クメン 化学特性,用途語,生産方法
外観
無色~ほとんど無色, 澄明の液体
性質
クメンは、常温常圧で特異臭を持つ無色の液体です。融点は-96℃、沸点は152℃、比重は0.86です。エタノール、エチルエーテル、アセトン、ベンゼン、石油エーテル、四塩化炭素に可溶で、水には非常に溶けにくい性質を有します。
労働安全衛生法において「危険物・引火性のもの」、「名称等を表示・通知すべき有害物質」に指定されています。PRTR法で第1種指定化学物質、消防法で「第4類引火性液体」、「第二石油類非水溶性液体」に指定されています。
芳香族炭化水素であるため、他の炭化水素と同様に発がん性の可能性が指摘されています。また、高温や加圧下で分解すると爆発する危険性があります。このため、取り扱いには注意が必要です。
溶解性
エタノール及びアセトンに溶け、水にほとんど溶けない。
解説
クメン,沸点153 ℃.d420"0.8620,nD25"1.489.各種有機溶剤とまざる.クメン法によるフェノール,アセトン製造の中間製品として利用される.
森北出版「化学辞典(第2版)
用途
有機合成原料。
用途
キュメンは芳香族炭化水素で、キュメン法によるフェノールとアセトン製造の原料として使われています。他に、合成中間体として使われています。
用途
フェノール?アセトン原料;有機合成原料(フェノール,アセトン),ラッカー希釈剤
製法
はじめクミン酸(p-イソプロピル安息香酸)を石灰と蒸留して得られたのでこの名称がある.現在は,ベンゼンにプロペンをフリーデル-クラフツ反応させて製造されるクメン.
合成
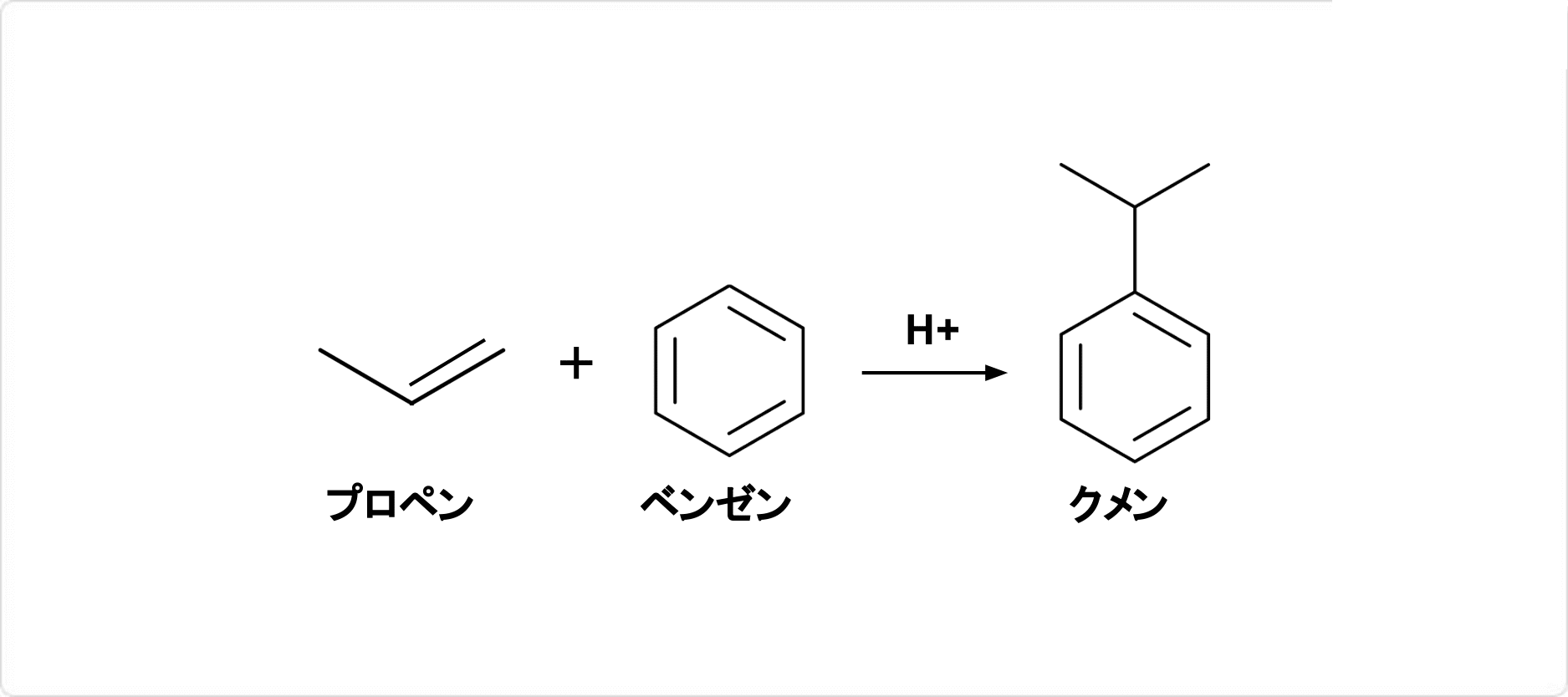
図3. フリーデル・クラフツ反応によるクメンの合成
クメンの合成ではフリーデル・クラフツ反応が利用されます。フリーデル・クラフツ反応は、有機化合物の芳香族置換反応の一種であり、ハロゲン化アルキルやアシルハライドなどのエレクトロフィルを芳香族環に付加する反応です。具体的なクメンの合成方法は以下の通りです。
プロピレンとベンゼンを混合し、ルイス酸である塩化アルミニウムを触媒として加熱します。次に、塩化アルミニウムがプロピレンに結合し、プロピレンカチオンが生成します。ベンゼンがプロピレンカチオンに攻撃され、クメンが生成します。この反応混合物を水で加水分解し、クメンを取り出します。
この反応では、塩化アルミニウムがプロピレンカチオンを安定化させ、芳香族環に付加させることができます。フリーデル・クラフツ反応によるクメンの合成は、工業的な生産方法として広く利用されています。
参考文献
説明
Cumene is a common name for isopropylbenzene, an organic
compound. Cumene is a volatile colorless liquid at room
temperature with a characteristic sharp, penetrating, aromatic
odor. It is insoluble in water but is soluble in alcohol and many
other organic solvents. Cumene is structurally a member of the
alkyl aromatic family of hydrocarbons, which also includes
toluene (methylbenzene) and ethylbenzene.Cumene can be
found in crude oil, refined fuels, and is a part of processed highoctane
gasoline.
Cumene is manufactured by reacting benzene with
propylene at elevated temperature and pressure in the presence
of a catalyst. It is considered an environmental pollutant
because it is a natural component of petroleum and is present
in tobacco smoke. Cumene vapor can be absorbed by the respiratory tract. Sufficiently high levels of exposure to cumene
causes central nervous system (CNS) depression leading to
death, internal bleeding of numerous organs, as well as irritation
of the eyes and respiratory system, skin, and mucous
membranes. Cumene is a high production volume chemical.
化学的特性
