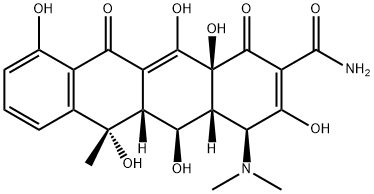
Oxytetracycline
- CAS No.
- 79-57-2
- Chemical Name:
- Oxytetracycline
- Synonyms
- OXYTETRACYCLINE BASE;Terramycin;oxytetracyclin;oxy-kesso-tetra;Oxytetracycline CRS;4-(dimethylamino)-1,4,4a,5,5a,6,11,12a-octahydro-3,5,6,10,12,12a-hexahydroxy-6-methyl-1,11-dioxo-, 2-Naphthacenecarboxamide;la200;Oxydon;oxypam;tetran
- CBNumber:
- CB6429340
- Molecular Formula:
- C22H24N2O9
- Molecular Weight:
- 460.43
- MDL Number:
- MFCD00003700
- MOL File:
- 79-57-2.mol
| Melting point | 183 °C |
|---|---|
| alpha | -223 º (c=1 0.03N HCl) |
| Boiling point | 565.29°C (rough estimate) |
| Density | 1.6340 |
| refractive index | 1.6500 (estimate) |
| storage temp. | Keep in dark place,Inert atmosphere,Room temperature |
| solubility | Sparingly soluble in aqueous solution at 0.6 mg/mL |
| form | Crystalline Powder |
| pka | pKa 3.27/7.32/9.11(H2O,t =25,I<0.01)(Approximate) |
| color | Beige to light yellow |
| Water Solubility | 0.2 g/L |
| InChIKey | IWVCMVBTMGNXQD-PXOLEDIWSA-N |
| LogP | -0.900 |
| FDA 21 CFR | 556.500; 558.450; 558.4 |
| CAS DataBase Reference | 79-57-2(CAS DataBase Reference) |
| FDA UNII | SLF0D9077S |
| Proposition 65 List | Oxytetracycline (internal use) |
| ATC code | D06AA03,G01AA07,J01AA06,J01AA56,S01AA04 |
| Pesticides Freedom of Information Act (FOIA) | Oxytetracycline |
| EPA Substance Registry System | Oxytetracycline (79-57-2) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS08,GHS09 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H361d-H411 | |||||||||
| Precautionary statements | P201-P202-P273-P280-P308+P313-P391 | |||||||||
| Hazard Codes | Xn,Xi | |||||||||
| Risk Statements | 63-36-36/37/38 | |||||||||
| Safety Statements | 22-24/25-36/37/39-26-36 | |||||||||
| WGK Germany | 2 | |||||||||
| RTECS | QI7875000 | |||||||||
| TSCA | Yes | |||||||||
| HS Code | 30029090 | |||||||||
| Toxicity | LD50 oral in rat: 4800mg/kg | |||||||||
| NFPA 704 |
|
Oxytetracycline price More Price(20)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | BP782 | Oxytetracycline British Pharmacopoeia (BP) Reference Standard | 79-57-2 | 100MG | $213 | 2024-03-01 | Buy |
| Sigma-Aldrich | 51239 | Oxytetra Selective Supplement suitableformicrobiology | 79-57-2 | 5vials | $73.3 | 2024-03-01 | Buy |
| Alfa Aesar | J62427 | Oxytetracycline, 96% | 79-57-2 | 10g | $59.6 | 2024-03-01 | Buy |
| Alfa Aesar | J62427 | Oxytetracycline, 96% | 79-57-2 | 50g | $217 | 2024-03-01 | Buy |
| Cayman Chemical | 18076 | Oxytetracycline ≥95% | 79-57-2 | 10g | $32 | 2024-03-01 | Buy |
Oxytetracycline Chemical Properties,Uses,Production
description
Oxytetracycline (PC Code 006304) is a broad-spectrum antibiotic produced from the actinomycete Streptomyces rimosus. Two related compounds, hydroxytetracycline monohydrochloride (PC Code 006308) and oxytetracycline calcium (PC Code 006321), are registered as pesticides, for use in preventing the growth of or killing bacteria, fungi and mycoplasma-like organisms; there is no active product for PC Code 006304. Oxytetracycline is also approved by the Food and Drug Administration (FDA) for use as a human and animal drug to treat a variety of bacterial infections. Specifically, oxytetracycline is used to treat infections caused by chlamydia, mycoplasma organisms, propionebacterium acnes, haemophilus influenzae, and rickettsiae. It acts by inhibiting protein synthesis by binding to the 30S ribosomal subunit of the bacteria. In conjunction with the FDA-approved animal drug uses, food-additive tolerances are established for residues of tetracyclines in commodities of beef cattle, dairy cattle, calves, swine, sheep, chickens, turkeys, finfish, and lobster (21 CFR §556.500).
