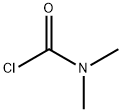
Dimethylcarbamoyl chloride
- CAS No.
- 79-44-7
- Chemical Name:
- Dimethylcarbamoyl chloride
- Synonyms
- Dimethylcarbamic chloride;dmcc;N,N-DIMETHYLCARBAMOYL CHLORIDE;DMCL;DIMETHYLCARBAMYL CHLORIDE;carbamyL;Dimethylcarbamoyl;N,N-DIMETHYLCARBAMYL CHLORIDE;DMCF;N,N-Dimethylaminoformyl
- CBNumber:
- CB7672462
- Molecular Formula:
- C3H6ClNO
- Molecular Weight:
- 107.54
- MDL Number:
- MFCD00000635
- MOL File:
- 79-44-7.mol
- MSDS File:
- SDS
| Melting point | −33 °C(lit.) |
|---|---|
| Boiling point | 167-168 °C775 mm Hg(lit.) |
| Density | 1.168 g/mL at 25 °C(lit.) |
| refractive index |
n |
| Flash point | 155 °F |
| solubility | Chloroform (Soluble), DMSO (Sparingly) |
| form | Liquid |
| pka | -1.85±0.70(Predicted) |
| Specific Gravity | 1.172 |
| color | Colorless to Yellow |
| Water Solubility | Decomposes |
| Sensitive | Moisture Sensitive |
| BRN | 878197 |
| Exposure limits | ACGIH: TWA 0.005 ppm (Skin) |
| Stability | Moisture Sensitive |
| CAS DataBase Reference | 79-44-7(CAS DataBase Reference) |
| EWG's Food Scores | 5 |
| FDA UNII | 5D54S95GT6 |
| Proposition 65 List | Dimethylcarbamoyl Chloride |
| IARC | 2A (Vol. 12, Sup 7, 71) 1999 |
| NIST Chemistry Reference | Carbamic chloride, dimethyl-(79-44-7) |
| EPA Substance Registry System | Dimethylcarbamoyl chloride (79-44-7) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS06,GHS08 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H302-H315-H319-H331-H335-H350 | |||||||||
| Precautionary statements | P201-P301+P312-P302+P352-P304+P340+P311-P305+P351+P338-P308+P313 | |||||||||
| Hazard Codes | T | |||||||||
| Risk Statements | 45-22-23-36/37/38 | |||||||||
| Safety Statements | 53-45-99 | |||||||||
| RIDADR | UN 2262 8/PG 2 | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | FD4200000 | |||||||||
| F | 9 | |||||||||
| TSCA | Yes | |||||||||
| HazardClass | 8 | |||||||||
| PackingGroup | II | |||||||||
| HS Code | 29241990 | |||||||||
| Toxicity | A designated animal carcinogen (IARC) and hazardous waste (EPA); a suspected carcinogen in humans. It is a liquid boiling at 165_x0003_C that is used primarily in the manufacture of drugs and pesticides. Dimethyl carbamoyl chloride is extremely toxic. | |||||||||
| NFPA 704 |
|
Dimethylcarbamoyl chloride price More Price(24)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | D152803 | Dimethylcarbamyl chloride 98% | 79-44-7 | 5g | $38.8 | 2024-03-01 | Buy |
| Sigma-Aldrich | D152803 | Dimethylcarbamyl chloride 98% | 79-44-7 | 100g | $44.7 | 2024-03-01 | Buy |
| TCI Chemical | D0695 | Dimethylcarbamoyl Chloride >98.0%(GC)(T) | 79-44-7 | 25g | $28 | 2024-03-01 | Buy |
| TCI Chemical | D0695 | Dimethylcarbamoyl Chloride >98.0%(GC)(T) | 79-44-7 | 500g | $120 | 2024-03-01 | Buy |
| Alfa Aesar | L03415 | Dimethylcarbamyl chloride, 96% | 79-44-7 | 25g | $30.65 | 2024-03-01 | Buy |
Dimethylcarbamoyl chloride Chemical Properties,Uses,Production
Overview
Dimethylcarbamoyl chloride is a useful intermediate in the production of pharmaceuticals, pesticides, and dyes. However, due to its high toxicity that may induce both acute effects and be of cancer risk, it is strictly limited for application[1].
