ChemicalBook >> CAS DataBase List >>3,4-DIHYDROXY-6-NITROBENZALDEHYDE
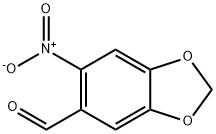
3,4-DIHYDROXY-6-NITROBENZALDEHYDE
- CAS No.
- 73635-75-3
- Chemical Name:
- 3,4-DIHYDROXY-6-NITROBENZALDEHYDE
- Synonyms
- 6-NITROPROTOCATECUALDEHYDE;6-NITROPROTOCATECHUALDEHYDE;3,4-DIHYDROXY-6-NITROBENZALDEHYDE;4,5-DIHYDROXY-2-NITROBENZALDEHYDE;2-nitro-4,5-dihydroxybenzaldehyde;Benzaldehyde, 4,5-dihydroxy-2-nitro-
- CBNumber:
- CB7210116
- Molecular Formula:
- C7H5NO5
- Molecular Weight:
- 183.12
- MDL Number:
- MFCD00016645
- MOL File:
- Mol file
Last updated:2023-04-23 13:52:06
| Melting point | 202-205°C |
|---|---|
| Boiling point | 417.0±45.0 °C(Predicted) |
| Density | 1.667±0.06 g/cm3(Predicted) |
| storage temp. | under inert gas (nitrogen or Argon) at 2-8°C |
| solubility | DMSO (Slightly), Ethyl Acetate (Slightly), Methanol (Slightly) |
| form | Solid |
| pka | 6.01±0.24(Predicted) |
| color | Dark Yellow to Very Dark Brown |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|---|---|
| Signal word | Warning |
| Hazard statements | H302-H315-H319-H332-H335 |
| Precautionary statements | P261-P280-P305+P351+P338 |
| Hazard Codes | Xi |
| Risk Statements | 36/37/38-43 |
| Safety Statements | 26-36-36/37 |
| Hazard Note | Irritant |
3,4-DIHYDROXY-6-NITROBENZALDEHYDE price
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| TRC | D453970 | 4,5-Dihydroxy-2-nitrobenzaldehyde | 73635-75-3 | 5g | $690 | 2021-12-16 | Buy |
| Biosynth Carbosynth | FD67691 | 3,4-Dihydroxy-6-nitrobenzaldehyde | 73635-75-3 | 25g | $625 | 2021-12-16 | Buy |
| Biosynth Carbosynth | FD67691 | 3,4-Dihydroxy-6-nitrobenzaldehyde | 73635-75-3 | 1g | $50 | 2021-12-16 | Buy |
| Biosynth Carbosynth | FD67691 | 3,4-Dihydroxy-6-nitrobenzaldehyde | 73635-75-3 | 2g | $84 | 2021-12-16 | Buy |
| Biosynth Carbosynth | FD67691 | 3,4-Dihydroxy-6-nitrobenzaldehyde | 73635-75-3 | 5G | $175 | 2021-12-16 | Buy |
3,4-DIHYDROXY-6-NITROBENZALDEHYDE Chemical Properties,Uses,Production
Uses
4,5-Dihydroxy-2-nitrobenzaldehyde is used as a reagent in the synthesis of 5,6-diacetoxyindole which is a stable eumelanin precursor. 4,5-Dihydroxy-2-nitrobenzaldehyde is also used in the preparation of 6-aminoisoproterenol which is an impurity of the non-selective beta-adrenergic agonist, Isoproterenol (I874200, HCl).
3,4-DIHYDROXY-6-NITROBENZALDEHYDE Preparation Products And Raw materials
Raw materials
1of2
Preparation Products
3,4-DIHYDROXY-6-NITROBENZALDEHYDE Suppliers
Global( 41)Suppliers
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Capot Chemical Co.,Ltd. | 571-85586718 +8613336195806 | sales@capotchem.com | China | 29797 | 60 |
| Hangzhou FandaChem Co.,Ltd. | 008657128800458; +8615858145714 | fandachem@gmail.com | China | 9348 | 55 |
| Dayang Chem (Hangzhou) Co.,Ltd. | 571-88938639 +8617705817739 | info@dycnchem.com | CHINA | 52867 | 58 |
| Hefei TNJ Chemical Industry Co.,Ltd. | 0551-65418684 +8618949823763 | sales@tnjchem.com | China | 25363 | 58 |
| Coresyn Pharmatech Co., Ltd. | +86-571-86626709 +86-18157142896 | cbc@coresyn.com | China | 9991 | 58 |
| GIHI CHEMICALS CO.,LIMITED | +8618058761490 | info@gihichemicals.com | China | 49999 | 58 |
| Shanghai Acmec Biochemical Technology Co., Ltd. | +undefined18621343501 | product@acmec-e.com | China | 33349 | 58 |
| Aladdin Scientific | +1-833-552-7181 | sales@aladdinsci.com | United States | 52927 | 58 |
| Amadis Chemical Company Limited | 571-89925085 | sales@amadischem.com | China | 131981 | 58 |
| Shanghai Sinch Parmaceuticals Tech. Co. Ltd. | +86-21-54098501 | sales@sinch.com.cn | China | 11605 | 64 |
4,5-DIHYDROXY-2-NITROBENZALDEHYDE
3,4-DIHYDROXY-6-NITROBENZALDEHYDE
6-NITROPROTOCATECHUALDEHYDE
6-NITROPROTOCATECUALDEHYDE
2-nitro-4,5-dihydroxybenzaldehyde
Benzaldehyde, 4,5-dihydroxy-2-nitro-
73635-75-3
Aromatic Aldehydes & Derivatives (substituted)