ベンゾイルヒドロペルオキシド 化学特性,用途語,生産方法
解説
過安息香酸.過酸化ベンゾイルを0 ℃ でナトリウムメトキシドで分解させるか,無水安息香酸を過酸化水素で酸化すると得られる.無色の結晶.融点41~43 ℃,沸点97~110 ℃(1.7~2.0 kPa).エーテル,クロロホルムなどの有機溶剤に可溶.強い酸化剤で,結合を容易にエポキシ化するので,合成反応や二重結合の定量に使われる.
化学的特性
Volatile solid with a pungent odor; sublimes;mp 41–43°C (105.8–109°F); bp 100–105°C(212–221°F) at 15 torr (partially decomposes); slightly soluble in water but mixesreadily with most organic solvents.
使用
Perbenzoic acid is used to convert ethylenic compounds into oxides; in analysis of unsatd compounds, to determine the number of double bonds.
製法
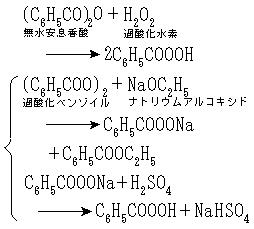
製法は無水安息香酸と過酸化水素との反応,または過酸化ベンゾイルにナトリウムアルコキシドを作用させて得られるナトリウム塩を冷希硫酸で分解する方法による。
強い酸化作用をもつため酸化剤として用いられるが,とくにエチレン結合のエポキシ化に有効である。
健康ハザード
The toxicity of peroxybenzoic acid is verylow on animals. In humans it is almostnontoxic. Peroxybenzoic acid caused skintumor in mice on prolonged contact (NIOSH1986). Its tumorigenic action was lower thanthat of peroxyacetic acid. Its carcinogenicityin humans is unknown.
火災危険
The presence of the peroxide functional
group renders its strong oxidizing properties similar to those of other peroxy compounds. However, this compound is not
hazardous like peroxyacetic or peroxyformic
acids. It is not shock sensitive. Violent
decomposition may occur only at high temperatures and/or in conjunction with certain organic contaminants that are easily
oxidizable. Although there is no report
of its explosion, general safety measures
for handling peroxy compounds should be
followed.
安全性プロファイル
Moderately irritating to skin, eyes, and mucous membranes by ingestion and inhalation. Questionable carcinogen with experimental tumorigenic data by skin contact. A dangerous fire hazard when exposed to heat, flame, or reducing materials. A powerful oxid
純化方法
Crystallise the peracid from *benzene or pet ether. It sublimes readily and is steam volatile. It is soluble in CHCl3, CCl4 and Et2O. [Braun Org Synth Coll Vol I 431 1941.] EXPLOSIVE.
ベンゾイルヒドロペルオキシド 上流と下流の製品情報
原材料
準備製品