Production
Dimethylcarbamoyl chloride has been produced since 1961(IARC 1999)[2]. In 2009, it was produced commercially by one manufac¬turer in Europe and two manufacturers in India[3] and was available from 17 suppliers worldwide, including 8 U.S. suppliers[4]. No data on U.S. imports or exports of dimethyl¬ carbamoyl chloride were found. Under the U.S. Environmental Pro¬tection Agency’s Toxic Substances Control Act Inventory Update Rule, production plus imports of dimethylcarbamoyl chloride totaled be¬ tween 10,000 and 500,000 lb in 1990; no other inventory update re¬ports were filed[5].
Dimethylcarbamoyl chloride can be manufactured by the following several methods[6-8]:
- The reaction between phosgene and dimethylamine(DMA), which is the earliest method[6];
- The reaction between phosgene with trimethylamine[7];
- The reaction between dimethylamine chloride with carbon monoxide under pressure, room temperature and using palladium as catalyst[8].
Physiochemical properties
Dimethylcarbamoyl chloride appears as a clear liquid at room temperature with a pungent odor and a tear-penetrating effect, which decomposes rapidly in water[1-2].
Dimethylcarbamoyl chloride is similar with an acid chloride whose chlorine atom can be exchanged for other nucleophiles. Therefore, it is capable of reacting with alcohols, phenols and oximes to the corresponding N, N-dimethylcarbamates, with thiols to thiolourethanes, with amines and hydroxylamine to substituted ureas, and with imidazoles and triazoles to carbamoylazoles[9].
Applications
Dimethylcarbamoyl chloride is used as an intermediate in the production of pharmaceuticals, pesticides, and dyes[10]. It is a reagent being capable of transferring a dimethylcarbamoyl group to alcoholic or phenolic hydroxyl groups forming dimethyl carbamates with pharmacological or pesticidal activities[11]. Dimethylcarbamoyl Chloride is also a reagent used in the induction of apoptosis in human lung adenocarcinoma by T-type calcium channel antagonist[12].
Dimethylcarbamoyl chloride is used as the starting material for the insecticide class of the dimethyl carbamates which act as inhibitors of acetylcholinesterase such as dimetilane and the related compounds isolane, pirimicarb and triazamate[13,14].
Dimethylcarbamoyl chloride can be used for the manufacturing of the quaternary ammonium compounds neostigmine, which finds pharmaceutical applications as acetylcholinesterase inhibitors[15].
Dimethylcarbamoyl chloride is also used in the synthesis of the benzodiazepine camazepam[16].
Warning and Risk
Dimethylcarbamoyl chloride can cause acute effect if not used properly. It is also a potential carcinogenic regent due to its high toxicity[17-20].
Acute effect
Workers exposing to dimethylcarbamoyl chloride can suffer from both eye irritation and liver disturbance. Acute inhalation exposure to dimethylcarbamoyl chloride has been found to result in damaged mucous membranes of the nose, throat, and lungs and cause difficulty in breathing in rats. Rats and rabbits experiments have also demonstrated that acute dermal exposure can cause skin irritation as well as conjunctivitis and keratitis in eyes. In addition, acute exposure of rats has also demonstrated dimethylcarbamoyl chloride to have high acute toxicity via inhalation and moderate acute toxicity via ingestion[17].
Cancer Risk
There are inadequate data on the carcinogenic effects of dimethylcarbamoyl chloride in humans[18, 21,22]. However, cancer risk of dimethylcarbamoyl chloride has been demonstrated in the rats and mice experiments. Inhalation exposure has been found to induce nasal tract carcinomas in rats and male hamsters. Skin tumors have also been observed among dermally exposed mice. Moreover, local sarcomas have been observed following subcutaneous injection in mice[21,22].