Physical and Chemical Properties
Isolated from the culture medium of Streptomyces fission; appears as a gray-yellow to yellow crystalline powder. Commonly in a form of hydrochloride; appears as yellow crystal, easily soluble in water, stable under acidic conditions but unstable in alkaline environment.
Its oxytetracycline appears as light yellow to dark yellow crystalline powder, insoluble in water. Adjusting the pH to over 8 or below 2 yields oxytetracycline sodium salt or hydrochloride, this is stable and easily soluble in water. The 10% aqueous solution should have a pH of 2.3 to 2.9.
Pharmacology
1. Pharmacodynamics; The drug is a tetracycline antibacterial drug with a broad spectrum of anti-pathogenic microorganisms; appears as a rapid bacteriostatic agent; exhibits bactericidal effect to some bacteria species at high concentration; have a slightly high potency than tetracycline for the treatment of intestinal infections (including amoebic dysentery).
- Mechanism of action; The drug can specifically bind to the A site of the 30s subunit of the ribosomes in pathogens, preventing the binding of aminoacyl-tRNA to this position, thereby inhibiting the growth of the peptide chain and affecting the protein synthesis of bacteria or other pathogenic microorganisms .
- Antibacterial spectrum The drug has strong antibacterial activity on Staphylococcus aureus, pneumococcus, Streptococcus pyogenes, Neisseria gonorrhoeae, meningococcal, Escherichia coli, Aerobacter aerogenes, Shigella, Yersinia and Listeria monocytogenes. It also has strong effect on rickettsia, mycoplasma, chlamydia, spirochetes, amoebae and some plasmodium and actinomyces.
Adverse reactions
- Bone; The medicine can be deposited in the teeth and bones, resulting in varying degrees of discoloration yellow teeth, enamel dysplasia and dental caries, and can cause bone dysplasia.
- Oral administration of the gastrointestinal tract can cause nausea, vomiting, abdominal discomfort, abdominal distension, diarrhea and other gastrointestinal reactions; occasional esophagitis and esophageal ulcer reported in patients with bed rest immediately after taking medicine.
- Liver; in rare cases, serum bilirubin, alkaline phosphatase and aminotransferase increase. Long-term medication can cause liver damage, usually leading to hepatic steatosis. People of liver and renal insufficiency, late pregnancy and women receiving high-dose intravenous administration are more likely to suffer. Occasionally also cause both pancreatic inflammation together with liver toxicity.
- Renal; in rare cases, blood urea nitrogen content increase. Patients with significant renal impairment may have aggravating azotemia, hyperphosphatemia and acidosis.
- Allergic reaction cases are less than penicillin. It can cause drug fever or rash, the latter can exhibit rash, urticaria, erythema multiforme, eczema-like erythema; can also cause photosensitive dermatitis. In addition, anaphylactic shock, asthma, purpura, etc. are also occasionally reported.
- Long-term use in blood can cause abnormal lymphocytes, granulocytic granules, hemolytic anemia, thrombocytopenia and neutropenia.
- Superinfection; Long-term medication can induce drug-resistant Staphylococcus aureus, Gram-negative bacteria and fungi, which lead to the infections in digestive tract, respiratory and urinary tract infections with sepsis occurring in severe cases.
