ジエチレングリコールジエチルエーテル 化学特性,用途語,生産方法
外観
無色~ほとんど無色, 澄明の液体
定義
本品は、ジエチレングリコール(*)のジエチルエーテルで、次の化学式で表される。参照表示名称:ジエチレングリコール
溶解性
20℃水に易溶。アルコール, エーテル, アセトンに易溶。水、エタノール及びアセトンに極めて溶けやすい。
用途
溶剤(有機合成反 用、ラッカー用)
用途
ニトロセルロース、樹脂、塗料の溶剤、高沸点の有機合成用溶媒
用途
溶剤。
化粧品の成分用途
溶剤
説明
Diethylene glycol diethyl ether, or 2-Ethoxyethyl ether, is an organic solvent used to study the activities of enzymes in aqueous organic mixtures. Diethylene glycol diethyl ether is a solvent for reactions performed at higher temperatures. It is involved in the preparation of nitrocellulose, resins, and adhesives. It is a scrubbing medium to absorb carbonyl sulfide (COS), an impurity in petroleum refineries. It could be used as an absorbent for CO2. This absorbent has a high boiling point (462 K, in SciFinder), indicating that energy for absorbent regeneration and loss were saved. In addition, this absorbent is chemically and thermally stable, non-corrosive, and has low density and viscosity. It is rich in philic-CO2 groups, which makes Diethylene glycol diethyl ether more effective for CO2 capture. Li et al. found that carbonyl and ether groups in the absorbents improved CO2 solubility, and the carbonyl group was more effective than the ether group[1].
化学的特性
colourless liquid
使用
Solvent; high boiling reaction medium.
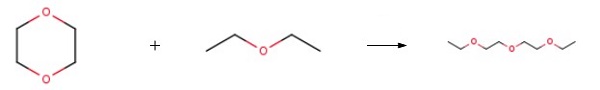
製造方法
2-Ethoxyethyl ether could be synthesized by reacting 1, 4-dioxane with diethyl ether. ZSM-5-type zeolite was used as the catalyst. This reaction is performed in an autoclave.
定義
ChEBI: A polyether that consists of undecane in which the carbon atoms at positions 3, 6 and 9 are replaced by oxygen atoms.
一般的な説明
Clear colorless viscous liquid.
空気と水の反応
Highly flammable. May be sensitive to prolonged exposure to air. Can form explosive peroxides. Vapor-air mixtures are explosive above the flash point. Water soluble.
反応プロフィール
2-Ethoxyethyl ether is incompatible with strong acids. 2-Ethoxyethyl ether is also incompatible with strong oxidizing agents.
火災危険
2-Ethoxyethyl ether is combustible.
安全性プロファイル
Moderately toxic by
ingestion. An experimental teratogen. Other
experimental reproductive effects. An eye
irritant. Flammable when exposed to heat or
flame. When heated to decomposition it
emits acrid smoke and irritating fumes. See
also GLYCOL ETHERS.
純化方法
Dry the ether with MgSO4, then CaH2 or LiAlH4, under N2. If sodium is used, the ether should be redistilled alone to remove any products which may be formed by the action of sodium on the ether. As a preliminary purification, the crude ether (2L) can be refluxed for 12hours with 25mL of conc HCl in 200mL of water, under reduced pressure, with slow passage of N2 to remove aldehydes and other volatile substances. After cooling, add sufficient solid KOH pellets (slowly and with shaking until no more dissolves) to give two liquid phases. The upper of these is decanted, dried with fresh KOH pellets, decanted, then refluxed over, and distilled from sodium. It can be passed through (alkaline) alumina prior to purification. [Beilstein 1 IV 2394.]
ジエチレングリコールジエチルエーテル 上流と下流の製品情報
原材料
準備製品