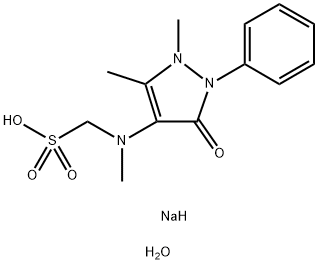
Metamizole Sodium Monohydrate
- CAS No.
- 5907-38-0
- Chemical Name:
- Metamizole Sodium Monohydrate
- Synonyms
- Analgin;METAMIZOLE;METAMIZOL;Dipirone;Sulpyrine;NORAMIDOPYRINI METHANOSULFONATE;[[(2,3-Dihydro-1,5-dimethyl-3-oxo-2-phenyl-1H-pyrazol)-4-yl]methylamino]methanesulfonic acid sodium salt;Dipyron;Novalgin;Sulpirine
- CBNumber:
- CB6715922
- Molecular Formula:
- C13H20N3NaO5S
- Molecular Weight:
- 353.37
- MDL Number:
- MFCD00150293
- MOL File:
- 5907-38-0.mol
- MSDS File:
- SDS
| Melting point | 221℃ |
|---|---|
| storage temp. | 2-8°C |
| solubility | H2O: soluble25mg/mL, clear |
| form | powder |
| color | white to beige |
| BRN | 3865381 |
| CAS DataBase Reference | 5907-38-0(CAS DataBase Reference) |
| FDA UNII | 6429L0L52Y |
Metamizole Sodium Monohydrate price More Price(10)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | PHR1800 | Metamizole sodium Pharmaceutical Secondary Standard; Certified Reference Material | 5907-38-0 | 100mg | $179 | 2024-03-01 | Buy |
| Sigma-Aldrich | 46232 | Dipyron monohydrate VETRANAL | 5907-38-0 | 250mg | $68 | 2024-03-01 | Buy |
| Sigma-Aldrich | SML1488 | Metamizole sodium ≥98% (HPLC) | 5907-38-0 | 10mg | $66.8 | 2024-03-01 | Buy |
| Sigma-Aldrich | SML1488 | Metamizole sodium ≥98% (HPLC) | 5907-38-0 | 50mg | $273 | 2024-03-01 | Buy |
| Sigma-Aldrich | M0600900 | Metamizole sodium European Pharmacopoeia (EP) Reference Standard | 5907-38-0 | m0600900 | $192 | 2024-03-01 | Buy |
Metamizole Sodium Monohydrate Chemical Properties,Uses,Production
Overview
Analgin[metamizole] is a painkiller, spasm reliever and fever reliever[1]. However, it is now either prescription or banned in most developed countries due to its potential for adverse events, including agranulocytosis. It belongs to the ampyrone sulfonate family of medicines.
Metamizole, a nonnarcotic analgesic, has been used to treat pain and fever for almost 90 years in some countries, while in others it is completely unknown or forgotten[2, 3]. The German company Hoechst AG synthesized it in 1920, and its mass production started in 1922. Metamizole remained freely available worldwide until the 1970s, when it was found that the drug poses a risk of causing agranulocytosis[4, 5]. Soon after that, metamizole was banned in the United States, Japan, Australia, and part of the European Union. However it is still widely used in some European countries, Turkey, Israel, India, Brazil, and Third World countries. Metamizole is available over-the-counter and remains one of the most popular analgesics in Bulgaria. Metamizole is a white or almost white crystalline powder, very soluble in water and soluble in alcohol. It is a well-established active substance and is the subject of a monograph in the European Pharmacopeia. Metamizole belongs to the following therapeutic groups: NSAIDs, nonnarcotic analgesics. ATC code is N02BB02[6, 7].