- Dysbacteriosis; long-term medication can also reduce the body's normal bacteria population, resulting in vitamin deficiency, fungal breeding, dry mouth, sore throat, angular cheilitis, glossitis, dark brown tongue or discoloration.
- Central nervous system; it has been reported of the cases of benign increased intracranial pressure, manifested as headache, vomiting, optic disc edema and so on.
Drugs interactions
- Quinolones: not suitable for combination usage with reduced efficacy and increased side effects.
- Cephalosporins: oxytetracycline can reduce the antibacterial effect of cephalosporins.
- Chlortetracycline, kanamycin, olaquindox, enramycin, North rifampin, furinomycin, flavomycin: incompatibility; oxytetracycline are incompatible with the latter combination.
- Tylosin and other macrolides: combination yields synergistic effect.
- Polymyxin: combination with oxytetracycline enhances the absorption of the latter, leading to synergistic effect.
- Penicillins: Antagonistic effect.
- Metronidazole: combination can reduce the effect of metronidazole, reducing the efficacy.
- Sodium bicarbonate: increase the pH of oxytetracycline; reduce the dissociation degree and absorption rate.
- Magnesium sulfate: oxytetracycline can form chelate with magnesium ions; reduce its own absorption and curative effect.
- TMP: significant synergistic effect.
- Vitamin C: The latter can inactivate oxytetracycline; oxytetracycline in turn causes faster excretion of vitamin C in the urine. Not suitable.
- Drugs and feeds containing calcium, magnesium, aluminum and other cationic; prevent the absorption of oxytetracycline and reduce its efficacy.
- Sodium sulfate: In order to avoid the effect of metal cations in feed; adding appropriate amount of sodium sulfate in the feed can improve the absorption of oxytetracycline and improve the curative effect.
- Diuretics: Combination can increase blood urea nitrogen, leading to increased renal toxicity.
- Berberine: combination can enhance antibacterial effect.
- Vitamin B complex: reduce the effect of oxytetracycline.
- Bacitracin: incompatibility should not be combined.
- Food, milk: oxytetracycline, when used with food, milk, the blood concentration is only 20% of that in the cases of fasting drug administration with remarkably compromised antibacterial effect.
- Trace elements: block the absorption of oxytetracycline, reduce the effect.
Description
Oxytetracycline also is one of the classic tetracyclines. It is produced by fermentation of Streptomyces rimosis and other soil microorganisms. The most hydrophilic tetracycline on the market, it has largely now been replaced by its semisynthetic descendants. It is primarily used today for IM injections.
Chemical Properties
beige to light yellow crystalline powder
Originator
Terramycin,Pfizer,US,1950
Uses
Oxytetracycline, which has seen significant usage in plant disease control, does not contain the Cl atom but has an additional -OH group at the C5 position on the tetracycline molecule (4). Tetracyclines are widely used therapeutic agents in clinical medicine [second to penicillins in total tons used each year (5)] for bacterial respiratory, periodontal, and urogenital tract diseases (6). Oxytetracycline is also used as a feed amendment for growth promotion and as a therapeutic agent for curing diseases of agricultural animals including fish.
Uses
Oxytetracycline (terramycin) is a linear, tetracyclic broad spectrum antibiotic originally isolated from Streptomyces rimosus in 1949. Like all tetracyclines, oxytetracycline shows broad spectrum antibacterial and antiprotozoan activity and acts by binding to the 30S and 50S ribosomal sub-units, blocking protein synthesis. Oxytetracycline, although less coloured than other tetracyclines, is still a pigment and is thus sensitive to environmental and storage conditions. Commercial oxytetracycline may contain significant levels of degradation products.
Uses
Oxytetracycline(Terramycin) was the second of the broad-spectrum tetracycline group of antibiotics to be discovered. Oxytetracycline works by interfering with the ability of bacteria to produce proteins that are essential to them. Without these proteins t
Definition
ChEBI: A tetracycline used for treatment of infections caused by a variety of Gram positive and Gram negative microorganisms including Mycoplasma pneumoniae, Pasteurella pestis, Escherichia coli, Haemophilus influenzae /ital> (respiratory infections), and Diplococcus pneumoniae.