References
- https://ntp.niehs.nih.gov/ntp/roc/content/profiles/dimethylcarbamoylchloride.pdf
- http://www.allfordrugs.com/tag/dimethylcarbamoyl-chloride/
- SRI. 2009. Directory of Chemical Producers. Menlo Park, CA: SRI Consulting. Database edition. Last accessed: 4/22/09.
- ChemSources. 2009. Chem Sources Chemical Search. Chemical Sources International. http://www. chemsources.com/chemonline.html and search on dimethylcarbamoyl chloride. Last accessed: 5/09.
- EPA. 2004. Non-confidential IUR Production Volume Information. U.S. Environmental Protection Agency. http://www.epa.gov/oppt/iur/tools/data/2002-vol.html and search on CAS number. Last accessed: 4/21/05.
- G. Karimipour; S. Kowkabi; A. Naghiha[2015], "New aminoporphyrins bearing urea derivative substituents: synthesis, characterization, antibacterial and antifungal activity"[in German], Braz. Arch. Biol. Technol. 58[3], doi:10.1590/S1516-891320500024
- H. Babad; A.G. Zeiler[1973], "Chemistry of Phosgene"[in German], Chem. Rev. 73[1]: pp. 75–91, doi:10.1021/cr60281a005
- T. Saegusa; T. Tsuda; Y. Isegawa[1971], "Carbamoyl chloride formation from chloramine and carbon monoxide"[in German], J. Org. Chem. 36[6]: pp. 858–860, doi:10.1021/jo00805a033
- C.B. Kreutzberger, R.A. Olofson[2007-02-01]. "Dimethylcarbamoyl Chloride"[in German]. John Wiley&Sons, Ltd. Retrieved 2016-09-27.
- https://www.epa.gov/sites/production/files/2016-09/documents/dimethylcarbamoyl-chloride.pdf
- R.P. Pohanish[2011][in German], Sittig’s Handbook of Toxic and Hazardous Chemicals and Carcinogens, 6th Edition, Amsterdam: Elsevier, pp. 1045–1047, ISBN 978-1437778694
- https://www.trc-canada.com/product-detail/?D471295
- T. Grauer, H. Urwyler, "Production of 1-N, N-dimethylcarbamoyl-5-methyl-3-N, N-dimethyl-carbamoyl-oxy-pyrazole"
- J.A. Aeschlimann; M. Reinert[1931], "Pharmacological action of some analogues of physostigmine"[in German], J. Pharmacol. Exp. Ther. 43[3]: pp. 413–444
- J.A. Aeschlimann, "Disubstituted carbamic acid esters of phenols containing a basic constituent"
- "Verfahren zur Herstellung des 3-N, N-Dimethylcarbamoyl-oxy-1-methyl-5-phenyl-7-chlor-1,3-dihydro-2H-1,4-benzodiazepin-2-on"
- https://www.epa.gov/sites/production/files/2016-09/documents/dimethylcarbamoyl-chloride.pdf
- International Agency for Research on Cancer[IARC]. IARC Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Man: Some Carbamates, Thiocarbamates and Carbazides. Volume 12. World Health Organization, Lyon. 1976.
- U.S. Department of Health and Human Services. Hazardous Substances Data Bank[HSDB, online database]. National Toxicology Information Program, National Library of Medicine, Bethesda, MD. 1993.
- U.S. Department of Health and Human Services. Registry of Toxic Effects of Chemical Substances[RTECS, online database]. National Toxicology Information Program, National Library of Medicine, Bethesda, MD. 1993.
- International Agency for Research on Cancer[IARC]. IARC Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans: Overall Evaluations of Carcinogenicity: An Updating of IARC Monographs Volumes 1 to 42. Supplement 7. World Health Organization, Lyon. 1987.
- U.S. Department of Health and Human Services[DHHS]. The 8th Report on Carcinogens. 1998 Summary. Public Health Service, National Toxicology Program. Research Triangle Park, NC. 1998.