Indication
Metamizole has various effects including analgesic effect; antipyretic effect, anti-inflammatory effect and can reduce platelet aggregation. Metamizole is commonly used to treat postoperative pain, colic pain, cancer pain and migraine[8, 9]. In many parts of the world, including most countries in the European Union and Latin America, it is the most popular non-opioid first-line analgesic and is sometimes even available over-the-counter. A few countries however, including the United States, the United Kingdom, Sweden, and most recently India[4], have banned metamizole because health authorities judged the risk of agranulocytosis to outweigh the benefits[5–7]. In addition, it can also be used in relieving fever, and treatment of respiratory and gastroenteritic diseases. It’s also helpful and efficacious in the treatment of blood poisoning and pyogenic dermatitis. Indications include asthma, pneumonia[10].
Pharmacological effects and Mode of action
The pharmacological mechanism of action of metamizole is not precisely known. Similar to paracetamol/acetaminophen, its effects may result from the interference with prostaglandin synthesis through the inhibitory potential on different cyclooxygenase[COX] isoenzymes. Yet, interactions with the endogenous opioid, peroxidase, cannabinoid, and glutamate systems are also discussed. The pharmacologically active metabolites of metamizole, MAA and AA, did not inhibit COX activity in vitro like classical COX inhibitors, but instead redirected the prostaglandin synthesis, ruling out inhibition of COX through binding to its active site[11,12]. There is the assumption that metamizole might be an inhibitor of the COX-3 isoenzyme, thereby reducing prostaglandin synthesis in the dorsal horn of the spinal cord[11, 12, 14, 18].
At least it may also have antinociceptive actions that are independent of the ability to inhibit COX. One component of the non-COXmediated antinociceptive effects is a consequence of direct actions on nociceptive afferent elements in the periphery. In addition to the action upon peripheral tissues, the NSAIDs exert their antinociceptive effect by acting upon the CNS, specifically upon the spinal cord and the periaqueductal grey matter[PAG], which triggers descending inhibition of spinal nociception[12, 13]. Inhibition of prostaglandin synthesis also leaves more arachidonic acid available for the synthesis of endocannabinoids, which have an antinociceptive action in the spinal cord.
Overall, It is still debated whether the site of metamizole action is in the CNS or in the periphery or both. The mode of action of metamizole has not been fully elucidated yet. Metamizole exerts its analgesic effects through several action mechanisms, among which the best described are COX inhibition[11], delayed activation of the L-arginine/NO/cGMP/K+ channel pathway at the periphery and at the spinal cord[14, 15], activation of the descending inhibitory pain control system, interaction with the glutamatergic system[16], and release of endogenous opioid peptides[17].
Adverse reaction
Although used for more than 90 years, the risks and harms of metamizole are not well documented, and information on adverse events related to metamizole is scarce. There are no large randomized controlled trials or conclusive summaries of the existing literature. Three current Cochrane reviews on the effectiveness and safety of metamizole for acute postoperative pain[8], acute renal colic pain[9], and acute primary headaches[19] concluded that metamizole offers good short-term pain relief. In each of these systematic reviews, however, the number of included participants was too small and the authors did not conduct a meta-analysis of safety issues, but they did associate metamizole with somnolence, gastric discomfort and nausea.
Warning and precaution
In severe cases, metamizole can cause severe adverse reactions including congenital defect including aplasia cutis, craniofacial malformations[facial dysmorphism; choanal atresia], gastrointestinal malformations[esophageal atresia with or without tracheoesophageal fistula], omphalocele and abnormalities of the omphalomesenteric duct. Moreover, it may cause agranulocytosis, liver toxicity and hypothyroidism[20].
References
- 1. Brayfield, A, ed.[13 December 2013]. "Dipyrone". Martindale: The Complete Drug Reference. Pharmaceutical Press. Retrieved 19 April 2014.