cumene is oxidized to its hydroperoxide, which is used to produce propene oxide. The alcohol produced is subsequently converted back to cumene over a copper-chromium oxide catalyst to be reused in the process. The advantage of this process is that cumene is easier to hydroperoxidate (more stable).
物理的性質
Colorless liquid with an aromatic odor. Experimentally determined detection and recognition odor
threshold concentrations were 40 μg/m
3 (8 ppb
v) and 230 μg/m
3 (47 ppb
v), respectively (Hellman
and Small, 1974). The taste threshold concentration in water is 60 ppb (Young et al., 1996).
使用
Around 98% of cumene is used in the production of phenol
and its coproduct, acetone, using cumene hydroperoxide as
chemical intermediate. However, the demand for cumene is
largely dependent on the performance of phenol’s derivatives,
which have resulted in healthy growth rates in demand for
cumene. It is also used as a starting material in the production
of acetophenone, α-methylstyrene, diisopropylbenzene, and
dicumylperoxide. Cumene is used as a thinner for paints,
lacquers, and enamels. It is also used in the manufacture of
acetophenone, methylstyrene, and other chemicals commonly
found in home cleaning products. Minor uses of cumene
include as a constituent of some petroleum-based solvents,
such as naphtha; in gasoline blending diesel fuel and highoctane
aviation fuel; and as a raw material for peroxides and
oxidation catalysts such as polymerization catalysts for acrylic
and polyester-type resins. It is also a good solvent for fats and
resins and has been suggested as a replacement for benzene in
many of its industrial applications.
一般的な説明
Pharmaceutical secondary standards for application in quality control, provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards.
空気と水の反応
Flammable. Insoluble in water.
反応プロフィール
Mixing CUMENE in equal molar portions with any of the following substances in a closed container caused the temperature and pressure to increase: chlorosulfonic acid, nitric acid, oleum, NFPA 1991.
危険性
Toxic by ingestion, inhalation, and skin
absorption; a narcotic. Moderate fire risk. Eye, skin,
and upper respiratory tract irritant, and central nervous system impairment. Possible carcinogen.