Description
DMCC is a liquid. Molecularweight= 107.55; Specific gravity (H2O:1) 5 1.2; Boilingpoint= 165℃; Freezing/Melting point = 2 33℃; Vaporpressure = 2.5 mmHg at 25℃; Flash point = 68.3℃.Hazard Identification (based on NFPA-704 M RatingSystem): Health 3, Flammability 2, Reactivity 1 . Reactivewith water; decomposes.
Chemical Properties
Clear colorless liquid
Uses
Dimethylcarbamyl chloride is used to make dyes and pharmaceuticals.
Uses
As a chemical intermediate in the manufacture of carbamate drugs and pesticides
Uses
Dimethylcarbamoyl chloride has been used primarily as a chemical intermediate in the production of dyes, pharmaceuticals, pesticides, and rocket fuel (IARC 1999, HSDB 2009).
Definition
ChEBI: Dimethylcarbamoyl chloride is a halide. It is functionally related to a carbamic acid.
General Description
A colorless to yellow liquid with a pungent odor. Burns to skin, eyes and mucous membranes. A lachrymator. Used to make dyes and pharmaceuticals.
Air & Water Reactions
Reacts with water or moisture in the air to form hydrochloric acid and dimethylcarbamic acid.
Reactivity Profile
Dimethylcarbamoyl chloride is water reactive. Incompatible with strong oxidizing agents, alcohols, bases (including amines). May react vigorously or explosively if mixed with diisopropyl ether or other ethers in the presence of trace amounts of metal salts [J. Haz. Mat., 1981, 4, 291]. Gives toxic fumes of NOx and HCl when burned [USCG, 1999].
Health Hazard
Material is extremely destructive to the mucous membranes, upper respiratory tract, eyes, and skin. Symptoms of exposure include burning sensation, coughing, wheezing, laryngitis, shortness of breath, headache, nausea, and vomiting.
Fire Hazard
Special Hazards of Combustion Products: Toxic fumes of NO x and HCl
Toxicology
The LD50 is 1170 mg/kg upon oral administration to rats and 350 mg/kg upon intraperitoneal administration to mice. The compound is irritating to the skin and eyes. All the test animals survived an 8-min inhalation test in an atmosphere saturated with vapors of the substance at 20℃. Longer exposure times led to death. No sensitization was observed in animal experiments.
Safety Profile
Confirmed carcinogen with experimental carcinogenic, neoplastigenic, and tumorigenic data. Poison by intraperitoneal route. Moderately toxic by inhalation and ingestion. Human mutation data reported. Can cause skin and paplllary tumors by skin contact, and squamous cell carcinoma by inhalation. Will react with water or steam to produce toxic and corrosive fumes. A powerful lachrymator. When heated to decomposition it emits very toxic fumes of Cland NOx. See also CHLORIDES.
Potential Exposure
AgriculturalChemical; Tumorigen, Mutagen. DMCC is used as a chemical intermediate in the production of pharmaceuticals, pesticides, rocket fuel, and dye synthesis. Human exposure islimited to, but not restricted to, chemical workers, pesticideformulators, dye makers, and pharmaceutical workers.DMCC has been found at levels up to 6 ppm during the production of phthaloyl chlorides. It is possible that levels ofexposure might be higher in facilities in which the chemicalis used for further synthesis. When DMCC is used as a dyeintermediate, exposure can occur from the amount of residue in the product and its ability to migrate.
First aid
If this chemical gets into the eyes, remove anycontact lenses at once and irrigate immediately for at least15 min, occasionally lifting upper and lower lids. Seek medical attention immediately. If this chemical contacts theskin, remove contaminated clothing and wash immediatelywith soap and water. Seek medical attention immediately. Ifthis chemical has been inhaled, remove from exposure,begin rescue breathing (using universal precautions, including resuscitation mask) if breathing has stopped and CPR ifheart action has stopped. Transfer promptly to a medicalfacility. When this chemical has been swallowed, get medical attention. If victim is conscious, administer water ormilk. Do not induce vomiting
Carcinogenicity
Dimethylcarbamoyl chloride is reasonably anticipated to be a human carcinogen based on sufficient evidence of carcinogenicity from studies in experimental animals.