- https://www.un.org/esa/coordination/CL12.pdf
Arellano, F, and J. A. Sacristán. "Metamizole: reassessment of its therapeutic role." European Journal of Clinical Pharmacology 38.6(1990]: 617-619. - Oricha, B. S., and M. B. Yauri. "Dipyrone[Novalgin Metamizole]: banned and unbanned: the dilemma of a commonly prescribed and over the counter analgesic, [letter]." Annals of African Medicine 2.2(2004]: 101-102.[5-8]
- Bhaumik S. India’s health ministry bans pioglitazone, metamizole, and flupentixol-melitracen. BMJ. 2013; 347: f4366. doi: 10.1136/bmj.f4366 PMID: 23833116
- Andersohn F, Konzen C, Garbe E. Systematic review: agranulocytosis induced by nonchemotherapy drugs. Ann Intern Med. 2007; 146: 657–665. PMID: 17470834
- Andrade SE, Martinez C, Walker AM. Comparative safety evaluation of non-narcotic analgesics. J Clin Epidemiol 1998; 51: 1357–1365. PMID: 10086830
- Heimpel H. Drug-induced agranulocytosis. Med Toxicol Adverse Drug Exp. 1988; 3: 449–462. PMID: 3063921
- Derry S, Faura C, Edwards J, McQuay HJ, Moore RA. Single-dose dipyrone for acute postoperative pain. Cochrane Database Syst Rev. 2013;CD003227. doi: 10.1002/14651858.CD003227.pub3 PMID: 24277663
- Edwards J, Meseguer F, Faura C, Moore RA, McQuay HJ. Single dose dipyrone for acute renal colic pain. Cochrane Database Syst Rev. 2011;CD003867. doi: 10.1002/14651858.CD003867 PMID: 12519613
- https://www.lybrate.com/medicine/analgin
- Chandrasekharan NV, Dai H, Roos KLT, et al. COX-3, a cyclooxygenase-1 variant inhibited by acetaminophen and other analgesic/antipyretic drugs: cloning, structure, and expression. Proc Natl Acad Sci U S A 2002; 99: 13926–13931.
- Vanegas, H, and H. G. Schaible. "Prostaglandins and cyclooxygenases [correction of cycloxygenases] in the spinal cord. " Progress in Neurobiology 64.4(2001]:327.
- Vanegas, H, and V. Tortorici. "Opioidergic effects of nonopioid analgesics on the central nervous system. " Cellular & Molecular Neurobiology 22.5-6(2002]: 655-661.
- Campos, Carmen, et al. "Regulation of cyclooxygenase activity by metamizol." European Journal of Pharmacology 378.3(1999]: 339-347.
- Aguirrebañuelos, P, and V. Granadossoto. "Evidence for a peripheral mechanism of action for the potentiation of the antinociceptive effect of morphine by dipyrone. " Journal of Pharmacological & Toxicological Methods 42.2(1999]:79-85.
- Beirith, A, et al. "Spinal and supraspinal antinociceptive action of dipyrone in formalin, capsaicin and glutamate tests. Study of the mechanism of action. " European Journal of Pharmacology345.3(1998]:233-245.
- Hernández-Delgadillo, G. P., and S. L. Cruz. "Endogenous opioids are involved in morphine and dipyrone analgesic potentiation in the tail flick test in rats." European Journal of Pharmacology 546.1(2006]:54-59.
- Soltesz, S, et al. "Parecoxib versus dipyrone[metamizole] for postoperative pain relief after hysterectomy : a prospective, single-centre, randomized, double-blind trial." Clinical Drug Investigation28.7(2008]:421-428.
- Ramacciotti AS, Soares BGO, Atallah AN. Dipyrone for acute primary headaches. Cochrane Database Syst Rev. 2007;CD004842. doi: 10.1002/14651858.CD004842.pub3 PMID: 17443558
- https://www.rxlist.com/tapazole-drug.htm#warnings_precautions
Chemical Properties
White or almost white, crystalline powder
Uses
antibacterial
Uses
Metamizol Sodium Monohydrate is used in methods of using Metamizole for treatment of Pyrexia in equines.
Definition
ChEBI: Sulpyrine is an organic molecular entity.
brand name
Diprofarn (Farmitalia, Societa Farmaceutici Italia, Italy); Novaldin (Sterling Winthrop).
General Description
Metamizole sodium is a nonsteroidal anti-inflammatory drug (NSAID). It is widely used as an analgesic and antipyretic agent.