健康ハザード
Narcotic action with long-lasting effects; depressant to central nervous system. Acute (short-term) inhalation exposure to cumene may cause headaches, dizziness, drowsiness, slight incoordination, and unconsciousness in humans. Cumene has a potent central nervous system (CNS) depressant action characterized by a slow induction period and long duration of narcotic effects in animals. Cumene is a skin and eye irritant. No information is available on the chronic (long-term), reproductive, developmental, or carcinogenic effects of cumene in humans. Animal studies have reported increased liver, kidney, and adrenal weights from inhalation exposure to cumene. EPA has classified cumene as a Group D, not classifiable as to human carcinogenicity.
火災危険
HIGHLY FLAMMABLE: Will be easily ignited by heat, sparks or flames. Vapors may form explosive mixtures with air. Vapors may travel to source of ignition and flash back. Most vapors are heavier than air. They will spread along ground and collect in low or confined areas (sewers, basements, tanks). Vapor explosion hazard indoors, outdoors or in sewers. Runoff to sewer may create fire or explosion hazard. Containers may explode when heated. Many liquids are lighter than water.
使用用途
1. プラスチック原料
クメンの実需の大部分はフェノールであり、クメンを原料として合成されたフェノールから、フェノール樹脂が製造されます。フェノール樹脂のビスフェノールAはポリカーボネートやエポキシ樹脂の原料として消費されます。
2. 燃料
燃料を燃焼させた際に得られるエネルギーを増加させる目的で、クメンを添加した航空向けの高オクタン価燃料が用いられています。
3. 溶剤
塗料やラッカー、シンナーの溶剤の一部に、少量のクメンが添加されている場合があります。
4. 過酸化物
クメンを酸化して得られるクメンヒドロペルオキシドのような過酸化物は、強力な酸化剤であり、還元性物質や可燃性物質と容易に反応して酸化させる作用を持っています。また、過酸化物を分解することでラジカルが発生し、高分子合成のラジカル重合における重合開始剤として使用されることがあります。
5. その他の原料
医薬品や香料の原料としてクメンが使用されています。
化学反応性
Reactivity with Water: No reaction; Reactivity with Common Materials: No reaction; Stability During Transport: Stable; Neutralizing Agents for Acids and Caustics: Not pertinent; Polymerization: Not pertinent; Inhibitor of Polymerization: Not pertinent.
安全性プロファイル
Moderately toxic by
ingestion. Mdly toxic by inhalation and skincontact. Human systemic effects by
inhalation: an antipsychotic, unspecified
changes in the sense of smell and respiratory
system. An eye and skin irritant. Potential
narcotic action. Central nervous system
depressant. There is no apparent difference
between the toxicity of natural cumene and
that derived from petroleum. See also
BENZENE and TOLUENE. Flammable
liquid when exposed to heat or flame; can
react with oxidizing materials. Violent
reaction with HNO3, oleum, chlorosulfonic
acid. To fight fKe, use foam, CO2, dry
chemical.
職業ばく露
Cumene is used primarily in the manufacture acetone and phenol which are widely used as solvents for paints, laquers, and varnishes and to make plastics. Cumene is used in gasoline blending and as a high-octane gasoline component. It is also found as a component in tobacco smoke.
発がん性
Cumene was not a developmental toxicant
in either rats or rabbits after exposure to levels
(1200ppm and 2300ppm, respectively) associated
with maternal toxicity.9 Most genotoxic
tests with cumene have been negative.
The LD50 for penetration of rabbit skin
was 12.3 ml/kg after 14 days.4 Contact of the
liquid with the skin causes erythema and irritation.
11 Eye contamination may produce conjunctival
irritation.
It generally is agreed that cumene has no
damaging effect on the hematopoietic system,
despite its chemical similarity to benzene.5
Furthermore, cumene is not anticipated to be
a significant carcinogenic hazard because it is
metabolically similar to toluene, a substance
that showed no carcinogenic activity in 2-year
inhalation studies.
輸送方法
UN1918 Cumene, Hazard Class: 3; Labels: 3-Flammable liquid
不和合性
Vapor may form explosive mixture with air. Incompatible with oxidizers (chlorates, nitrates, peroxides, permanganates, perchlorates, chlorine, bromine, 942 Cumene fluorine, etc.); contact may cause fires or explosions. Keep away from alkaline materials, strong bases, strong acids, oxoacids, epoxides. Air contact forms cumene hydroperoxide. Attacks rubber. May accumulate static electrical charges, and may cause ignition of its vapors.
廃棄物の処理
Dissolve or mix the material with a combustible solvent and burn in a chemical incinerator equipped with an afterburner and scrubber. All federal, state, and local environmental regulations must be observed.
クメン 上流と下流の製品情報
原材料
準備製品