbonate or hydroxide.
Flammability and Explosibility
Not classified
Safety Profile
Moderately toxic by subcutaneous route. When heated to decomposition it emits toxic fumes of Fand Na2O.
Purification Methods
Crystallise the fluoroborate from hot water (50mL/g) by cooling to 0o. Alternatively, free it from insoluble material by dissolving it in a minimum amount of water; then fluoride ions are removed by adding concentrated lanthanum nitrate in excess. After removing lanthanum fluoride by centrifugation, the supernatant is passed through a cation-exchange column (Dowex 50, Na+-form) to remove any remaining lanthanum [Anbar & Guttman J Phys Chem 64 1896 1960]. It has also been recrystallised from anhydrous MeOH and dried in a vacuum at 70o for 16hours. Keep it dry as it is hygrosopic. [Delville et al. J Am Chem Soc 109 7293 1987.]
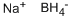
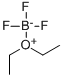
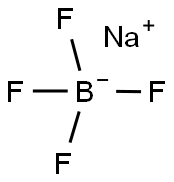


Sodium tetrafluoroborate Preparation Products And Raw materials
Raw materials
1of3
Preparation Products
1of3
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Henan Fengda Chemical Co., Ltd | +86-371-86557731 +86-13613820652 | info@fdachem.com | China | 7377 | 58 |
| Capot Chemical Co.,Ltd. | 571-85586718 +8613336195806 | sales@capotchem.com | China | 29797 | 60 |
| Shanghai Daken Advanced Materials Co.,Ltd | +86-371-66670886 | info@dakenam.com | China | 15371 | 58 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21695 | 55 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418679 +86-18949832763 | info@tnjchem.com | China | 2989 | 55 |
| Shanghai Zheyan Biotech Co., Ltd. | 18017610038 | zheyansh@163.com | CHINA | 3620 | 58 |
| career henan chemical co | +86-0371-86658258 | sales@coreychem.com | China | 29914 | 58 |
| Hubei Jusheng Technology Co.,Ltd. | 18871490254 | linda@hubeijusheng.com | CHINA | 28180 | 58 |
| Hebei Guanlang Biotechnology Co., Ltd. | +86-19930503282 | alice@crovellbio.com | China | 8822 | 58 |
| Xiamen AmoyChem Co., Ltd | +86-592-6051114 +8618959220845 | sales@amoychem.com | China | 6387 | 58 |
View Lastest Price from Sodium tetrafluoroborate manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2024-01-23 | Sodiumtetrafluoroborate
13755-29-8
|
US $9.00-0.90 / KG | 1KG | 99% | g-kg-tons, free sample is available | Henan Fengda Chemical Co., Ltd | |
 |
2021-09-29 | Sodium tetrafluoroborate
13755-29-8
|
US $10.00 / KG | 1KG | 99% | 5tons | Hebei Guanlang Biotechnology Co., Ltd. | |
 |
2021-07-13 | Sodium tetrafluoroborate
13755-29-8
|
US $15.00-10.00 / KG | 1KG | 99%+ HPLC | Monthly supply of 1 ton | Zhuozhou Wenxi import and Export Co., Ltd |
-

- Sodiumtetrafluoroborate
13755-29-8
- US $9.00-0.90 / KG
- 99%
- Henan Fengda Chemical Co., Ltd
-

- Sodium tetrafluoroborate
13755-29-8
- US $10.00 / KG
- 99%
- Hebei Guanlang Biotechnology Co., Ltd.
-

- Sodium tetrafluoroborate
13755-29-8
- US $15.00-10.00 / KG
- 99%+ HPLC
- Zhuozhou Wenxi import and Export Co., Ltd
13755-29-8(Sodium tetrafluoroborate)Related Search:
1of4