Biochem/physiol Actions
Metamizole (Dipyrone) is a potent analgesic and antipyretic. Metamizole inhibits prostaglandin synthesis in both the periphery and the central nervous system. Metamizole may act as a prodrug. It is rapidly hydrolyzed to N-methyl-4-aminoantipyrine (MAA) and 4-aminoantipyrine (AA) which inhibit cyclo-oxygenase activity and can be further acylated with arachidonic acid forming compounds that bind to central and peripheral cannabinoid (CB1 and CB2) receptors and TRPV1 receptors. It is believed that these metabolites play a major role in metamizole activity. Metamizole has been withdrawn from clinical use in several countries because of a side-effect of agranulocytosis in some patients.
Clinical Use
Metamizol is the water
soluble sodium sulfonate of amidopyrine.
After oral application it is rapidly hydrolyzed
to 4-methylaminoantipyrine and metabolized to
various active metabolites . Metamizol has
strong analgesic, spasmolytic, and antipyretic,
but no anti-inflammatory properties. The exact
mechanism of action is unknown but may include
inhibition of prostaglandin synthesis. Inhibition
of both COX isoenzymes by metamizol
has been demonstrated only when very high
concentrations of this drug were used, thus questioning
the relevance of this activity.
COX inhibition may be induced by the
metabolites, but this is not yet systematically investigated.
Metamizol is used for the treatment
of medium to severe pain, often in combination
with opioids, for fever reduction and for
the treatment of colic pain. It is given by mouth
in doses of 500 mg up to 4 g daily, by intravenous
or by rectal application. Metamizol is relatively
free of acute side effects but in rare cases
may cause severe and life-threatening allergic reactions like agranulocytosis, allergic skin reactions,
and allergic shock. The compound is
not used in the UK, USA, and in Scandinavian
countries.
Metamizole Sodium Monohydrate Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Hebei Mojin Biotechnology Co., Ltd | +8613288715578 | sales@hbmojin.com | China | 12456 | 58 |
| shandong perfect biotechnology co.ltd | +86-53169958659; +8618596095638 | sales@sdperfect.com | China | 294 | 58 |
| Xiamen Wonderful Bio Technology Co., Ltd. | +8613043004613 | Sara@xmwonderfulbio.com | China | 305 | 58 |
| Sigma Audley | +86-18336680971 +86-18126314766 | nova@sh-teruiop.com | China | 525 | 58 |
| Shaanxi TNJONE Pharmaceutical Co., Ltd | +8618740459177 | sarah@tnjone.com | China | 893 | 58 |
| Capot Chemical Co.,Ltd. | 571-85586718 +8613336195806 | sales@capotchem.com | China | 29797 | 60 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21691 | 55 |
| Hubei XinRunde Chemical Co., Ltd. | +8615102730682 | bruce@xrdchem.cn | CHINA | 566 | 55 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418679 +86-18949832763 | info@tnjchem.com | China | 2989 | 55 |
| Hebei Guanlang Biotechnology Co., Ltd. | +86-19930503282 | alice@crovellbio.com | China | 8823 | 58 |
View Lastest Price from Metamizole Sodium Monohydrate manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2024-04-11 | Analgin
5907-38-0
|
US $0.00 / kg | 1kg | 99% | 20tons | Shaanxi TNJONE Pharmaceutical Co., Ltd | |
 |
2024-03-16 | Analgin
5907-38-0
|
US $0.00 / KG | 100g | 98%+ | 100kg | WUHAN CIRCLE POWDER TECHNOLOGY CO.,LTD | |
 |
2024-03-15 | Metamizole Sodium Monohydrate
5907-38-0
|
US $35.00-25.00 / kg | 1kg | 99.8% | 200tons/year | Sigma Audley |
-

- Analgin
5907-38-0
- US $0.00 / kg
- 99%
- Shaanxi TNJONE Pharmaceutical Co., Ltd
-

- Analgin
5907-38-0
- US $0.00 / KG
- 98%+
- WUHAN CIRCLE POWDER TECHNOLOGY CO.,LTD
-

- Metamizole Sodium Monohydrate
5907-38-0
- US $35.00-25.00 / kg
- 99.8%
- Sigma Audley