Manufacturing Process
The pH was adjusted to 7.0 with sodium hydroxide and calcium carbonate was added at the rate of 1 g/l.
500 ml portions of the above medium were added to Fernbach flasks which
were then sterilized at 121°C for 30 minutes. Upon cooling, the flasks were inoculated with a suspension of the growth of s. rimosus obtained from the surface of beef lactose agar slants, and the flasks were shaken for 4 days at 28°C on a rotary shaker having a displacement of 2" at an rpm of 200. At the end of this period the broth was found to contain 640 C.D.U/ml and 400 chloramphenicol units/ml. The mycelium was separated from the broth by filtration and the latter was adjusted to pH 9.0. The antibiotic was extracted from the broth with n-butanol, and when the ultraviolet absorption spectrum was observed on the butanol solution of the antibiotic, peaks in the absorption curve were found at 385 and 270 millimicrons.
brand name
Terramycin (Pfizer).
Therapeutic Function
Antibiotic
Antimicrobial activity
It is slightly less active than other tetracyclines against most common pathogenic bacteria.
Pharmaceutical Applications
A fermentation product of certain strains of Streptomyces rimosus, supplied as the dihydrate or hydrochloride for oral or parenteral administration.
Pharmacokinetics
Oral absorption: c.60%
Cmax 500 mg oral: 3–4 mg/L after 2–4 h
Plasma half-life: c.9 h
Volume of distribution: c.1.8 L/kg
Plasma protein binding: 20–35%
Oxytetracycline is moderately well absorbed from the upper
gastrointestinal tract. Food decreases plasma levels by approximately
50%. Although widely distributed in the tissues, it achieves lower concentrations than related agents such as minocycline.
Sputum concentrations of 1 mg/L have been recorded
on a daily dosage of 2 g. Approximately 60% is excreted in the
urine and the half-life is prolonged in renal insufficiency.
Clinical Use
It offers no unique therapeutic advantages, although it is one of the cheaper preparations.
Side effects
Gastrointestinal intolerance is responsible for most side effects, and tends to be more severe than with other tetracyclines. Esophageal irritation may result from the local effects of the swallowed drug. Potentially serious adverse reactions have included neuromuscular paralysis following intravenous administration to patients with myasthenia gravis. Thrombocytopenic purpura and lupus erythematosus syndrome have been reported, although a direct role for the drug in the latter remains uncertain. Apart from the effect on nitrogen balance common to many tetracyclines, a metabolic effect on glucose homeostasis has been noted in type 1 diabetes mellitus. Allergic contact sensitivity reactions have also been reported.
Synthesis
Oxytetracycline, 4-dimethylamino-1,4,4a,5,5a,6,11,12a-octahydro- 3,6,10,12,12a-hexahydroxy-6-methyl-1,11-dioxo-2-naphthacencarboxamide (32.3.2), is synthesized biosynthetically as a result of the activity of actinomycete S. rimosus.
Veterinary Drugs and Treatments
Oxytetracycline products are approved for use in dogs and cats (no known products are being marketed, however), calves, non-lactating dairy cattle, beef cattle, swine, fish, and poultry. For more information, refer to the Doses section, below.
Drug interactions
Potentially hazardous interactions with other drugs
Anticoagulants: possibly enhanced anticoagulant
effect of coumarins and phenindione.
Oestrogens: possibly reduced contraceptive effects of
oestrogens (risk probably small).
Retinoids: possible increased risk of benign
intracranial hypertension with tetracyclines and
retinoids - avoid.
Metabolism
Metabolism is negligible. The tetracyclines are excreted in the urine and in the faeces. Renal clearance is by glomerular filtration. Up to 60% of an intravenous dose of tetracycline, and up to 55% of an oral dose, is eliminated unchanged in the urine. The tetracyclines are excreted in the bile, where concentrations 5-25 times those in plasma can occur. There is some enterohepatic reabsorption and considerable quantities occur in the faeces after oral doses.