storage
(1) Color Code—White: Corrosive or ContactHazard; Store separately in a corrosion-resistant location.(2) Color Code—Blue: Health Hazard/Poison: Store in asecure poison location. Prior to working with DMCC youshould be trained on its proper handling and storage. Storein tightly closed containers in a cool, well-ventilated areaaway from strong oxidizers, strong acids, strong bases, andmoisture, since violent reactions can occur. Metal containers involving the transfer of this chemical should begrounded and bonded. Where possible, automatically pumpliquid from drums or other storage containers to processcontainers. Drums must be equipped with self-closingvalves, pressure vacuum bungs, and flame arresters. Useonly nonsparking tools and equipment, especially whenopening and closing containers of this chemical. Sources ofignition, such as smoking and open flames, are prohibitedwhere this chemical is used, handled, or stored in a mannerthat could create a potential fire or explosion hazard.Wherever this chemical is used, handled, manufactured, orstored, use explosion-proof electrical equipment and fittings. A regulated, marked area should be established wherethis chemical is handled, used, or stored in compliance withOSHA Standard 1910.1045.
Shipping
This compound requires a shipping label of“CORROSIVE.” It falls in Hazard Class 8 and PackingGroup II.
Purification Methods
It must be distilled under high vacuum to avoid decomposition. [Beilstein 4 IV 224.]
Incompatibilities
Strong oxidizers; strong acids; strongbases. Reacts with water (rapidly hydrolyzes to carbondioxide, dimethylamine, dimethylcarbamic acid, and hydrochloric acid). Attacks metals in the presence of moisture.
Dimethylcarbamoyl chloride Preparation Products And Raw materials
Raw materials
1of2
Preparation Products
1of5
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Career Henan Chemica Co | +86-0371-86658258 15093356674; | laboratory@coreychem.com | China | 30255 | 58 |
| Hangzhou MolCore BioPharmatech Co.,Ltd. | +86-057181025280; +8617767106207 | sales@molcore.com | China | 49739 | 58 |
| Capot Chemical Co.,Ltd. | 571-85586718 +8613336195806 | sales@capotchem.com | China | 29797 | 60 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21691 | 55 |
| Nanjing ChemLin Chemical Industry Co., Ltd. | 025-83697070 | product@chemlin.com.cn | CHINA | 3012 | 60 |
| career henan chemical co | +86-0371-86658258 | sales@coreychem.com | China | 29914 | 58 |
| Hubei Jusheng Technology Co.,Ltd. | 18871490254 | linda@hubeijusheng.com | CHINA | 28180 | 58 |
| Hubei xin bonus chemical co. LTD | 86-13657291602 | linda@hubeijusheng.com | CHINA | 22968 | 58 |
| Shandong chuangyingchemical Co., Ltd. | 18853181302 | sale@chuangyingchem.com | CHINA | 5909 | 58 |
| Changzhou Carman Chemical Co., Ltd. | 15061933924 | CHINA | 514 | 58 |
View Lastest Price from Dimethylcarbamoyl chloride manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2023-01-31 | Dimethylcarbamoyl chloride
79-44-7
|
US $100.00 / kg | 1kg | 99% | 50000KG/month | Hebei Mojin Biotechnology Co., Ltd | |
 |
2020-02-21 | Dimethylcarbamoyl chloride
79-44-7
|
US $8.00 / KG | 1g | >98% HPLC | kg--ton | Career Henan Chemical Co | |
 |
2019-08-30 | Dimethylcarbamoyl chloride
79-44-7
|
US $1.00-1.00 / KG | 1KG | 98% | 1kg;10kg;100kg | Career Henan Chemical Co |
-

- Dimethylcarbamoyl chloride
79-44-7
- US $100.00 / kg
- 99%
- Hebei Mojin Biotechnology Co., Ltd
-

- Dimethylcarbamoyl chloride
79-44-7
- US $8.00 / KG
- >98% HPLC
- Career Henan Chemical Co
-

- Dimethylcarbamoyl chloride
79-44-7
- US $1.00-1.00 / KG
- 98%
- Career Henan Chemical Co