Purification Methods
Terramycin crystallises (as dihydrate) from water or aqueous EtOH and is soluble in MeOH, EtOH, Me2CO and H2O (0.25mg/mL at 25o ) but insoluble in Et2O and pet ether. It is amphoteric, and an aqueous solution has pH 2.0-5.0. It has max at 247, 275 and 353nm in 0.1 M phosphate (pH 4.5). [Finlay et al. Science 111 85 1951.] The hydrochloride, [2058-46-0] M 496.9 [Beilstein 14 IV 2630], crystallises from MeOH in needles and from H2O at 50o it forms plates. Terramycin has also been purified via the hydrochloride by dissolving it in H2O, adjusting to pH 6, and the solid is filtered off after 1hour. The crystals of the dihydrate are dried to constant weight in vacuum/CaCl2/25o. Drying at 60o in vacuo gives the anhydrous base m 184.5-185.5o (sintering at 180o). The dihydrate has m 181-182o. Its optical rotation in MeOH decreases from [] D 25 +26o (c 0.5%) to [] D 25 +11.3o after standing for 16hours. It forms a sodium salt and a CaCl2 complex. [Regna et al. J Am Chem Soc 73 4211 1951, Beilstein 14 IV 2633.]
Toxicity evaluation
Tetracycline is a potent inhibitor of bacterial protein biosynthesis, with less activity on mammalian cells. It binds to the 30S and 50S bacterial ribosomal subunits, and it inhibits the binding of aminoacyl–tRNA and the termination factors RF1 and RF2 to the A site of bacterial ribosomes (21). Acute oral LD50 for mice >7 g/kg; for rats >10 g/kg, acute intravenous 100 ~ 200 mg/kg. Tlm for black bass: 250 ppm (24 h).
Oxytetracycline Preparation Products And Raw materials
Raw materials
1of2
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Hebei Yime New Material Technology Co., Ltd. | +86-66697723 +86-17703311139 | admin@china-yime.com | China | 563 | 58 |
| Hebei Guanlang Biotechnology Co,.LTD | +8619930503252 | daisy@crovellbio.com | China | 5964 | 58 |
| shandong perfect biotechnology co.ltd | +86-53169958659; +8618596095638 | sales@sdperfect.com | China | 294 | 58 |
| Henan Bao Enluo International TradeCo.,LTD | +86-17331933971 +86-17331933971 | deasea125996@gmail.com | China | 2503 | 58 |
| Nantong Guangyuan Chemicl Co,Ltd | +undefined17712220823 | admin@guyunchem.com | China | 616 | 58 |
| Hebei Jingbo New Material Technology Co., Ltd | +8619931165850 | hbjbtech@163.com | China | 1000 | 58 |
| Capot Chemical Co.,Ltd. | 571-85586718 +8613336195806 | sales@capotchem.com | China | 29797 | 60 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21695 | 55 |
| Hangzhou FandaChem Co.,Ltd. | 008657128800458; +8615858145714 | fandachem@gmail.com | China | 9350 | 55 |
| Hubei XinRunde Chemical Co., Ltd. | +8615102730682 | bruce@xrdchem.cn | CHINA | 566 | 55 |
View Lastest Price from Oxytetracycline manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2024-04-22 | Oxytetracycline
79-57-2
|
US $15.00 / KG | 100KG | 99% | 1000KG/day | Hebei Yime New Material Technology Co., Ltd. | |
 |
2024-03-16 | Oxytetracycline
79-57-2
|
US $0.00 / KG | 100g | 98%+ | 100kg | WUHAN CIRCLE POWDER TECHNOLOGY CO.,LTD | |
 |
2024-03-12 | Oxytetracycline
79-57-2
|
US $0.00 / g | 1g | 98% HPLC | 100kg | shandong perfect biotechnology co.ltd |
-

- Oxytetracycline
79-57-2
- US $15.00 / KG
- 99%
- Hebei Yime New Material Technology Co., Ltd.
-

- Oxytetracycline
79-57-2
- US $0.00 / KG
- 98%+
- WUHAN CIRCLE POWDER TECHNOLOGY CO.,LTD
-

- Oxytetracycline
79-57-2
- US $0.00 / g
- 98% HPLC
- shandong perfect biotechnology co.ltd